Korean J Physiol Pharmacol.
2009 Jun;13(3):189-194. 10.4196/kjpp.2009.13.3.189.
Forskolin Changes the Relationship between Cytosolic Ca2+ and Contraction in Guinea Pig Ileum
- Affiliations
-
- 1Department of Internal Medicine, Gangnung Asan Hospital, Gangnung 210-711, Korea.
- 2Department of Physiology, College of Medicine, Kwandong University, Gangneung 210-751, Korea. skwon2028@kd.ac.kr
- KMID: 2071670
- DOI: http://doi.org/10.4196/kjpp.2009.13.3.189
Abstract
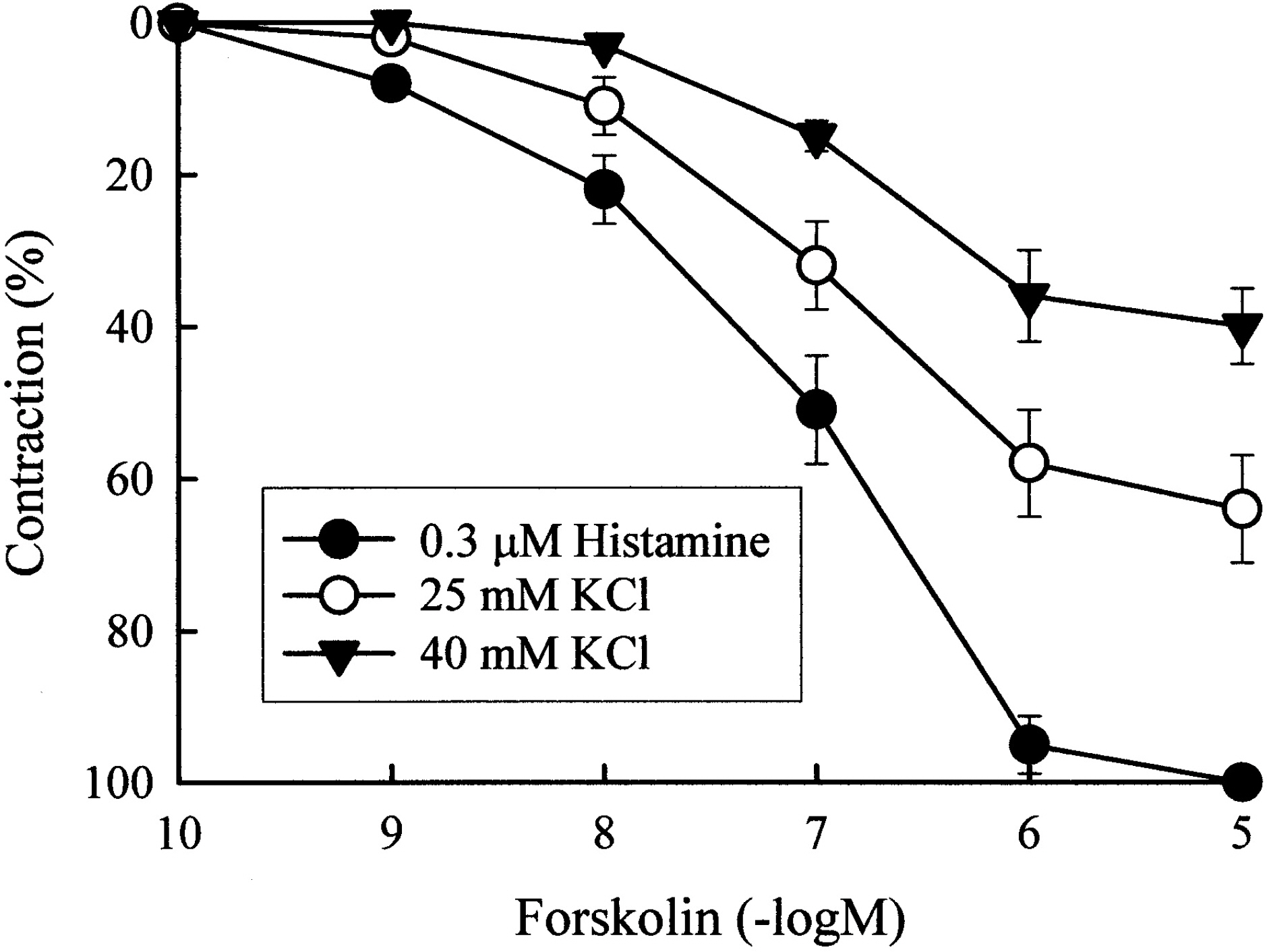
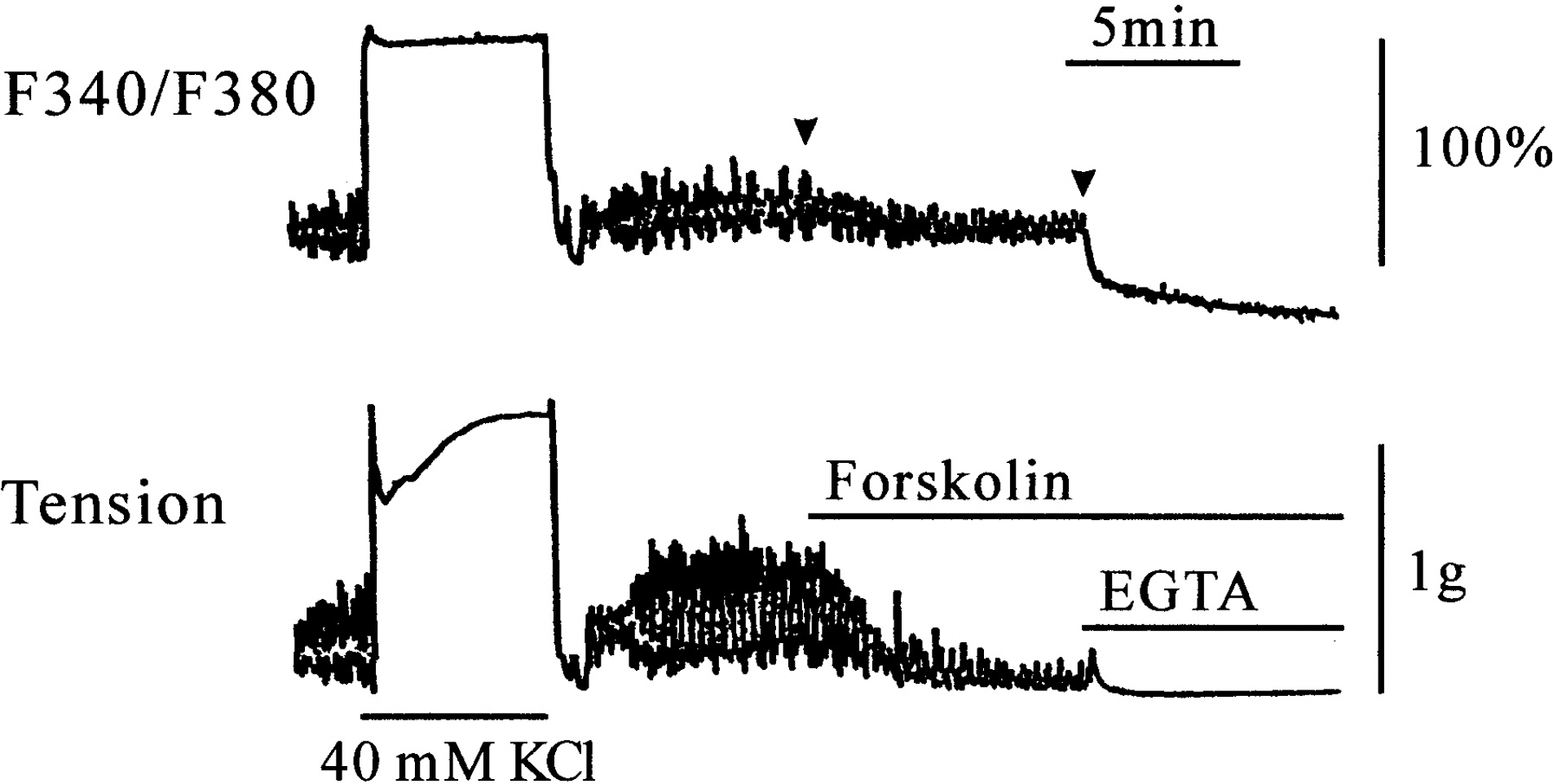
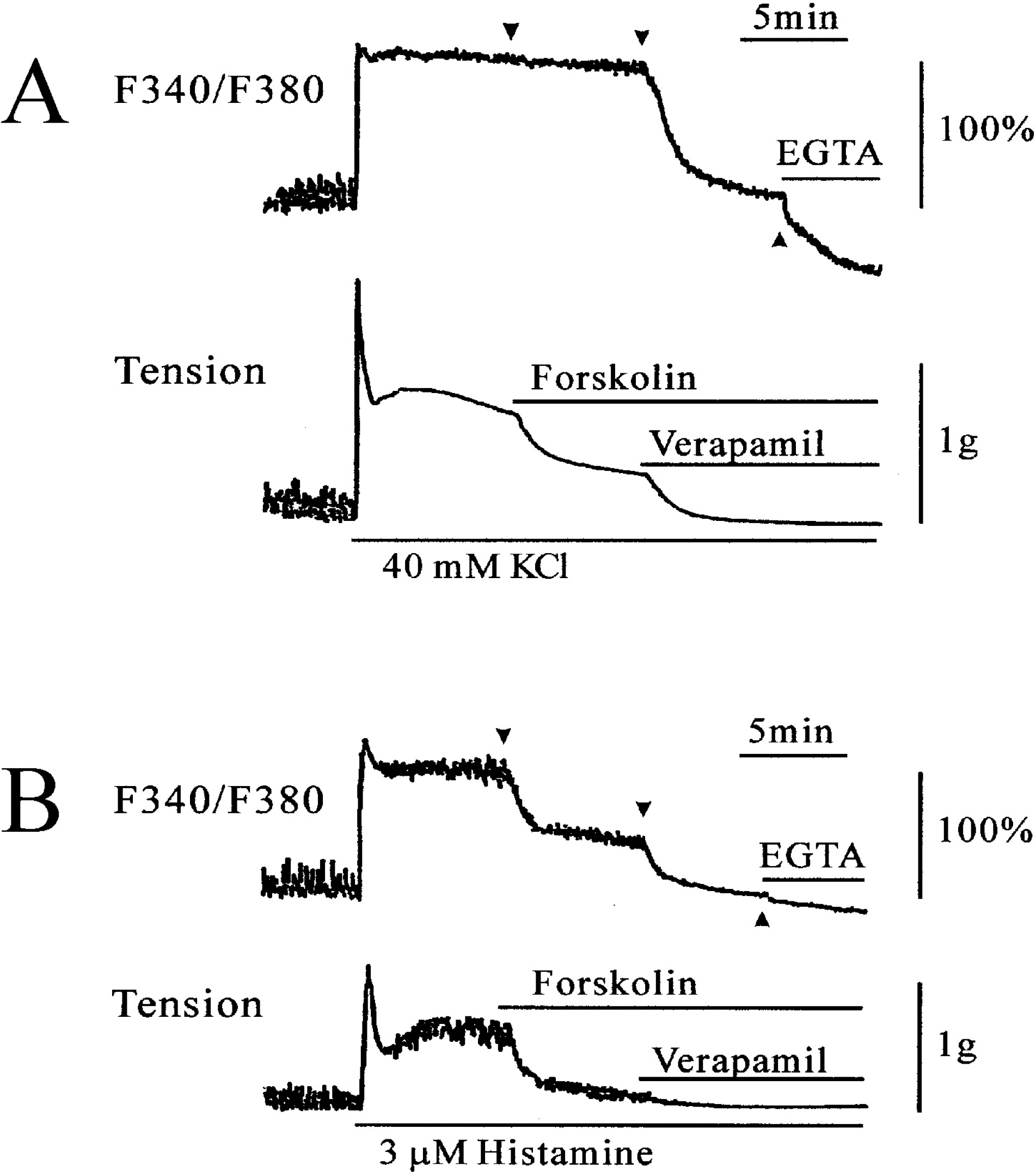
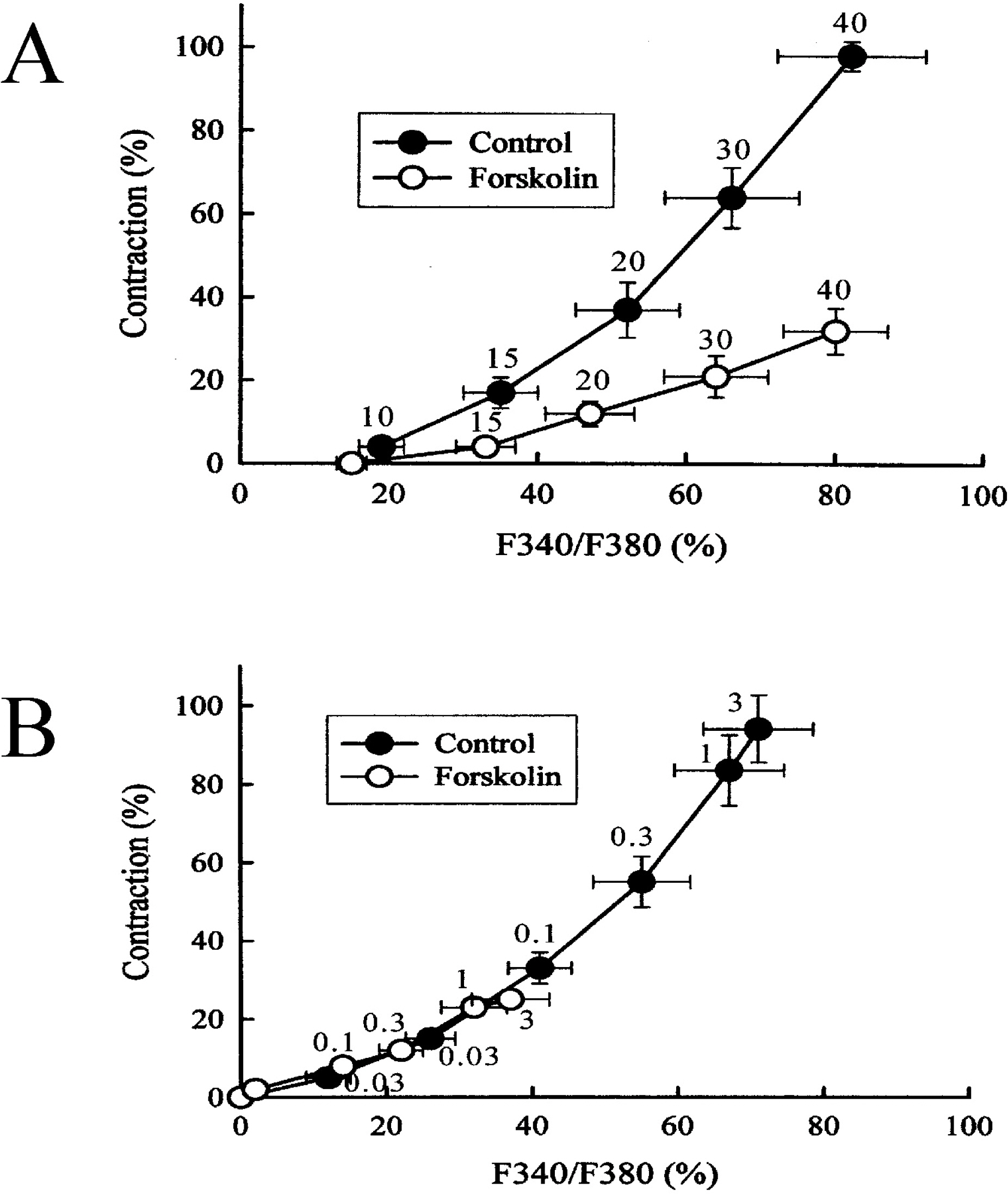
