J Adv Prosthodont.
2011 Sep;3(3):145-151. 10.4047/jap.2011.3.3.145.
The effect of immobilization of heparin and bone morphogenic protein-2 to bovine bone substitute on osteoblast-like cell's function
- Affiliations
-
- 1Department of Prosthodontics, School of Dentistry, Pusan National University, Yangsan, Korea.
- 2Department of Orthopedic Surgery & Rare Diseases Institute, Korea University Medical Center, Guro Hospital, Seoul, Korea.
- 3Department of Prosthodontics, Institute for Clinical Dental Research, Korea University Guro Hospital, Seoul, Korea. swshin@korea.ac.kr
- KMID: 2118124
- DOI: http://doi.org/10.4047/jap.2011.3.3.145
Abstract
- PURPOSE
This study was performed to investigate the ability of recombinant human-bone morphogenic protein-2 immobilized on a heparin-grafted bone substrate to enhance the osteoblastic functions.
MATERIALS AND METHODS
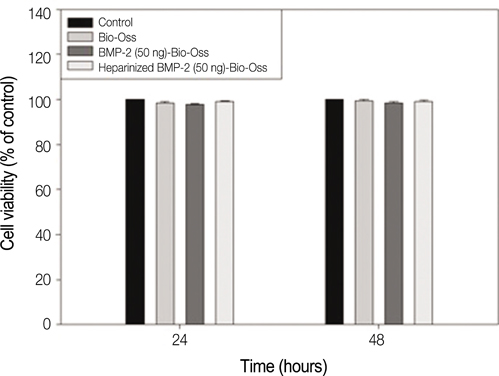
The Bio-Oss(R), not coated with any material, was used as a control group. In rhBMP-2-Bio-Oss(R) group, rhBMP-2 was coated with Bio-Oss(R) using only deep and dry methods (50 ng/mL, 24 h). In heparinized rhBMP-2-Bio-Oss(R) group, dopamine was anchored to the surface of Bio-Oss(R), and coated with heparin. rhBMP-2 was immobilized onto the heparinized- Bio-Oss(R) surface. The release kinetics of the rhBMP-2-Bio-Oss(R) and heparinized rhBMP-2-Bio-Oss(R) were analyzed using an enzyme-linked immunosorbent assay. The biological activities of the MG63 cells on the three groups were investigated via cytotoxicity assay, cell proliferation assay, alkaline phosphatase (ALP) measurement, and calcium deposition determination. Statistical comparisons were carried out by one-way ANOVA test. Differences were considered statistically significant at *P<.05 and **P<.001.
RESULTS
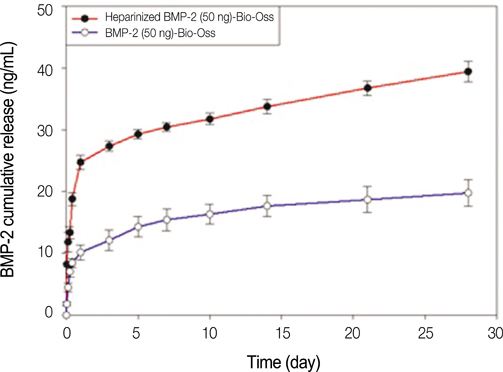
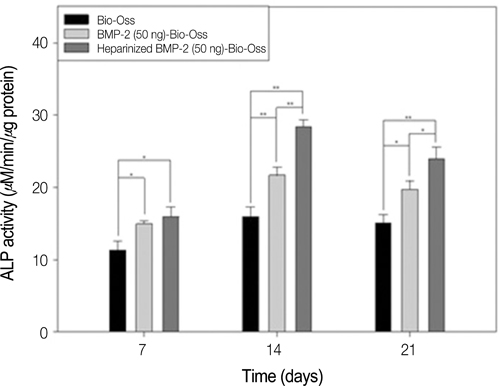
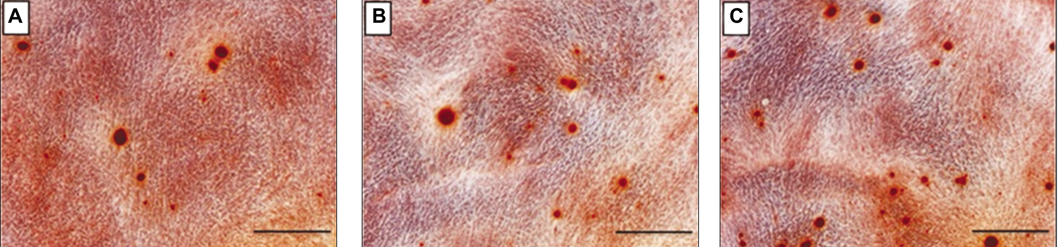
The heparinized rhBMP-2-Bio-Oss(R) showed more sustained release compared to the rhBMP-2-Bio-Oss(R) over an extended time. In the measurement of the ALP activity, the heparinized group showed a significantly higher ALP activity when compared with the non-heparinized groups (P<.05). The MG63 cells cultivated in the group with rhBMP-2 showed increased calcium deposition, and the MG63 cells from the heparinized group increased more than those that were cultivated in the non-heparinized groups.
