J Gynecol Oncol.
2015 Jul;26(3):193-200. 10.3802/jgo.2015.26.3.193.
High-dose oral tegafur-uracil maintenance therapy in patients with uterine cervical cancer
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Kumamoto University Faculty of Life Sciences, Kumamoto, Japan. isakakuh@gmail.com
- 2Department of Obstetrics and Gynecology, Japanese Red Cross Kumamoto Hospital, Kumamoto, Japan.
- 3Department of Obstetrics and Gynecology, Kumamoto Rosai Hospital, Yatsushiro, Japan.
- 4Department of Obstetrics and Gynecology, Arao Municipal Hospital, Arao, Japan.
- 5Department of Obstetrics and Gynecology, National Hospital Organization Kumamoto Medical Center, Kumamoto, Japan.
- 6School & Graduate School of Health Sciences, Kumamoto University Faculty of Life Sciences, Kumamoto, Japan.
- KMID: 2160805
- DOI: http://doi.org/10.3802/jgo.2015.26.3.193
Abstract
OBJECTIVE
The aim of this study was to determine the efficacy and toxicity of oral administration of tegafur-uracil (UFT) at a high dose, 600 mg/day, based on the tegafur dose, against uterine cervical cancer.
METHODS
This study consisted of a retrospective analysis. From April 1986 to March 1997, 309 patients with uterine cervical cancer were registered. Oral UFT was administered to 162 patients for maintenance therapy after an initial treatment (the UFT group). The other 147 patients were not treated with UFT (the control group). The survival rate was calculated for both groups and statistically analyzed using the log-rank test. Adverse events were compared between the UFT and control groups.
RESULTS
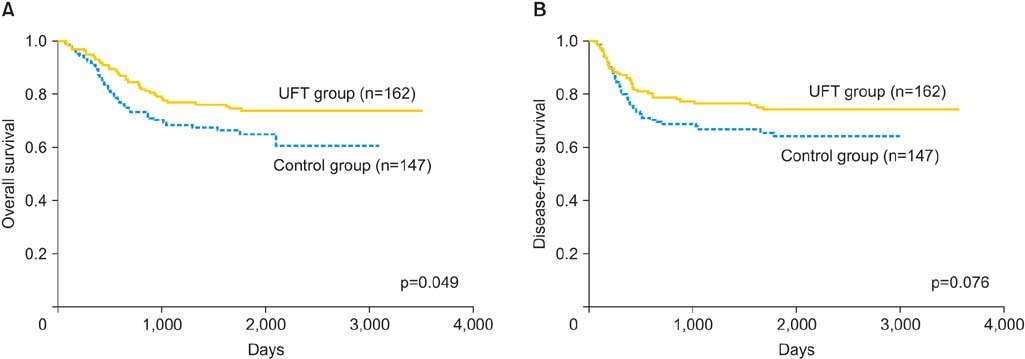
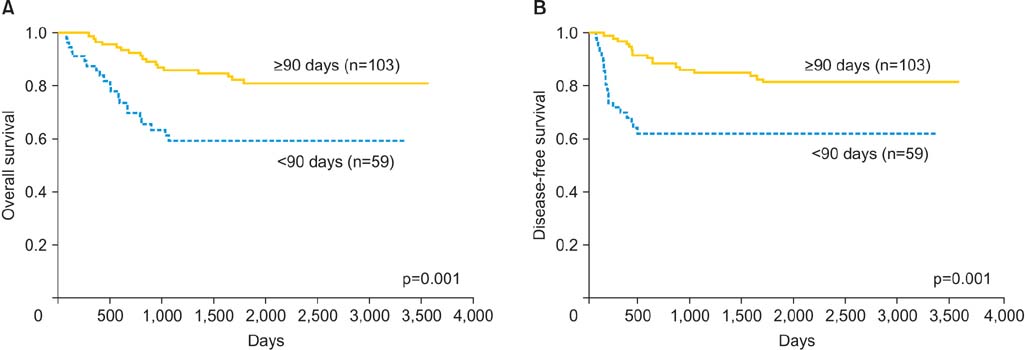
In the UFT group, 103 patients (63.6%) received UFT for > or =90 days. The drug dose was 600 mg/day for 137 patients (84.6%) and 300 to 400 mg/day for the remainder. The overall survival rate was significantly higher in the UFT group than in the control group (p<0.05). The prognosis was particularly favorable in stage III cases, in cases of squamous cell carcinoma, and in cases that were treated by radiotherapy. The most frequent side effects were nausea/vomiting (12.2%), appetite loss (10.1%), and leukopenia/neutropenia (5.8%).
