Cancer Res Treat.
2006 Jun;38(3):139-143.
Occludin Expression in Brain Tumors and its Relevance to Peritumoral Edema and Survival
- Affiliations
-
- 1Department of Neurosurgery, Hanyang University College of Medicine, Seoul, Korea. kch5142@hanyang.ac.kr
Abstract
-
PURPOSE: Peritumoral brain edema (PTBE) is a serious causative factor that contributes the morbidity or mortality of brain tumors. The development of PTBE is influenced by many factors, including such tight junction proteins as occludin. We evaluated the PTBE volume and survival time with respect to the occludin expression in various pathological types of brain tumors.
MATERIALS AND METHODS
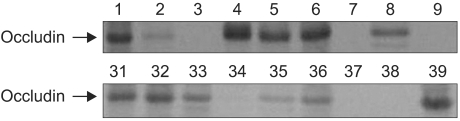
Fresh-frozen specimens from sixty patients who had brain tumors were obtained during surgery and the tumors were confirmed pathologically. The occludin expression was investigated by Western blot analysis. The PTBE volume was measured by using preoperative magnetic resonance (MR) imaging, and the survival time in each patient was estimated retrospectively.
RESULTS
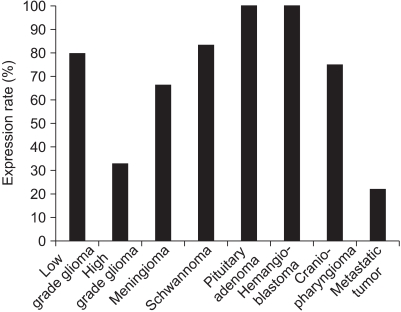
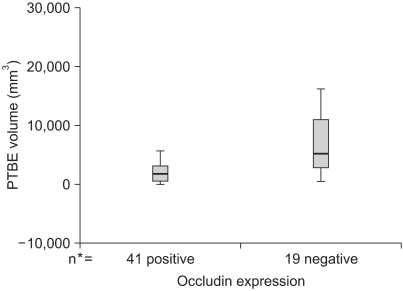
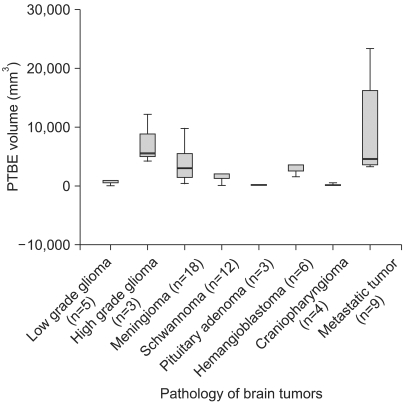
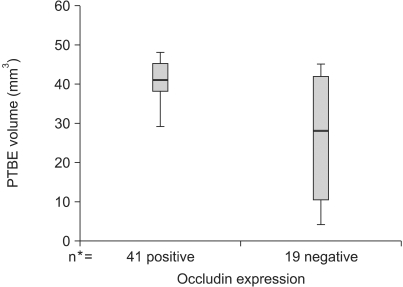
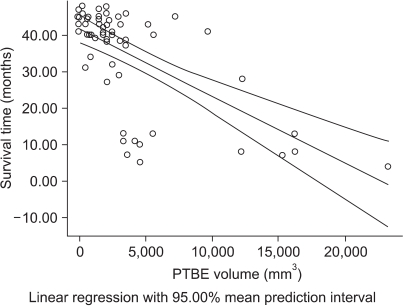
Occludin was detected in 41 (68.3%) of the cases with brain tumors and it was not expressed in the other 19 (31.7%) cases. Although the lowest expression was revealed in high-grade gliomas, its expression was variable according to the pathology of the brain tumors (p>0.05). The difference of PTBE volume between occludin-positive and negative brain tumors was statistically significant (2072.46+/-328.73 mm3 vs. 7452.42+/-1504.19 mm3, respectively, p=0.002). The mean survival time was longer in the occludin-positive tumor group than in the occludin-negative group (38.63+/-1.57 months vs. 26.16+/-3.83 months, respectively; p=0.016).
CONCLUSIONS
This study suggests that the occludin expression is highly correlated to the development of PTBE in brain tumors and it might be a prognostic indicator for patient survival.
MeSH Terms
Figure
Reference
-
1. Yoshioka H, Hama S, Taniguchi E, Sugiyama K, Arita K, Kurisu K. Peritumoral brain edema associated with meningioma: influence of vascular endothelial growth factor expression and vascular blood supply. Cancer. 1999; 85:36–44.2. Kaal EC, Vecht CJ. The management of brain edema in brain tumors. Curr Opin Oncol. 2004; 16:593–600. PMID: 15627023.
Article3. Burger PC, Heinz ER, Shibata T, Kleihues P. Topographic anatomy and CT correlations in the untreated glioblastoma multiforme. J Neurosurg. 1988; 68:698–704. PMID: 2833587.