CONCLUSION
Heparin increased the rhBMP-2 release amount and made sustained release possible, and heparinized Bio-Oss(R) with rhBMP-2 successfully improved the osteoblastic functions.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Effect of immobilization of the recombinant human bone morphogenetic protein 2 (rhBMP-2) on anodized implants coated with heparin for improving alveolar ridge augmentation in beagle dogs: Radiographic observations
So-Hyoun Lee, Jae-Young Jo, Mi-Jung Yun, Young-Chan Jeon, Jung-Bo Huh, Chang-Mo Jeong
J Korean Acad Prosthodont. 2013;51(4):307-314. doi: 10.4047/jkap.2013.51.4.307.
Reference
-
1. Amerio P, Vianale G, Reale M, Muraro R, Tulli A, Piattelli A. The effect of deproteinized bovine bone on osteoblast growth factors and proinflammatory cytokine production. Clin Oral Implants Res. 2010. 21:650–655.2. Del Fabbro M, Rosano G, Taschieri S. Implant survival rates after maxillary sinus augmentation. Eur J Oral Sci. 2008. 116:497–506.3. Mangano C, Scarano A, Iezzi G, Orsini G, Perrotti V, Mangano F, Montini S, Piccirilli M, Piattelli A. Maxillary sinus augmentation using an engineered porous hydroxyapatite: a clinical, histological, and transmission electron microscopy study in man. J Oral Implantol. 2006. 32:122–131.4. Esposito M, Grusovin MG, Kwan S, Worthington HV, Coulthard P. Interventions for replacing missing teeth: bone augmentation techniques for dental implant treatment. Cochrane Database Syst Rev. 2008. 3:CD003607.5. Fulmer NL, Bussard GM, Gampper TJ, Edlich RF. Anorganic bovine bone and analogs of bone mineral as implants for craniofacial surgery: a literature review. J Long Term Eff Med Implants. 1998. 8:69–78.6. Kübler A, Neugebauer J, Oh JH, Scheer M, Zöller JE. Growth and proliferation of human osteoblasts on different bone graft substitutes: an in vitro study. Implant Dent. 2004. 13:171–179.7. Turhani D, Weissenböck M, Watzinger E, Yerit K, Cvikl B, Ewers R, Thurnher D. In vitro study of adherent mandibular osteoblast-like cells on carrier materials. Int J Oral Maxillofac Surg. 2005. 34:543–550.8. Reddi AH. Symbiosis of biotechnology and biomaterials: applications in tissue engineering of bone and cartilage. J Cell Biochem. 1994. 56:192–195.9. Reddi AH. Bone morphogenetic proteins, bone marrow stromal cells, and mesenchymal stem cells. Maureen Owen revisited. Clin Orthop Relat Res. 1995. 313:115–119.10. Duneas N, Crooks J, Ripamonti U. Transforming growth factor-beta 1: induction of bone morphogenetic protein genes expression during endochondral bone formation in the baboon, and synergistic interaction with osteogenic protein-1 (BMP-7). Growth Factors. 1998. 15:259–277.11. Hall J, Sorensen RG, Wozney JM, Wikesjö UM. Bone formation at rhBMP-2-coated titanium implants in the rat ectopic model. J Clin Periodontol. 2007. 34:444–451.12. Chen D, Zhao M, Mundy GR. Bone morphogenetic proteins. Growth Factors. 2004. 22:233–241.13. Bessa PC, Balmayor ER, Azevedo HS, Nürnberger S, Casal M, van Griensven M, Reis RL, Redl H. Silk fibroin microparticles as carriers for delivery of human recombinant BMPs. Physical characterization and drug release. J Tissue Eng Regen Med. 2010. 4:349–355.14. Sykaras N, Iacopino AM, Triplett RG, Marker VA. Effect of recombinant human bone morphogenetic protein-2 on the osseointegration of dental implants: a biomechanics study. Clin Oral Investig. 2004. 