CONCLUSION
High-dose oral UFT maintenance treatment prolonged the disease-free survival and overall survival of patients with uterine cervical cancer, particularly of those with advanced disease.
Keyword
MeSH Terms
-
Administration, Oral
Antineoplastic Combined Chemotherapy Protocols/*therapeutic use
Disease-Free Survival
Female
Humans
Kaplan-Meier Estimate
Middle Aged
Retrospective Studies
Tegafur/administration & dosage/adverse effects
Treatment Outcome
Uracil/administration & dosage/adverse effects
Uterine Cervical Neoplasms/*drug therapy/mortality
Tegafur
Uracil
Figure
Reference
-
1. Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011; 61:69–90.2. Fujii S, Kitano S, Ikenaka K, Shirasaka T. Effect of coadministration of uracil or cytosine on the anti-tumor activity of clinical doses of 1-(2-tetrahydrofuryl)-5-fluorouracil and level of 5-fluorouracil in rodents. Gan. 1979; 70:209–214.3. Tanaka F, Fukuse T, Wada H, Fukushima M. The history, mechanism and clinical use of oral 5-fluorouracil derivative chemotherapeutic agents. Curr Pharm Biotechnol. 2000; 1:137–164.4. Ikenaka K, Shirasaka T, Kitano S, Fujii S. Effect of uracil on metabolism of 5-fluorouracil in vitro. Gan. 1979; 70:353–359.5. Wada H, Hitomi S, Teramatsu T. Adjuvant chemotherapy after complete resection in non-small-cell lung cancer. West Japan Study Group for Lung Cancer Surgery. J Clin Oncol. 1996; 14:1048–1054.6. Kato H, Ichinose Y, Ohta M, Hata E, Tsubota N, Tada H, et al. A randomized trial of adjuvant chemotherapy with uracil-tegafur for adenocarcinoma of the lung. N Engl J Med. 2004; 350:1713–1721.7. Nakagawa K, Tada H, Akashi A, Yasumitsu T, Iuchi K, Taki T, et al. Randomised study of adjuvant chemotherapy for completely resected p-stage I-IIIA non-small cell lung cancer. Br J Cancer. 2006; 95:817–821.8. Kinoshita T, Nakajima T, Ohashi Y. National Surgical Adjuvant Study Group for Gastric Cancer. Adjuvant chemotherapy with uraciltegafur (UFT) for serosa negative advanced gastric cancer: results of a randomized trial by national surgical adjuvant study of gastric cancer. J Clin Oncol. 2005; 23:16 Suppl. 313S.9. Nakajima T, Kinoshita T, Nashimoto A, Sairenji M, Yamaguchi T, Sakamoto J, et al. Randomized controlled trial of adjuvant uraciltegafur versus surgery alone for serosa-negative, locally advanced gastric cancer. Br J Surg. 2007; 94:1468–1476.10. Akasu T, Moriya Y, Ohashi Y, Yoshida S, Shirao K, Kodaira S, et al. Adjuvant chemotherapy with uracil-tegafur for pathological stage III rectal cancer after mesorectal excision with selective lateral pelvic lymphadenectomy: a multicenter randomized controlled trial. Jpn J Clin Oncol. 2006; 36:237–244.11. Noguchi S, Koyama H, Uchino J, Abe R, Miura S, Sugimachi K, et al. Postoperative adjuvant therapy with tamoxifen, tegafur plus uracil, or both in women with node-negative breast cancer: a pooled analysis of six randomized controlled trials. J Clin Oncol. 2005; 23:2172–2184.12. Yamamoto K, Izumi R, Hasegawa K, Nakajima H, Ohashi K, Kudo R, et al. Adjuvant oral 5-fluorouracil for cervical cancer: Japanese Gynecologic Oncology Group report. Int J Oncol. 