J Korean Med Sci.
2015 May;30(5):559-568. 10.3346/jkms.2015.30.5.559.
A Novel Angiotensin Type I Receptor Antagonist, Fimasartan, Prevents Doxorubicin-induced Cardiotoxicity in Rats
- Affiliations
-
- 1Division of Cardiology, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. eunseok.jeon@samsung.com
- 2Cardiovascular Imaging Center, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 3Department of Biomedical Science, Jungwon University, Goesan-gun, Korea.
- KMID: 2155468
- DOI: http://doi.org/10.3346/jkms.2015.30.5.559
Abstract
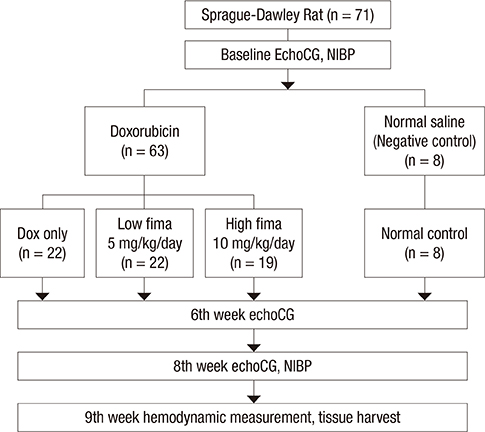
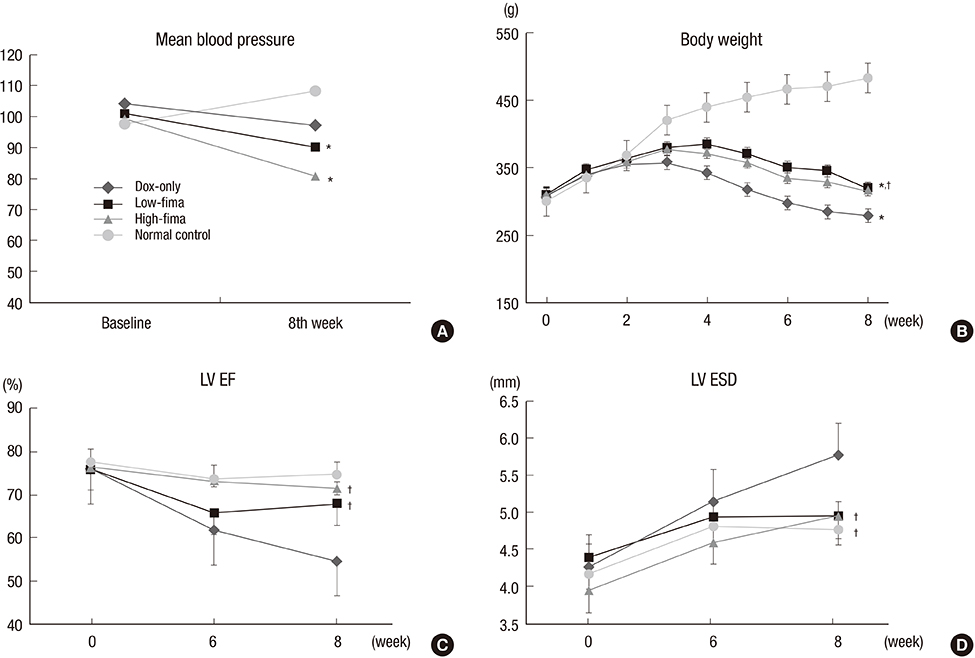
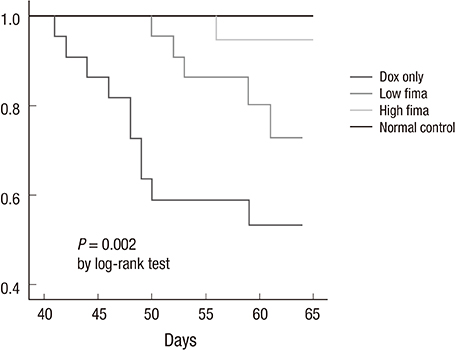
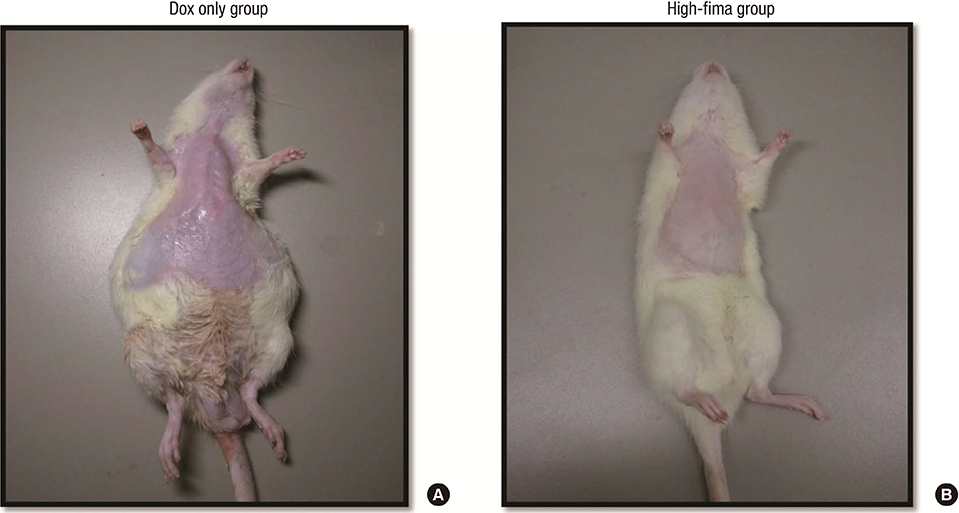
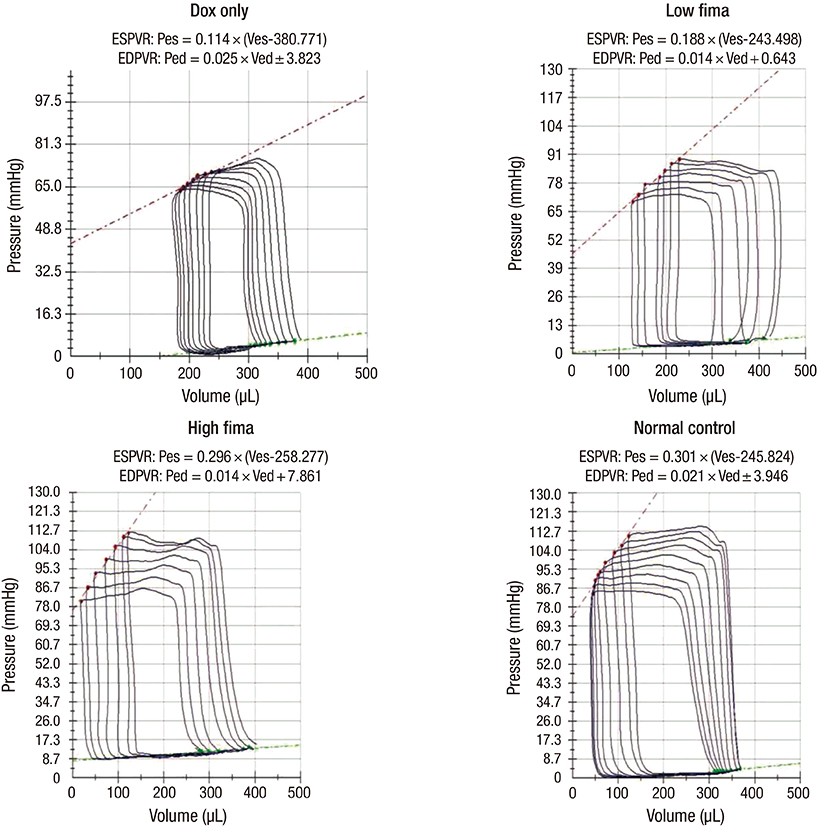
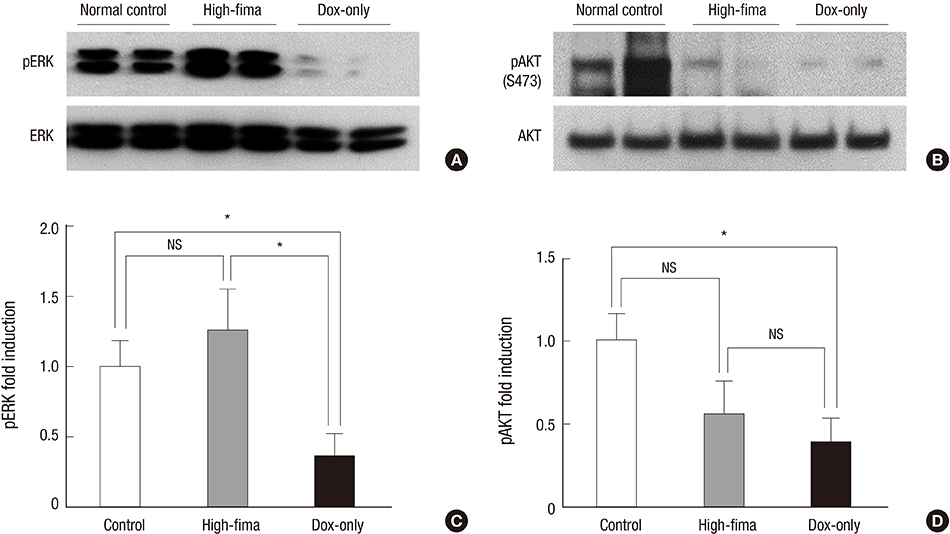
- Angiotensin receptor blockers (ARBs) have organ-protective effects in heart failure and may be also effective in doxorubicin-induced cardiomyopathy (DOX-CMP); however, the efficacy of ARBs on the prevention of DOX-CMP have not been investigated. We performed a preclinical experiment to evaluate the preventive effect of a novel ARB, fimasartan, in DOX-CMP. All animals