J Gynecol Oncol.
2014 Jul;25(3):214-220. 10.3802/jgo.2014.25.3.214.
Conservative therapy with metformin plus megestrol acetate for endometrial atypical hyperplasia
- Affiliations
-
- 1Department of Gynecology, Obstetrics and Gynecology Hospital of Fudan University, Shanghai, China. cxjlh@hotmail.com
- 2Shanghai Key Laboratory of Female Reproductive Endocrine Related Diseases, Shanghai, China.
- 3Department of Pathology, Obstetrics and Gynecology Hospital of Fudan University, Shanghai, China.
- 4Department of Gynecology, Shanghai First People's Hospital, Shanghai Jiao Tong University, Shanghai, China.
- KMID: 2130102
- DOI: http://doi.org/10.3802/jgo.2014.25.3.214
Abstract
OBJECTIVE
To compare the efficacy of metformin plus megestrol acetate (MA) with that of MA alone for treating endometrial atypical hyperplasia (EAH).
METHODS
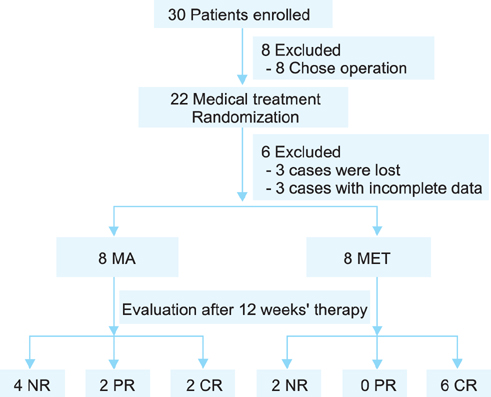
This pilot study included 16 EAH patients who met at least one metabolic syndrome (MS) criterion and received either adjunctive metformin plus MA (MET group) or MA monotherapy (MA group). Each patient in the MA group received 160 mg of MA daily, whereas patients in the MET group received the same dose of MA plus 0.5 g of metformin thrice daily. Treatment response was assessed by histological examination of dilation and curettage specimens obtained after 12 weeks of therapy.
RESULTS
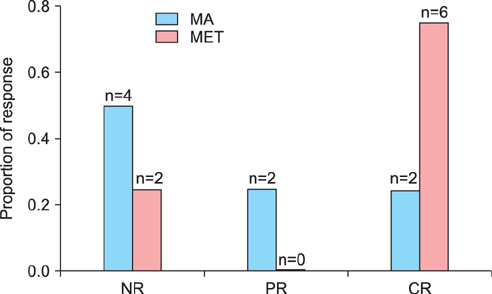
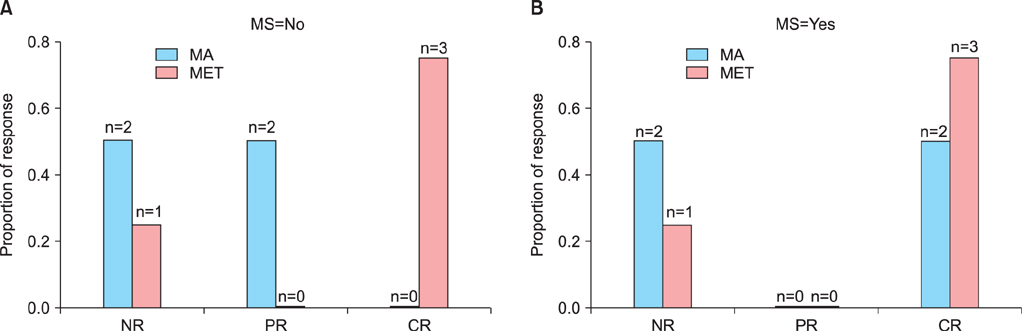
Each group had eight patients, and half of the patients in each group were diagnosed with MS. The complete response (CR) rate was 75% (6/8) in the MET group and 25% (2/8) in the MA group (p=0.105). Complications of MS did not affect the response rates in either group. In the MET group, 75% (3/4) of the patients had CR in the presence or absence of MS. In the MA group, 50% (2/4) of the patients with MS had CR, whereas no patient without MS had CR. No irreversible toxicities were observed.
