Immune Netw.
2011 Jun;11(3):163-168. 10.4110/in.2011.11.3.163.
Formulation and Characterization of Antigen-loaded PLGA Nanoparticles for Efficient Cross-priming of the Antigen
- Affiliations
-
- 1College of Pharmacy, Chungbuk National University, Cheongju 361-763, Korea. cklee@chungbuk.ac.kr
- 2College of Pharmacy, ShamYook University, Seoul 139-742, Korea.
- KMID: 2150703
- DOI: http://doi.org/10.4110/in.2011.11.3.163
Abstract
- BACKGROUND
Nanoparticles (NPs) prepared from biodegradable polymers, such as poly (D,L-lactic acid-co-glycolic acid) (PLGA), have been studied as vehicles for the delivery of antigens to phagocytes. This paper describes the preparation of antigen-loaded PLGA-NPs for efficient cross-priming.
METHODS
NPs containing a similar amount of ovalbumin (OVA) but different sizes were produced using a micromixer-based W/O/W solvent evaporation procedure, and the efficiency of the NPs to induce the cross-presentation of OVA peptides were examined in dendritic cells (DCs). Cellular uptake and biodistribution studies were performed using fluorescein isothiocyanate (FITC)-loaded NPs in mice.
RESULTS
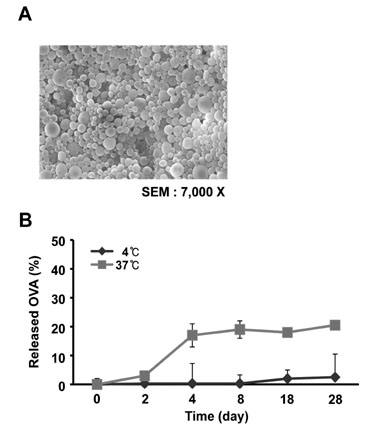
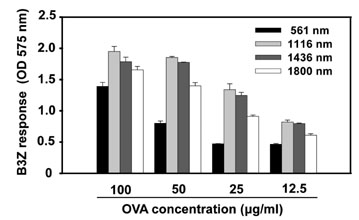
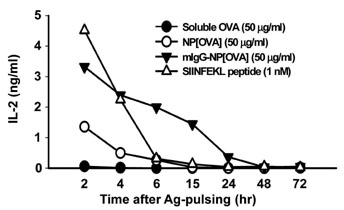
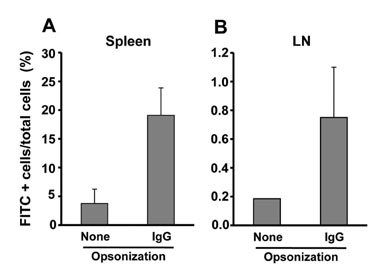
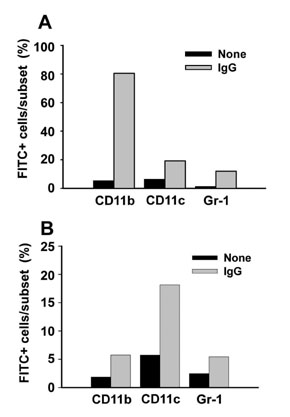
The NPs in the range of 1.1~1.4microm in size were the most and almost equally efficient in inducing the cross-presentation of OVA peptides via H-2Kb molecules. Cellular uptake and biodistribution studies showed that opsonization of the NPs with mouse IgG greatly increased the percentage of FITC-positive cells in the spleen and lymph nodes. The major cell type of FITC-positive cells in the spleen was macrophages, whereas that of lymph nodes was DCs.
CONCLUSION
These results show that IgG-opsonized PLGA-NPs with a mean size of 1.1microm would be the choice of biodegradable carriers for the targeted-delivery of protein antigens for cross-priming in vivo.
Keyword
MeSH Terms
Figure
Cited by 2 articles
-
Induction of Potent Antigen-specific Cytotoxic T Cell Response by PLGA-nanoparticles Containing Antigen and TLR Agonist
Young-Ran Lee, Young-Hee Lee, Ki-Hyang Kim, Sun-A Im, Chong-Kil Lee
Immune Netw. 2013;13(1):30-33. doi: 10.4110/in.2013.13.1.30.Nanoliposomes of L-lysine-conjugated poly(aspartic acid) Increase the Generation and Function of Bone Marrow-derived Dendritic Cells
Sun-A Im, Ki-Hyang Kim, Hong-Geun Ji, Hyoung-Gyoung Yu, Sun-Ki Park, Chong-Kil Lee
Immune Netw. 2011;11(5):281-287. doi: 10.4110/in.2011.11.5.281.
Reference
-
1. Bevan MJ. Cross-priming for a secondary cytotoxic response to minor H antigens with H-2 congenic cells which do not cross-react in the cytotoxic assay. J Exp Med. 1976. 143:1283–1288.
Article2. Champion JA, Walker A, Mitragotri S. Role of particle size in phagocytosis of polymeric microspheres. Pharm Res. 2008. 25:1815–1821.
Article3. Elamanchili P, Diwan M, Cao M, Samuel J. Characterization of poly(D,L-lactic-co-glycolic acid) based nanoparticulate system for enhanced delivery of antigens to dendritic cells. Vaccine. 2004. 22:2406–2412.
Article4. Gerelchuluun T, Lee YH, Lee YR, Im SA, Song S, Park JS, Han K, Kim K, Lee CK. Dendritic cells process antigens encapsulated in a biodegradable polymer, poly(D,L-lactide-co-glycolide), via an alternate class I MHC processing pathway. Arch Pharm Res. 2007. 30:1440–1446.