underwent echocardiography and were randomly assigned into three groups: treated daily with vehicle (DOX-only group, n=22), 5 mg/kg of fimasartan (Low-fima group, n=22), and 10 mg/kg of fimasartan (High-fima group, n=19). DOX was injected once a week for six weeks. Echocardiography and hemodynamic assessment was performed at the 8th week using a miniaturized conductance catheter. Survival rate of the High-fima group was greater (100%) than that of the Low-fima (75%) and DOX-only groups (50%). Echocardiography showed preserved left ventricular (LV) ejection fraction in the High-fima group, but not in the DOX-only group (P=0.002). LV dimensions increased in the DOX-only group; however, remodeling was attenuated in the Low-fima and High-fima groups. Hemodynamic assessment showed higher dP/dt in the High-fima group compared with the DOX-only group. A novel ARB, fimasartan, may prevent DOX-CMP and improve survival rate in a dose-dependent manner in a rat model of DOX-CMP and could be a treatment option for the prevention of DOX-CMP.
MeSH Terms
-
Angiotensin Receptor Antagonists/*therapeutic use
Animals
Biphenyl Compounds/*therapeutic use
Cardiomyopathies/chemically induced/mortality/*prevention & control
Doxorubicin/*toxicity
Echocardiography
Hemodynamics
Pyrimidines/*therapeutic use
Rats
Rats, Sprague-Dawley
Receptor, Angiotensin, Type 1/chemistry/*metabolism
Survival Rate
Tetrazoles/*therapeutic use
Ventricular Function, Left/physiology
Angiotensin Receptor Antagonists
Biphenyl Compounds
Doxorubicin
Pyrimidines
Receptor, Angiotensin, Type 1
Tetrazoles
Figure
Cited by 2 articles
-
Therapeutic Effect of Fimasartan in a Rat Model of Myocardial Infarction Evaluated by Cardiac Positron Emission Tomography with [18F]FPTP
Hyukjin Park, Hyeon Sik Kim, Young Joon Hong, Jung-Joon Min, Han Byul Kim, Min Chul Kim, Doo Sun Sim, Ju Han Kim, Dong-Yeon Kim, Jae Sung Lee, Youngkeun Ahn, Myung Ho Jeong
Chonnam Med J. 2019;55(2):109-115. doi: 10.4068/cmj.2019.55.2.109.Fimasartan attenuates renal ischemia-reperfusion injury by modulating inflammation-related apoptosis
Jang-Hee Cho, Soon-Youn Choi, Hye-Myung Ryu, Eun-Joo Oh, Ju-Min Yook, Ji-Sun Ahn, Hee-Yeon Jung, Ji-Young Choi, Sun-Hee Park, Chan-Duck Kim, Yong-Lim Kim
Korean J Physiol Pharmacol. 2018;22(6):661-670. doi: 10.4196/kjpp.2018.22.6.661.
