Cancer Res Treat.
2009 Sep;41(3):145-154.
Apoptosis and Expression of AQP5 and TGF-beta in the Irradiated Rat Submandibular Gland
- Affiliations
-
- 1Department of Radiation Oncology, Chung-Ang University Hospital, Seoul, Korea.
- 2Department of Radiation Oncology, Seoul National University College of Medicine, Seoul, Korea. wuhg@snu.ac.kr
- 3Department of Pathology, Seoul National University College of Medicine, Seoul, Korea.
- 4Cancer Research Institute, Seoul National University College of Medicine, Seoul, Korea.
- 5Institute of Radiation Medicine, Medical Research Center, Seoul National University, Seoul, Korea.
Abstract
- PURPOSE
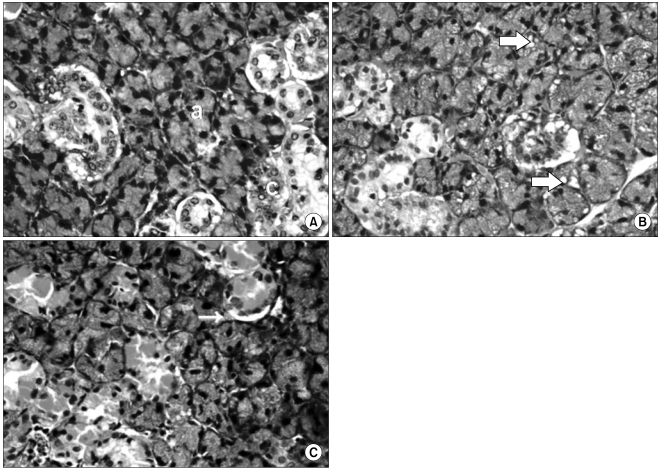
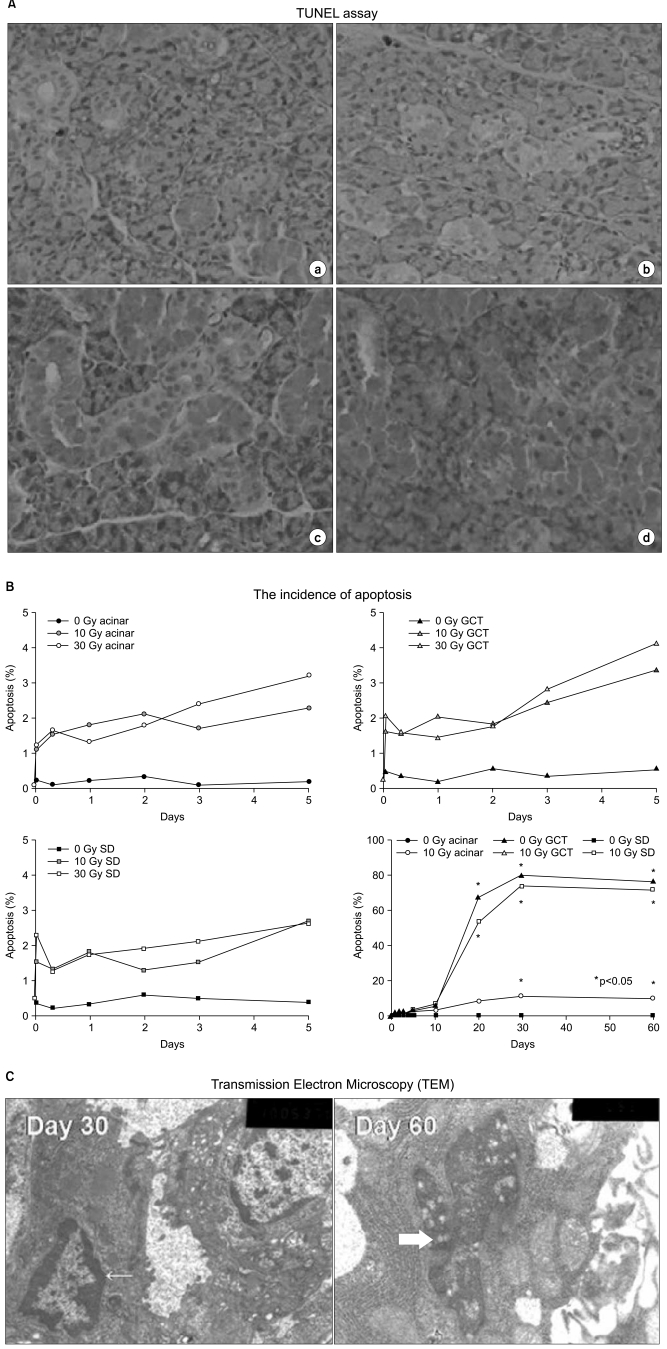
To evaluate the effect of X-ray irradiation on apoptosis and change of expression of aquaporin 5 (AQP5) and transforming growth factor-beta(TGF-beta) in the rat submandibular gland (SMG). MATERIALS AND METHODS: SMGs of 120 male Sprague-Dawley rats were irradiated with a single X-ray dose (3, 10, 20, or 30 Gy). At the early and late post-irradiation phase, apoptosis was measured by the terminal deoxynucleotidyl transferase biotin-dUTP nick end labeling (TUNEL) method, and expression of AQP5 and TGF-beta was determined by immunohistochemical staining. RESULTS: At the late post-irradiation phase, increased apoptosis was evident and marked decreases of expression of AQP5 expression by acinar cells and TGF-beta expression by ductal cells were evident. CONCLUSION: Apoptosis after X-ray irradiation develops relatively late in rat SMG. Irradiation reduces AQP5 and TGF-beta expression in different SMG cell types.
Keyword
MeSH Terms
Figure
Reference
-
1. Taylor JC, Terrell JE, Ronis DL, Fowler KE, Bishop C, Lambert MT, et al. Disability in patients with head and neck cancer. Arch Otolaryngol Head Neck Surg. 2004; 130:764–769. PMID: 15210560.