2004; 24:1175–1179.13. Ho DH, Pazdur R, Covington W, Brown N, Huo YY, Lassere Y, et al. Comparison of 5-fluorouracil pharmacokinetics in patients receiving continuous 5-fluorouracil infusion and oral uracil plus N1-(2'-tetrahydrofuryl)-5-fluorouracil. Clin Cancer Res. 1998; 4:2085–2088.14. Taguchi T, Ikeda Y, Mikami Y, Kawai S, Arai Y, Mochimatsu I, et al. Combined radiotherapy and chemotherapy with carboplatin and UFT for head and neck squamous cell carcinoma. Anticancer Res. 2003; 23(1B):713–717.15. Ichinose Y, Yano T, Asoh H, Yokoyama H, Maruyama R, Ushijima C, et al. UFT plus cisplatin with concurrent radiotherapy for locally advanced non-small-cell lung cancer. Oncology (Williston Park). 1999; 13:7 Suppl 3. 98–101.16. Cellier P, Leduc B, Martin L, Vie B, Chevelle C, Vendrely V, et al. Phase II study of preoperative radiation plus concurrent daily tegafururacil (UFT) with leucovorin for locally advanced rectal cancer. BMC Cancer. 2011; 11:98.17. Lorvidhaya V, Chitapanarux I, Sangruchi S, Lertsanguansinchai P, Kongthanarat Y, Tangkaratt S, et al. Concurrent mitomycin C, 5-fluorouracil, and radiotherapy in the treatment of locally advanced carcinoma of the cervix: a randomized trial. Int J Radiat Oncol Biol Phys. 2003; 55:1226–1232.18. Duenas-Gonzalez A, Zarba JJ, Patel F, Alcedo JC, Beslija S, Casanova L, et al. Phase III, open-label, randomized study comparing concurrent gemcitabine plus cisplatin and radiation followed by adjuvant gemcitabine and cisplatin versus concurrent cisplatin and radiation in patients with stage IIB to IVA carcinoma of the cervix. J Clin Oncol. 2011; 29:1678–1685.19. Tanaka F, Yanagihara K, Otake Y, Miyahara R, Wada H. Angiogenesis and the efficacy of postoperative administration of UFT in pathologic stage I non-small cell lung cancer. Cancer Sci. 2004; 95:371–376.20. Tanaka F, Otake Y, Yanagihara K, Kawano Y, Miyahara R, Li M, et al. Correlation between apoptotic index and angiogenesis in non-small cell lung cancer: comparison between CD105 and CD34 as a marker of angiogenesis. Lung Cancer. 2003; 39:289–296.21. Tanaka F, Otake Y, Yanagihara K, Yamada T, Miyahara R, Kawano Y, et al. Apoptosis and p53 status predict the efficacy of postoperative administration of UFT in non-small cell lung cancer. Br J Cancer. 2001; 84:263–269.22. Nakagawa T, Tanaka F, Takata T, Matsuoka K, Miyahara R, Otake Y, et al. Predictive value of dihydropyrimidine dehydrogenase expression in tumor tissue, regarding the efficacy of postoperatively administered UFT (Tegafur + Uracil) in patients with p-stage I nonsmall-cell lung cancer. J Surg Oncol. 2002; 81:87–92.23. Nakano J, Huang C, Liu D, Masuya D, Nakashima T, Yokomise H, et al. Evaluations of biomarkers associated with 5-FU sensitivity for non-small-cell lung cancer patients postoperatively treated with UFT. Br J Cancer. 2006; 95:607–615.24. Nagai N, Mukai K, Hirata E, Jin HH, Komatsu M, Yunokawa M. UFT and its metabolite gamma-butyrolactone (GBL) inhibit angiogenesis induced by vascular endothelial growth factor in advanced cervical carcinoma. Med Oncol. 2008; 25:214–221.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Fixed Drug Eruption Induced by Tegafur-Uracil(TEGASIL)
- Discoid Lupus Erythematosus-like Eruption Induced by Tegafur - Uracil (UFT)
- Clinical Pharmacokinetics of Tegafur Administered with Epirubicin and Cisplatin in Patients with Advanced Gastric Cancer
- Oral Tegafur-uracil Plus Folinic Acid versus Intravenous 5-fluorouracil Plus Folinic Acid as Adjuvant Chemotherapy of Colon Cancer
- Accuracy of Dose Estimation in High Dose Rate Intracavitary Radiotherapy of Carcinoma of the Uterine Cervix