Reference
-
1. Singal PK, Iliskovic N. Doxorubicin-induced cardiomyopathy. N Engl J Med. 1998; 339:900–905.2. Lefrak EA, Pitha J, Rosenheim S, Gottlieb JA. A clinicopathologic analysis of adriamycin cardiotoxicity. Cancer. 1973; 32:302–314.3. Von Hoff DD, Layard MW, Basa P, Davis HL Jr, Von Hoff AL, Rozencweig M, Muggia FM. Risk factors for doxorubicin-induced congestive heart failure. Ann Intern Med. 1979; 91:710–717.4. Minow RA, Benjamin RS, Lee ET, Gottlieb JA. Adriamycin cardiomyopathy--risk factors. Cancer. 1977; 39:1397–1402.5. Mitani I, Jain D, Joska TM, Burtness B, Zaret BL. Doxorubicin cardiotoxicity: prevention of congestive heart failure with serial cardiac function monitoring with equilibrium radionuclide angiocardiography in the current era. J Nucl Cardiol. 2003; 10:132–139.6. Georgakopoulos D, Mitzner WA, Chen CH, Byrne BJ, Millar HD, Hare JM, Kass DA. In vivo murine left ventricular pressure-volume relations by miniaturized conductance micromanometry. Am J Physiol. 1998; 274:H1416–H1422.7. Singal PK, Li T, Kumar D, Danelisen I, Iliskovic N. Adriamycin-induced heart failure: mechanism and modulation. Mol Cell Biochem. 2000; 207:77–86.8. Lebrecht D, Kokkori A, Ketelsen UP, Setzer B, Walker UA. Tissue-specific mtDNA lesions and radical-associated mitochondrial dysfunction in human hearts exposed to doxorubicin. J Pathol. 2005; 207:436–444.9. Lipshultz SE, Rifai N, Dalton VM, Levy DE, Silverman LB, Lipsitz SR, Colan SD, Asselin BL, Barr RD, Clavell LA, et al. The effect of dexrazoxane on myocardial injury in doxorubicin-treated children with acute lymphoblastic leukemia. N Engl J Med. 2004; 351:145–153.10. Speyer JL, Green MD, Kramer E, Rey M, Sanger J, Ward C, Dubin N, Ferrans V, Stecy P, Zeleniuch-Jacquotte A, et al. Protective effect of the bispiperazinedione ICRF-187 against doxorubicin-induced cardiac toxicity in women with advanced breast cancer. N Engl J Med. 1988; 319:745–752.11. Cohn JN, Tognoni G. Valsartan Heart Failure Trial Investigators. A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure. N Engl J Med. 2001; 345:1667–1675.12. McMurray JJ, Ostergren J, Swedberg K, Granger CB, Held P, Michelson EL, Olofsson B, Yusuf S, Pfeffer MA. CHARM Investigators and Committees. Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function taking angiotensin-converting-enzyme inhibitors: the CHARM-Added trial. Lancet. 2003; 362:767–771.13. Heidenreich PA, Lee TT, Massie BM. Effect of beta-blockade on mortality in patients with heart failure: a meta-analysis of randomized clinical trials. J Am Coll Cardiol. 1997; 30:27–34.14. Kalay N, Basar E, Ozdogru I, Er O, Cetinkaya Y, Dogan A, Inanc T, Oguzhan A, Eryol NK, Topsakal R, et al. Protective effects of carvedilol against anthracycline-induced cardiomyopathy. J Am Coll Cardiol. 2006; 48:2258–2262.15. Cardinale D, Colombo A, Sandri MT, Lamantia G, Colombo N, Civelli M, Martinelli G, Veglia F, Fiorentini C, Cipolla CM. Prevention of high-dose chemotherapy-induced cardiotoxicity in high-risk patients by angiotensin-converting enzyme inhibition. Circulation. 2006; 114:2474–2481.16. Sjølie AK, Klein R, Porta M, Orchard T, Fuller J, Parving HH, Bilous R, Chaturvedi N. DIRECT Programme Study Group. Effect of candesartan on progression and regression of retinopathy in type 2 diabetes (DIRECT-Protect 2): a randomised placebo-controlled trial. Lancet. 2008; 372:1385–1393.17. Konstam MA, Neaton JD, Dickstein K, Drexler H, Komajda M, Martinez FA, Riegger GA, Malbecq W, Smith RD, Guptha S, et al. Effects of high-dose versus low-dose losartan on clinical outcomes in patients with heart failure (HEAAL study): a randomised, double-blind trial. Lancet. 