Article2. Konings AW, Coppes RP, Vissink A. On the mechanism of salivary gland radiosensitivity. Int J Radiat Oncol Biol Phys. 2005; 62:1187–1194. PMID: 15990024.
Article3. Abok K, Brunk U, Jung B, Ericsson J. Morphologic and histochemical studies on the differing radiosensitivity of ductular and acinar cells of the rat submandibular gland. Virchows Arch B Cell Pathol Incl Mol Pathol. 1984; 45:443–460. PMID: 6145251.
Article4. Peter B, Van Waarde MA, Vissink A, s-Gravenmade EJ, Konings AW. The role of secretory granules in the radiosensitivity of rat salivary gland acini--a morphological study. Radiat Res. 1994; 140:419–428. PMID: 7972696.5. Stephens LC, King GK, Peters LJ, Ang KK, Schultheiss TE, Jardine JH. Acute and late radiation injury in rhesus monkey parotid glands. Evidence of interphase cell death. Am J Pathol. 1986; 124:469–478. PMID: 3766705.6. Paardekooper GM, Cammelli S, Zeilstra LJ, Coppes RP, Konings AW. Radiation-induced apoptosis in relation to acute impairment of rat salivary gland function. Int J Radiat Biol. 1998; 73:641–648. PMID: 9690682.
Article7. Coppes RP, Zeilstra LJ, Kampinga HH, Konings AW. Early to late sparing of radiation damage to the parotid gland by adrenergic and muscarinic receptor agonists. Br J Cancer. 2001; 85:1055–1063. PMID: 11592779.
Article8. Zeilstra LJ, Vissink A, Konings AW, Coppes RP. Radiation induced cell loss in rat submandibular gland and its relation to gland function. Int J Radiat Biol. 2000; 76:419–429. PMID: 10757322.9. Dorr W, Hendry JH. Consequential late effects in normal tissues. Radiother Oncol. 2001; 61:223–231. PMID: 11730991.10. He X, Tse CM, Donowitz M, Alper SL, Gabriel SE, Baum BJ. Polarized distribution of key membrane transport proteins in the rat submandibular gland. Pflugers Arch. 1997; 433:260–268. PMID: 9064641.