CONCLUSION
Metformin plus MA may be a potential alternative therapy for treating EAH, and the MS status of patients may have no effect on the efficacy of metformin plus MA therapy.
MeSH Terms
-
Adult
Antineoplastic Agents, Hormonal/*therapeutic use
Drug Therapy, Combination
Endometrial Hyperplasia/complications/*drug therapy/metabolism
Female
Humans
Hypoglycemic Agents/*therapeutic use
Megestrol Acetate/*therapeutic use
Metabolic Syndrome X/complications/metabolism
Metformin/*therapeutic use
Pilot Projects
Receptors, Estrogen/metabolism
Receptors, Progesterone/metabolism
Single-Blind Method
Treatment Outcome
Antineoplastic Agents, Hormonal
Hypoglycemic Agents
Megestrol Acetate
Metformin
Receptors, Estrogen
Receptors, Progesterone
Figure
Cited by 2 articles
-
Insulin resistance and overweight prolonged fertility-sparing treatment duration in endometrial atypical hyperplasia patients
Bingyi Yang, Liying Xie, Hongwei Zhang, Qin Zhu, Yan Du, Xuezhen Luo, Xiaojun Chen
J Gynecol Oncol. 2018;29(3):. doi: 10.3802/jgo.2018.29.e35.Fertility sparing treatment for early stage endometrial cancer: current situation and new strategy
Kimio Ushijima
J Gynecol Oncol. 2019;30(6):. doi: 10.3802/jgo.2019.30.e117.
Reference
-
1. Baker J, Obermair A, Gebski V, Janda M. Efficacy of oral or intrauterine device-delivered progestin in patients with complex endometrial hyperplasia with atypia or early endometrial adenocarcinoma: a meta-analysis and systematic review of the literature. Gynecol Oncol. 2012; 125:263–270.2. Nucera G, Mandato VD, Gelli MC, Palomba S, La Sala GB. Gonadotropin releasing hormone agonist and levonorgestrel-intrauterine device followed by in vitro fertilization program as management strategy for an infertile endometrial cancer patient: a case report. Gynecol Endocrinol. 2013; 29:219–221.3. Brown AJ, Westin SN, Broaddus RR, Schmeler K. Progestin intrauterine device in an adolescent with grade 2 endometrial cancer. Obstet Gynecol. 2012; 119(2 Pt 2):423–426.4. Jasonni VM, Franceschetti F, Ciotti P, Bulletti C, Vignudelli A, Marabini A, et al. Treatment of endometrial hyperplasia with cyproterone acetate histological and hormonal aspects. Acta Obstet Gynecol Scand. 1986; 65:685–687.5. Ushijima K, Yahata H, Yoshikawa H, Konishi I, Yasugi T, Saito T, et al. Multicenter phase II study of fertility-sparing treatment with medroxyprogesterone acetate for endometrial carcinoma and atypical hyperplasia in young women. J Clin Oncol. 2007; 25:2798–2803.6. Gallos ID, Yap J, Rajkhowa M, Luesley DM, Coomarasamy A, Gupta JK. Regression, relapse, and live birth rates with fertility-sparing therapy for endometrial cancer and atypical complex endometrial hyperplasia: a systematic review and metaanalysis. Am J Obstet Gynecol. 2012; 207:266.7. Penner KR, Dorigo O, Aoyama C, Ostrzega N, Balzer BL, Rao J, et al. Predictors of resolution of complex atypical hyperplasia or grade 1 endometrial adenocarcinoma in premenopausal women treated with progestin therapy. Gynecol Oncol. 2012; 124:542–548.8. Ricciardi E, Maniglio P, Frega A, Marci R, Caserta D, Moscarini M. Fertility-sparing treatment of endometrial cancer precursors among young women: a reproductive point of view. Eur Rev Med Pharmacol Sci. 2012; 16:1934–1937.9. Shen ZQ, Zhu HT, Lin JF. Reverse of progestin-resistant atypical endometrial hyperplasia by metformin and oral contraceptives. Obstet Gynecol. 2008; 112(2 Pt 2):465–467.10. Campagnoli C, Abba C, Ambroggio S, Brucato T, Pasanisi P. Life-style and metformin for the prevention of endometrial pathology in postmenopausal women. Gynecol Endocrinol. 2013; 29:119–124.11. Session DR, Kalli KR, Tummon IS, Damario MA, Dumesic DA. Treatment of atypical endometrial hyperplasia with an insulin-sensitizing agent. Gynecol Endocrinol. 