Korean J Obstet Gynecol.
2010 Sep;53(9):816-824. 10.5468/kjog.2010.53.9.816.
Interrelationship of aging and mitochondrial DNA deletion in luteinized granulosa cells
- Affiliations
-
- 1Department of Obstetrics and Gynecology, Konyang University Hospital, Daejeon, Korea. 3884@medigate.net
- 2Konyang University Myunggok Medical Research Institute, Daejeon, Korea.
- 3Daejeon Maria Infertility Clinic, Daejeon, Korea.
- KMID: 2077999
- DOI: http://doi.org/10.5468/kjog.2010.53.9.816
Abstract
OBJECTIVE
Many clinical trials have proven the close relationship between the loss of human mitochondrial DNA and aging process. The purpose of this study was to evaluate the different types of mitochondrial DNA deletion and its frequency in luteinized granulosa cells in different aged groups of women undergoing in vitro fertilization (IVF).
METHODS
The ovum pick up was done in 51 women who participated in Konyang University IVF program, and mitochondrial DNAs extracted from luteinized granulosa cells, were screened to search for different types of deletion and its frequency. The deleted mitochondrial DNA were analyzed by polymerase chain reaction method. DNA sequencing was performed to reveal exact deletion point.
RESULTS
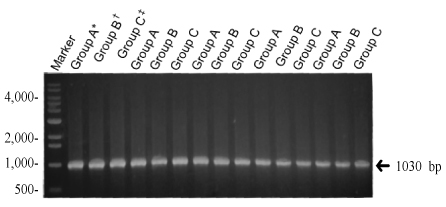
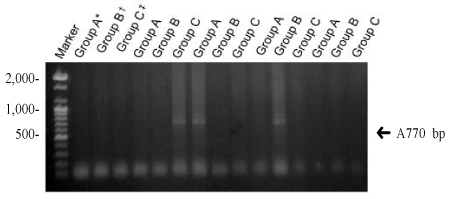
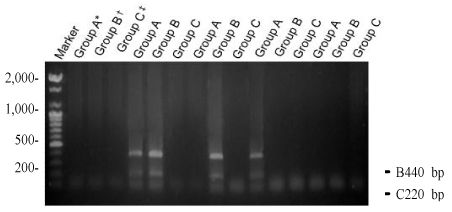
Three different types of deletions (4,977 bp, 7,150 bp, and 5,777 bp) were confirmed. To find the difference between the aged groups, we have divided women into groups younger than 32 years, between 32 to 37 years, and older than 37 years. The deletion of 4,977 bp was 60.9% (14/23) in younger than 32 years, 46.2% (6/13) in 32 to 37 years, 46.7% (7/15) in older than 37 years. There was no statistical significance between aged groups and the incidence of the deletion. The deletion of 7150 bp was 34.8% (8/23), in younger than 32 years, 30.8% (4/13) in 32 to 37 years, 40% (6/15) in older than 37 years. We investigated relationship between the frequency of deletion and the aging, but there was no statistical significance. In case of 5,777 bp, the deletion was 43.5% (10/23) in younger than 32 years, 30.8% (4/13) in 32 to 37 years, 53.3% (8/15) in older than 37 years. It showed no statistical significance as well as other types.
CONCLUSION
In this study we have found three different types of deletion of mitochondrial DNA obtained from luteinized granulosa cells in women with infertility. There were no significant differnces of each type of deletion in 3 different aged groups of infantile women. The limitation of this study is that the analyze were done qualitatively. If we could provide the quantitative analyze it could be applied clinically.