8:196–205.15. Sellers RS, Zhang R, Glasson SS, Kim HD, Peluso D, D'Augusta DA, Beckwith K, Morris EA. Repair of articular cartilage defects one year after treatment with recombinant human bone morphogenetic protein-2 (rhBMP-2). J Bone Joint Surg Am. 2000. 82:151–160.16. Bax BE, Wozney JM, Ashhurst DE. Bone morphogenetic protein-2 increases the rate of callus formation after fracture of the rabbit tibia. Calcif Tissue Int. 1999. 65:83–89.17. Sheehan JP, Kallmes DF, Sheehan JM, Jane JA Jr, Fergus AH, diPierro CG, Simmons NE, Makel DD, Helm GA. Molecular methods of enhancing lumbar spine fusion. Neurosurgery. 1996. 39:548–554.18. King GN, King N, Cruchley AT, Wozney JM, Hughes FJ. Recombinant human bone morphogenetic protein-2 promotes wound healing in rat periodontal fenestration defects. J Dent Res. 1997. 76:1460–1470.19. Sellers RS, Peluso D, Morris EA. The effect of recombinant human bone morphogenetic protein-2 (rhBMP-2) on the healing of full-thickness defects of articular cartilage. J Bone Joint Surg Am. 1997. 79:1452–1463.20. Zellin G, Linde A. Importance of delivery systems for growth-stimulatory factors in combination with osteopromotive membranes. An experimental study using rhBMP-2 in rat mandibular defects. J Biomed Mater Res. 1997. 35:181–190.21. Kim J, Park Y, Tae G, Lee KB, Hwang CM, Hwang SJ, Kim IS, Noh I, Sun K. Characterization of low-molecular-weight hyaluronic acid-based hydrogel and differential stem cell responses in the hydrogel microenvironments. J Biomed Mater Res A. 2009. 88:967–975.22. Kim J, Kim IS, Cho TH, Lee KB, Hwang SJ, Tae G, Noh I, Lee SH, Park Y, Sun K. Bone regeneration using hyaluronic acid-based hydrogel with bone morphogenic protein-2 and human mesenchymal stem cells. Biomaterials. 2007. 28:1830–1837.23. Lee TC, Ho JT, Hung KS, Chen WF, Chung YH, Yang YL. Bone morphogenetic protein gene therapy using a fibrin scaffold for a rabbit spinal-fusion experiment. Neurosurgery. 2006. 58:373–380. discussion 373-80.24. Jeon O, Song SJ, Yang HS, Bhang SH, Kang SW, Sung MA, Lee JH, Kim BS. Long-term delivery enhances in vivo osteogenic efficacy of bone morphogenetic protein-2 compared to short-term delivery. Biochem Biophys Res Commun. 2008. 369:774–780.25. Sasisekharan R, Ernst S, Venkataraman G. On the regulation of fibroblast growth factor activity by heparin-like glycosaminoglycans. Angiogenesis. 1997. 1:45–54.26. Perets A, Baruch Y, Weisbuch F, Shoshany G, Neufeld G, Cohen S. Enhancing the vascularization of three-dimensional porous alginate scaffolds by incorporating controlled release basic fibroblast growth factor microspheres. J Biomed Mater Res A. 2003. 65:489–497.27. Ishibe T, Goto T, Kodama T, Miyazaki T, Kobayashi S, Takahashi T. Bone formation on apatite-coated titanium with incorporated BMP-2/heparin in vivo. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009. 108:867–875.28. von Walter M, Herren C, Gensior TJ, Steffens GC, Hermanns-Sachweh B, Jahnen-Dechent W, Rüger M, Erli HJ. Biomimetic modification of the TiO(2)/glass composite Ecopore with heparinized collagen and the osteoinductive factor BMP-2. Acta Biomater. 2008. 4:997–1004.29. Kodama T, Goto T, Miyazaki T, Takahashi T. Bone formation on apatite-coated titanium incorporated with bone morphogenetic protein and heparin. Int J Oral Maxillofac Implants. 2008. 23:1013–1019.30. Ishibe T, Goto T, Kodama T, Miyazaki T, Kobayashi S, Takahashi T. Bone formation on apatite-coated titanium with incorporated BMP-2/heparin in vivo. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009. 108:867–875.31. Kim SE, Song SH, Yun YP, Choi BJ, Kwon IK, Bae MS, Moon HJ, Kwon YD. The effect of immobilization of heparin and bone morphogenic protein-2 (BMP-2) to titanium surfaces on inflammation and osteoblast function. Biomaterials. 2011. 32:366–373.32. Tadjoedin ES, de Lange GL, Bronckers AL, Lyaruu DM, Burger EH. Deproteinized cancellous bovine bone (Bio-Oss) as bone substitute for sinus floor elevation. A retrospective, histomorphometrical study of five cases. J Clin Periodontol. 2003. 30:261–270.33. Bessho K, Konishi Y, Kaihara S, Fujimura K, Okubo Y, Iizuka T. Bone induction by Escherichia coli -derived recombinant human bone morphogenetic protein-2 compared with Chinese hamster ovary cell-derived recombinant human bone morphogenetic protein-2. Br J Oral Maxillofac Surg. 2000. 38:645–649.34. Tokuhara Y, Wakitani S, Imai Y, Kawaguchi A, Fukunaga K, Kim M, Kadoya Y, Takaoka K. Repair of experimentally induced large osteochondral defects in rabbit knee with various concentrations of Escherichia coli-derived recombinant human bone morphogenetic protein-2. Int Orthop. 2010. 34:761–767.35. Lee JH, Kim CS, Choi KH, Jung UW, Yun JH, Choi SH, Cho KS. The induction of bone formation in rat calvarial defects and subcutaneous tissues by recombinant human BMP-2, produced in Escherichia coli. Biomaterials. 2010. 31:3512–3519.36. Ho YC, Mi FL, Sung HW, Kuo PL. Heparin-functionalized chitosan-alginate scaffolds for controlled release of growth factor. Int J Pharm. 2009. 376:69–75.37. Lin H, Zhao Y, Sun W, Chen B, Zhang J, Zhao W, Xiao Z, Dai J. The effect of crosslinking heparin to demineralized bone matrix on mechanical strength and specific binding to human bone morphogenetic protein-2. Biomaterials. 2008. 29:1189–1197.38. Park YJ, Kim KH, Lee JY, Ku Y, Lee SJ, Min BM, Chung CP. Immobilization of bone morphogenetic protein-2 on a nanofibrous chitosan membrane for enhanced guided bone regeneration. Biotechnol Appl Biochem. 2006. 43:17–24.39. Turksen K, Bhargava U, Moe HK, Aubin JE. Isolation of monoclonal antibodies recognizing rat bone-associated molecules in vitro and in vivo. J Histochem Cytochem. 1992. 40:1339–1352.40. van den Beucken JJ, Walboomers XF, Boerman OC, Vos MR, Sommerdijk NA, Hayakawa T, Fukushima T, Okahata Y, Nolte RJ, Jansen JA. Functionalization of multilayered DNA-coatings with bone morphogenetic protein 2. J Control Release. 2006. 113:63–72.41. Bancroft GN, Sikavitsas VI, van den Dolder J, Sheffield TL, Ambrose CG, Jansen JA, Mikos AG. Fluid flow increases mineralized matrix deposition in 3D perfusion culture of marrow stromal osteoblasts in a dose-dependent manner. Proc Natl Acad Sci USA. 2002. 99:12600–12605.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Evaluation of deproteinized bovine bone mineral as a bone graft substitute: A comparative analysis of basic characteristics of three commercially available bone substitutes
- Bone Substitutes: From Basic to Current Update
- Bone Cell Viability after Exposure to Air
- Bone Morphogenic Protein-2-Conjugated Three-DimensionalPrinted Poly (L-Lactic Acid) (PLLA) Scaffold is likely Promising as an Effective Bone Substitute
- The BMPs expression and histomorphometric study of beta-TCP /rhBMP-2 Grafting on the rabbit cranial bone defects