Article4. Furuse M, Fujita K, Hiiragi T, Fujimoto K, Tsukita S. Claudin-1 and -2: novel integral membrane proteins localizing at tight junctions with no sequence similarity to occludin. J Cell Biol. 1998; 141:1539–1550. PMID: 9647647.
Article5. Furuse M, Hirase T, Itoh M, Nagafuchi A, Yonemura S, Tsukita S. Occludin: a novel integral membrane protein localizing at tight junctions. J Cell Biol. 1993; 123:1777–1788. PMID: 8276896.
Article6. Kleihues P, Cavanee P. WHO Classification of tumors: pathology and genetic of tumors of the nervous system. 2000. Lyon: IARC Press;p. 9–54.7. Thapar K, Rutka JT, Laws ER Jr. Kaye AH, Laws ER, editors. Brain edema, increased intracranial pressure, vascular effects, and other epiphenomena of human brain tumors. Brain tumors: an encyclopedic approach. 1995. Edinburgh: Churchill Livingstone;p. 163–189.8. Sinha S, Bastin ME, Wardlaw JM, Armitage PA, Whittle IR. Effects of dexamethasone on peritumoural oedematous brain: a DT-MRI study. J Neurol Neurosurg Psychiatry. 2004; 75:1632–1635. PMID: 15489404.
Article9. Muresan Z, Paul DL, Goodenough DA. Occludin 1B, a variant of the tight junction protein occludin. Mol Biol Cell. 2000; 11:627–634. PMID: 10679019.
Article10. Wong V. Phosphorylation of occludin correlates with occludin localization and function at the tight junction. Am J Physiol. 1997; 273:1859–1867.11. Papadopoulos MC, Saadoun S, Woodrow CJ, Davies DC, Costa-Martins P, Moss RF, et al. Occludin expression in microvessels of neoplastic and non-neoplastic human brain. Neuropathol Appl Neurobiol. 2001; 27:384–395. PMID: 11679090.
Article12. Bar-Sella P, Front D, Hardoff R, Peyser E, Borovich B, Nir I. Ultrastructural basis for different pertechnetate uptake patterns by various human brain tumours. J Neurol Neurosurg Psychiatry. 1979; 42:924–930. PMID: 229200.
Article13. Gumbiner BM. Breaking through the tight junction barrier. J Cell Biol. 1993; 123:1631–1633. PMID: 8276885.
Article14. Saitou M, Fujimoto K, Doi Y, Itoh M, Fujimoto T, Furuse M, et al. Occludin-deficient embryonic stem cells can differentiate into polarized epithelial cells bearing tight junctions. J Cell Biol. 1998; 141:397–408. PMID: 9548718.
Article15. Li D, Mrsny RJ. Oncogenic Raf-1 disrupts epithelial tight junctions via downregulation of occludin. J Cell Biol. 2000; 148:791–800. PMID: 10684259.
Article16. Liebner S, Fischmann A, Rascher G, Duffner F, Grote EH, Kalbacher H, et al. Claudin-1 and claudin-5 expression and tight junction morphology are altered in blood vessels of human glioblastoma multiforme. Acta Neuropathol (Berl). 2000; 100:323–331. PMID: 10965803.
Article17. Otsuka S, Tamiya T, Ono Y, Michiue H, Kurozumi K, Daido S, et al. The relationship between peritumoral brain edema and the expression of vascular endothelial growth factor and its receptors in intracranial meningiomas. J Neurooncol. 2004; 70:349–357. PMID: 15662977.
Article18. Jiang WG, Martin TA, Matsumoto K, Nakamura T, Mansel RE. Hepatocyte growth factor/scatter factor decreases the expression of occludin and transendothelial resistance (TER) and increases paracellular permeability in human vascular endothelial cells. J Cell Physiol. 1999; 181:319–329. PMID: 10497311.
Article19. Kim TY, Park JT, Moon SK, Han WC. Expression of vascular endothelial growth factor in astrocytic tumors: correlation to peritumoral brain edema and microvasculature. J Korean Neurosurg Soc. 2000; 29:1303–1308.20. Duffy HS, John GR, Lee SC, Brosnan CF, Spray DC. Reciprocal regulation of the junctional proteins claudin-1 and connexin43 by interleukin-1 beta in primary human fetal astrocytes. J Neurosci. 2000; 20(114):1–6. PMID: 10627575.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Differential Expression of the Tight Junction Protein, Occludin, in Brain Tumors
- Expression of Vascular Endothelial Growth Factor in Astrocytic Tumors: Correlation to Peritumoral Brain Edema and Microvasculature
- Solid Cerebellar Hemangioblastoma with Peritumoral Edema: 5-Years Follow up
- A Study about Peritumoral Brain Edema in Meningiomas using Angiographic Pattern and MIB-1
- Expression of Vascular Endothelial Growth Factor and Peritumoral Brain Edema in Intracranial Meningiomas