MeSH Terms
Figure
Reference
-
1. Cortopassi GA, Arnheim N. Detection of a specific mitochondrial DNA deletion in tissues of older humans. Nucleic Acids Res. 1990. 18:6927–6933.2. Cortopassi GA, Shibata D, Soong NW, Arnheim N. A pattern of accumulation of a somatic deletion of mitochondrial DNA in aging human tissues. Proc Natl Acad Sci USA. 1992. 89:7370–7374.3. Yen TC, Pang CY, Hsieh RH, Su CH, King KL, Wei YH. Age-dependent 6kb deletion in human liver mitochondrial DNA. Biochem Int. 1992. 26:457–468.4. Hayakawa M, Sugiyama S, Hattori K, Takasawa M, Ozawa T. Age-associated damage in mitochondrial DNA in human hearts. Mol Cell Biochem. 1993. 119:95–103.5. Richter C. Reactive oxygen and DNA damage in mitochondria. Mutat Res. 1992. 275:249–255.6. Chance B, Sies H, Boveris A. Hydroperoxide metabolism in mammalian organs. Physiol Rev. 1979. 59:527–605.7. Harman D. The biologic clock: the mitochondria? J Am Geriatr Soc. 1972. 20:145–147.8. Clayton DA. Replication and transcription of vertebrate mitochondrial DNA. Annu Rev Cell Biol. 1991. 7:453–478.9. Hammans SR. Mitochondrial DNA and disease. Essays Biochem. 1994. 28:99–112.10. Yakes FM, Van Houten B. Mitochondrial DNA damage is more extensive and persists longer than nuclear DNA damage in human cells following oxidative stress. Proc Natl Acad Sci USA. 1997. 94:514–519.11. Grossman LI, Shoubridge EA. Mitochondrial genetics and human disease. Bioessays. 1996. 18:983–991.12. Ozawa T. Genetic and functional changes in mitochondria associated with aging. Physiol Rev. 1997. 77:425–464.13. Meissner C, von Wurmb N, Oehmichen M. Detection of the age-dependent 4977 bp deletion of mitochondrial DNA. A pilot study. Int J Legal Med. 1997. 110:288–291.14. Larsson NG, Clayton DA. Molecular genetic aspects of human mitochondrial disorders. Annu Rev Genet. 1995. 29:151–178.15. Cortopassi GA, Arnheim N. Using the polymerase chain reaction to estimate mutation frequencies and rates in human cells. Mutat Res. 1992. 277:239–249.16. Corral-Debrinski M, Horton T, Lott MT, Shoffner JM, Beal MF, Wallace DC. Mitochondrial DNA deletions in human brain: regional variability and increase with advanced age. Nat Genet. 1992. 2:324–329.17. Kitagawa T, Suganuma N, Nawa A, Kikkawa F, Tanaka M, Ozawa T, et al. Rapid accumulation of deleted mitochondrial deoxyribonucleic acid in postmenopausal ovaries. Biol Reprod. 1993. 49:730–736.18. Samuels DC, Schon EA, Chinnery PF. Two direct repeats cause most human mtDNA deletions. Trends Genet. 2004. 20:393–398.19. Chomyn A, Attardi G. MtDNA mutations in aging and apoptosis. Biochem Biophys Res Commun. 2003. 304:519–529.20. Khrapko K, Bodyak N, Thilly WG, van Orsouw NJ, Zhang X, Coller HA, et al. Cell-by-cell scanning of whole mitochondrial genomes in aged human heart reveals a significant fraction of myocytes with clonally expanded deletions. Nucleic Acids Res. 1999. 27:2434–2441.21. Seifer DB, Gardiner AC, Ferreira KA, Peluso JJ. Apoptosis as a function of ovarian reserve in women undergoing in vitro fertilization. Fertil Steril. 1996. 66:593–598.22. Klein NA, Battaglia DE, Woodruff TK, Padmanabhan V, Giudice LC, Bremner WJ, et al. Ovarian follicular concentrations of activin, follistatin, inhibin, insulin-like growth factor I (IGF-I), IGF-II, IGF-binding protein-2 (IGFBP-2), IGFBP-3, and vascular endothelial growth factor in spontaneous menstrual cycles of normal women of advanced reproductive age. J Clin Endocrinol Metab. 2000. 85:4520–4525.23. Hsieh RH, Tsai NM, Au HK, Chang SJ, Wei YH, Tzeng CR. Multiple rearrangements of mitochondrial DNA in unfertilized human oocytes. Fertil Steril. 2002. 77:1012–1017.24. Au HK, Lin SH, Huang SY, Yeh TS, Tzeng CR, Hsieh RH. Deleted mitochondrial DNA in human luteinized granulosa cells. Ann N Y Acad Sci. 2005. 1042:136–141.25. Seifer DB, DeJesus V, Hubbard K. Mitochondrial deletions in luteinized granulosa cells as a function of age in women undergoing in vitro fertilization. Fertil Steril. 2002. 78:1046–1048.26. Lee HC, Pang CY, Hsu HS, Wei YH. Differential accumulations of 4,977 bp deletion in mitochondrial DNA of various tissues in human ageing. Biochim Biophys Acta. 1994. 1226:37–43.27. Anderson S, Bankier AT, Barrel BG. Sequence and organization of the human mitochondrial genome. Nature. 1981. 290:457–465.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical Significance of Apoptotic Rates of Human Luteinized Granulosa Cells in In Vitro Fertilization and Embryo Transfer
- Interrelationship of ageing of human ovary and mitochondrial DNA deletion
- Flow cytometric quantification of apoptotosis in luteinized granulosa cells recovered from patients participating in an in vitro fertilization program
- Apoptosis of human luteinized granulosa cell via change of mitochondria
- The Effect of Nitric Oxide on Apoptosis in Human Luteinized Granulosa Cells