2003; 17:405–407.12. Zhang P, Li H, Tan X, Chen L, Wang S. Association of metformin use with cancer incidence and mortality: a meta-analysis. Cancer Epidemiol. 2013; 37:207–218.13. Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009; 120:1640–1645.14. Chen CH, Huang MC, Kao CF, Lin SK, Kuo PH, Chiu CC, et al. Effects of adjunctive metformin on metabolic traits in nondiabetic clozapine-treated patients with schizophrenia and the effect of metformin discontinuation on body weight: a 24-week, randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2013; 74:e424–e430.15. Kurman RJ, Ellenson LH, Ronnett BM. Blaustein's pathology of female genital tract. 6th ed. New York: Springer;2011.16. Vang R, Gown AM, Barry TS, Wheeler DT, Ronnett BM. Immunohistochemistry for estrogen and progesterone receptors in the distinction of primary and metastatic mucinous tumors in the ovary: an analysis of 124 cases. Mod Pathol. 2006; 19:97–105.17. Mentrikoski MJ, Shah AA, Hanley KZ, Atkins KA. Assessing endometrial hyperplasia and carcinoma treated with progestin therapy. Am J Clin Pathol. 2012; 138:524–534.18. Gunderson CC, Fader AN, Carson KA, Bristow RE. Oncologic and reproductive outcomes with progestin therapy in women with endometrial hyperplasia and grade 1 adenocarcinoma: a systematic review. Gynecol Oncol. 2012; 125:477–482.19. Gallos ID, Shehmar M, Thangaratinam S, Papapostolou TK, Coomarasamy A, Gupta JK. Oral progestogens vs levonorgestrel-releasing intrauterine system for endometrial hyperplasia: a systematic review and metaanalysis. Am J Obstet Gynecol. 2010; 203:547.20. Ganie MA, Khurana ML, Nisar S, Shah PA, Shah ZA, Kulshrestha B, et al. Improved efficacy of low-dose spironolactone and metformin combination than either drug alone in the management of women with polycystic ovary syndrome (PCOS): a six-month, open-label randomized study. J Clin Endocrinol Metab. 2013; 98:3599–3607.21. Lexis CP, van der Horst IC, Lipsic E, van der Harst P, van der Horst-Schrivers AN, Wolffenbuttel BH, et al. Metformin in non-diabetic patients presenting with ST elevation myocardial infarction: rationale and design of the glycometabolic intervention as adjunct to primary percutaneous intervention in ST elevation myocardial infarction (GIPS)-III trial. Cardiovasc Drugs Ther. 2012; 26:417–426.22. Xie Y, Wang YL, Yu L, Hu Q, Ji L, Zhang Y, et al. Metformin promotes progesterone receptor expression via inhibition of mammalian target of rapamycin (mTOR) in endometrial cancer cells. J Steroid Biochem Mol Biol. 2011; 126:113–120.23. Zhang Z, Dong L, Sui L, Yang Y, Liu X, Yu Y, et al. Metformin reverses progestin resistance in endometrial cancer cells by downregulating GloI expression. Int J Gynecol Cancer. 2011; 21:213–221.24. Hanna RK, Zhou C, Malloy KM, Sun L, Zhong Y, Gehrig PA, et al. Metformin potentiates the effects of paclitaxel in endometrial cancer cells through inhibition of cell proliferation and modulation of the mTOR pathway. Gynecol Oncol. 2012; 125:458–469.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Conservative treatment for atypical endometrial hyperplasia: what is the most effective therapeutic method?
- A successful twin pregnancy after conservative therapy of endometrial cancer
- Treatment Efficacy of High-Dose Megestrol Acetate (Megace) in Young Women with Early Stage of Endometrial Carcinoma
- A case of treatment using high-dose Megestrol Acetate (Megace(R)) combined with PDT (Photodynamic Therapy) on early stage of endometrial carcinoma, in nulliparous woman
- Treatment of endometrial hyperplasia with Medroxyprogesterone acetate(MPA)