2009; 374:1840–1848.18. Toko H, Oka T, Zou Y, Sakamoto M, Mizukami M, Sano M, Yamamoto R, Sugaya T, Komuro I. Angiotensin II type 1a receptor mediates doxorubicin-induced cardiomyopathy. Hypertens Res. 2002; 25:597–603.19. Soga M, Kamal FA, Watanabe K, Ma M, Palaniyandi S, Prakash P, Veeraveedu P, Mito S, Kunisaki M, Tachikawa H, et al. Effects of angiotensin II receptor blocker (candesartan) in daunorubicin-induced cardiomyopathic rats. Int J Cardiol. 2006; 110:378–385.20. Ibrahim MA, Ashour OM, Ibrahim YF, El-Bitar HI, Gomaa W, Abdel-Rahim SR. Angiotensin-converting enzyme inhibition and angiotensin AT(1)-receptor antagonism equally improve doxorubicin-induced cardiotoxicity and nephrotoxicity. Pharmacol Res. 2009; 60:373–381.21. Arozal W, Watanabe K, Veeraveedu PT, Thandavarayan RA, Harima M, Sukumaran V, Suzuki K, Kodama M, Aizawa Y. Effect of telmisartan in limiting the cardiotoxic effect of daunorubicin in rats. J Pharm Pharmacol. 2010; 62:1776–1783.22. Kim JH, Lee JH, Paik SH, Kim JH, Chi YH. Fimasartan, a novel angiotensin II receptor antagonist. Arch Pharm Res. 2012; 35:1123–1126.23. Sim DS, Jeong MH, Song HC, Kim J, Chong A, Bom HS, Jeong IS, Oh SG, Kim JM, Park DS, et al. Cardioprotective effect of fimasartan, a new angiotensin receptor blocker, in a porcine model of acute myocardial infarction. J Korean Med Sci. 2015; 30:34–43.24. Omura T, Yoshiyama M, Ishikura F, Kobayashi H, Takeuchi K, Beppu S, Yoshikawa J. Myocardial ischemia activates the JAK-STAT pathway through angiotensin II signaling in in vivo myocardium of rats. J Mol Cell Cardiol. 2001; 33:307–316.25. Inamura K, Matsuzaki Y, Uematsu N, Honda A, Tanaka N, Uchida K. Rapid inhibition of MAPK signaling and anti-proliferation effect via JAK/STAT signaling by interferon-alpha in hepatocellular carcinoma cell lines. Biochim Biophys Acta. 2005; 1745:401–410.26. Xuan YT, Guo Y, Zhu Y, Wang OL, Rokosh G, Messing RO, Bolli R. Role of the protein kinase C-epsilon-Raf-1-MEK-1/2-p44/42 MAPK signaling cascade in the activation of signal transducers and activators of transcription 1 and 3 and induction of cyclooxygenase-2 after ischemic preconditioning. Circulation. 2005; 112:1971–1978.27. Li Y, Takemura G, Okada H, Miyata S, Maruyama R, Li L, Higuchi M, Minatoguchi S, Fujiwara T, Fujiwara H. Reduction of inflammatory cytokine expression and oxidative damage by erythropoietin in chronic heart failure. Cardiovasc Res. 2006; 71:684–694.28. Lu Y, Zhou J, Xu C, Lin H, Xiao J, Wang Z, Yang B. JAK/STAT and PI3K/AKT pathways form a mutual transactivation loop and afford resistance to oxidative stress-induced apoptosis in cardiomyocytes. Cell Physiol Biochem. 2008; 21:305–314.29. Lipshultz SE, Lipsitz SR, Sallan SE, Dalton VM, Mone SM, Gelber RD, Colan SD. Chronic progressive cardiac dysfunction years after doxorubicin therapy for childhood acute lymphoblastic leukemia. J Clin Oncol. 2005; 23:2629–2636.30. Uemura H, Ishiguro H, Nakaigawa N, Nagashima Y, Miyoshi Y, Fujinami K, Sakaguchi A, Kubota Y. Angiotensin II receptor blocker shows antiproliferative activity in prostate cancer cells: a possibility of tyrosine kinase inhibitor of growth factor. Mol Cancer Ther. 2003; 2:1139–1147.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Fimasartan, an angiotensin II receptor antagonist, ameliorates an in vivo zebrafish model of heart failure
- Angiotensin II Type 1 Receptor Blocker, Fimasartan, Reduces Vascular Smooth Muscle Cell Senescence by Inhibiting the CYR61 Signaling Pathway
- Pathophysiology and preventive strategies of anthracycline-induced cardiotoxicity
- Downregulation of Angiotensin II-Induced 12-Lipoxygenase Expression and Cell Proliferation in Vascular Smooth Muscle Cells from Spontaneously Hypertensive Rats by CCL5
- Comparative effects of angiotensin II and angiotensin-(4-8) on blood pressure and ANP secretion in rats