Article5. Harding CV, Song R. Phagocytic processing of exogenous particulate antigens by macrophages for presentation by class I MHC molecules. J Immunol. 1994. 153:4925–4933.6. Heath WR, Carbone FR. Cross-presentation, dendritic cells, tolerance and immunity. Annu Rev Immunol. 2001. 19:47–64.
Article7. Huang AY, Golumbek P, Ahmadzadeh M, Jaffee E, Pardoll D, Levitsky H. Role of bone marrow-derived cells in presenting MHC class I-restricted tumor antigens. Science. 1994. 264:961–965.
Article8. Jiang W, Gupta RK, Deshpande MC, Schwendeman SP. Biodegradable poly(lactic-co-glycolic acid) microparticles for injectable delivery of vaccine antigens. Adv Drug Deliv Rev. 2005. 57:391–410.
Article9. Johansen P, Men Y, Merkle HP, Gander B. Revisiting PLA/PLGA microspheres: an analysis of their potential in parenteral vaccination. Eur J Pharm Biopharm. 2000. 50:129–146.
Article10. Karttunen J, Sanderson S, Shastri N. Detection of rare antigen-presenting cells by the lacZ T-cell activation assay suggests an expression cloning strategy for T-cell antigens. Proc Natl Acad Sci U S A. 1992. 89:6020–6024.
Article11. Kocbek P, Obermajer N, Cegnar M, Kos J, Kristl J. Targeting cancer cells using PLGA nanoparticles surface modified with monoclonal antibody. J Control Release. 2007. 120:18–26.
Article12. Lee JK, Lee MK, Yun YP, Kim Y, Kim JS, Kim YS, Kim K, Han SS, Lee CK. Acemannan purified from Aloe vera induces phenotypic and functional maturation of immature dendritic cells. Int Immunopharmacol. 2001. 1:1275–1284.
Article13. Lee YH, Lee YR, Kim KH, Im SA, Song S, Lee MK, Kim Y, Hong JT, Kim K, Lee CK. Baccatin III, a synthetic precursor of taxol, enhances MHC-restricted antigen presentation in dendritic cells. Int Immunopharmacol. 2011. [Epub ahead of print].
Article14. Lee YR, Yang IH, Lee YH, Im SA, Song S, Li H, Han K, Kim K, Eo SK, Lee CK. Cyclosporin A and tacrolimus, but not rapamycin, inhibit MHC-restricted antigen presentation pathways in dendritic cells. Blood. 2005. 105:3951–3955.
Article15. Mundargi RC, Babu VR, Rangaswamy V, Patel P, Aminabhavi TM. Nano/micro technologies for delivering macromolecular therapeutics using poly(D,L-lactide-co-glycolide) and its derivatives. J Control Release. 2008. 125:193–209.
Article16. Newman KD, Sosnowski DL, Kwon GS, Samuel J. Delivery of MUC1 mucin peptide by Poly(d,l-lactic-co-glycolic acid) microspheres induces type 1 T helper immune responses. J Pharm Sci. 1998. 87:1421–1427.
Article17. Panyam J, Labhasetwar V. Biodegradable nanoparticles for drug and gene delivery to cells and tissue. Adv Drug Deliv Rev. 2003. 55:329–347.
Article18. Sahoo SK, Panyam J, Prabha S, Labhasetwar V. Residual polyvinyl alcohol associated with poly (D,L-lactide-co-glycolide) nanoparticles affects their physical properties and cellular uptake. J Control Release. 2002. 82:105–114.
Article19. Shen H, Ackerman AL, Cody V, Giodini A, Hinson ER, Cresswell P, Edelson RL, Saltzman WM, Hanlon DJ. Enhanced and prolonged cross-presentation following endosomal escape of exogenous antigens encapsulated in biodegradable nanoparticles. Immunology. 2006. 117:78–88.
Article20. Sigal LJ, Crotty S, Andino R, Rock KL. Cytotoxic T-cell immunity to virus-infected non-haematopoietic cells requires presentation of exogenous antigen. Nature. 1999. 398:77–80.
Article21. Venkataprasad N, Coombes AG, Singh M, Rohde M, Wilkinson K, Hudecz F, Davis SS, Vordermeier HM. Induction of cellular immunity to a mycobacterial antigen adsorbed on lamellar particles of lactide polymers. Vaccine. 1999. 17:1814–1819.
Article22. Waeckerle-Men Y, Groettrup M. PLGA microspheres for improved antigen delivery to dendritic cells as cellular vaccines. Adv Drug Deliv Rev. 2005. 57:475–482.
Article23. Yewdell JW, Haeryfar SM. Understanding presentation of viral antigens to CD8+ T cells in vivo: the key to rational vaccine design. Annu Rev Immunol. 2005. 23:651–682.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Induction of Potent Antigen-specific Cytotoxic T Cell Response by PLGA-nanoparticles Containing Antigen and TLR Agonist
- Docetaxel-loaded PLGA nanoparticles to increase pharmacological sensitivity in MDA-MB-231 and MCF-7 breast cancer cells
- Encapsulation of Low Metronidazole Dose in Poly (D,L-lactide-co-glycolide) (PLGA) Nanoparticles Improves Giardia intestinalis Treatment
- Effect of Poly(Lactide-Co-Glycolide) Nanoparticles on Local Retention of Fluorescent Material: An Experimental Study in Mice
- Immunogenicity of glycine nanoparticles containing a chimeric antigen as Brucella vaccine candidate