Article11. Verrecchia F, Mauviel A. Transforming growth factor-beta signaling through the smad pathway: role in extracellular matrix gene expression and regulation. J Invest Dermatol. 2002; 118:211–215. PMID: 11841535.12. Nagler RM. The enigmatic mechanism of irradiation-induced damage to the major salivary glands. Oral Dis. 2002; 8:141–146. PMID: 12108758.
Article13. Guchelaar HJ, Vermes A, Meerwaldt JH. Radiation-induced xerostomia: pathophysiology, clinical course and supportive treatment. Support Care Cancer. 1997; 5:281–288. PMID: 9257424.
Article14. Muhvic-Urek M, Bralic M, Curic S, Pezelj-Ribaric S, Borcic J, Tomac J. Apoptosis after Imbalance between apoptosis and proliferation causes late radiation damage of salivary gland in mouse. Physiol Res. 2006; 55:89–95. PMID: 15857161.15. Li Z, Zhao D, Gong B, Xu Y, Sun H, Yang B, et al. Decreased saliva secretion and down-regulation of AQP5 in submandibular gland in irradiated rats. Radiat Res. 2006; 165:678–687. PMID: 16802868.
Article16. Krane CM, Melvin JE, Nguyen HV, Richardson L, Towne JE, Doetschman T, et al. Salivary acinar cells from aquaporin 5-deficient mice have decreased membrane water permeability and altered cell volume regulation. J Biol Chem. 2001; 276:23413–23420. PMID: 11290736.
Article17. Yi ES, Bedoya A, Lee H, Chin E, Kim SJ, Danielpour D, et al. Radiation-induced lung injury in vivo: expression of transforming growth factor-beta precedes fibrosis. Inflammation. 1996; 20:339–352. PMID: 8872498.
Article18. Rubin P, Finkelstein J, Shapiro D. Molecular biology mechanisms in the radiation induction of pulmonary injury syndromes: interrelationship between the alveolar macrophage and the septal fibroblast. Int J Radiat Oncol Biol Phys. 1992; 24:93–101. PMID: 1512168.
Article19. Anscher MS, Murase T, Prescott DM, Marks LB, Reisenbichler H, Bentel GC, et al. Changes in plasma TGF beta levels during pulmonary radiotherapy as a predictor of the risk of developing radiation pneumonitis. Int J Radiat Oncol Biol Phys. 1994; 30:671–676. PMID: 7928499.20. Flanders KC, Burmester JK. Medical applications of transforming growth factor-beta. Clin Med Res. 2003; 1:13–20. PMID: 15931280.21. Feltl D, Zavadova E, Pala M, Hozak P. Post-treatment plasma transforming growth factor beta 1 (TGF-beta1) level predicts for late morbidity in patients with advanced head and neck cancer. Neoplasma. 2005; 52:393–397. PMID: 16151583.22. Mukunoki H, Uchihashi K, Nishikawa Y. Effect of TGF-β on paracellulat transport in the rat submandibular gland acini. J Osaka Dent Univ. 2004; 38:41–52.23. Higuchi H. Regulation of paracellular barrier function in mouse submandibular gland acini. J Osaka Dent Univ. 2001; 35:13–25.24. Takai N, Uchihashi K, Miyao H, Murakami H, Yoshida Y. Chorda-evoked opening of tight junctions in rat submandibular salivary acini demonstrated by microperoxidase. Arch Oral Biol. 1995; 40:1077–1080. PMID: 8670027.
Article25. Uchio T. Permeability changes in the rat submandibular gland with experimental sialothiasis. J Osaka Odont Soc. 1988; 51:291–307.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Immunolocalization of AQP5 in Salivary Glands of Rat
- Study on the Changes in Distributions and Expressions of Aquaporin5 (AQP5) in Salivary Glands of Mice After Alcohol Ingestion
- Effect of irradiation on the acinar cells of submandibular gland in streptozotocin-induced diabetic rats
- Altered expression of sodium transporters and water channels following sympathetic and parasympathetic denervation in rat submandibular gland
- Effect of radiation dose variation on expression of caspase-3 in rat submandibular glands