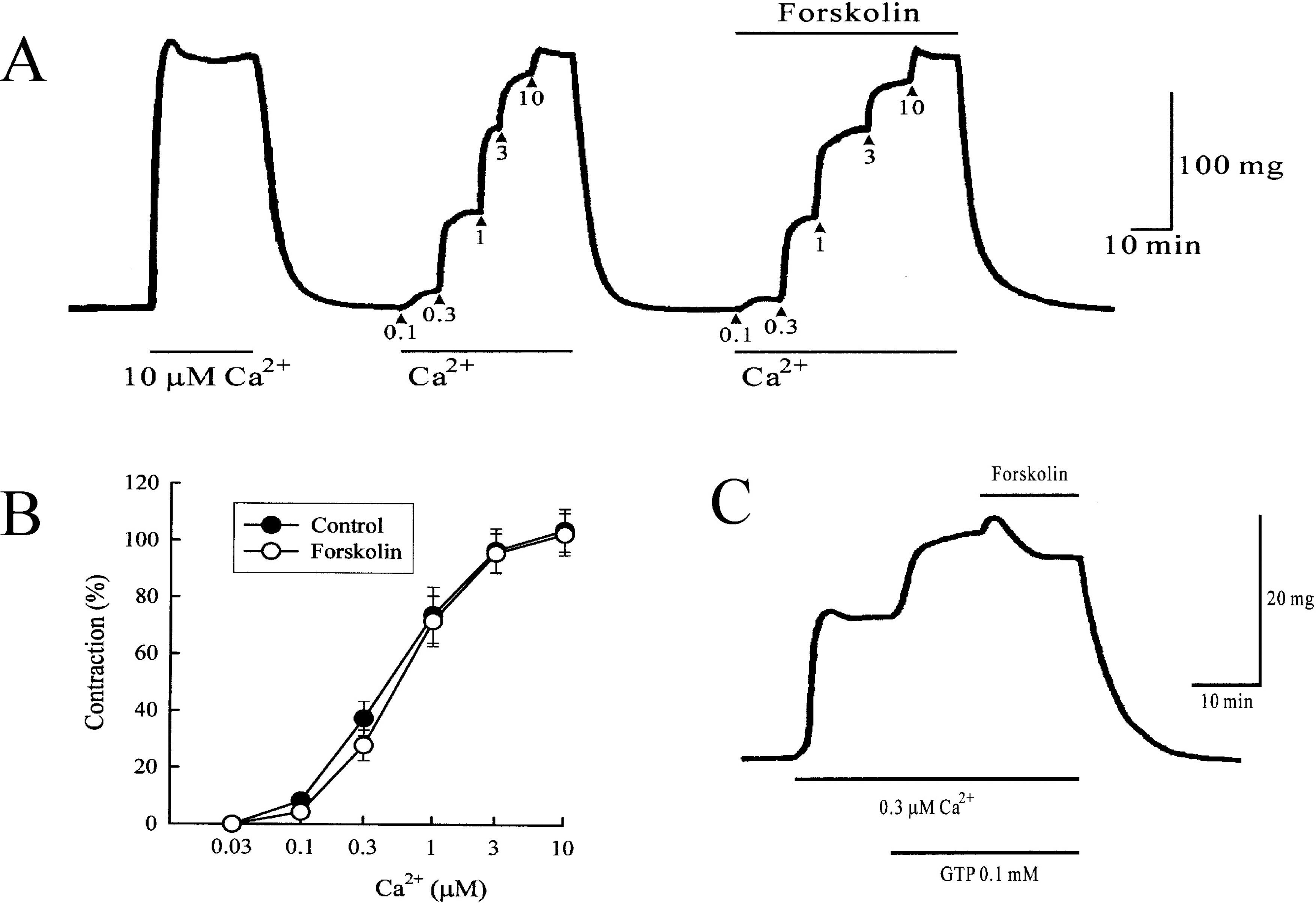
- This study was designed to clarify the mechanism of the inhibitory effect of forskolin on contraction, cytosolic Ca2+ level ([Ca2+]i), and Ca2+ sensitivity in guinea pig ileum. Forskolin (0.1 nM~10 micrometer) inhibited high K+ (25 mM and 40 mM)- or histamine (3 micrometer)-evoked contractions in a concentration-dependent manner. Histamine-evoked contractions were more sensitive to forskolin than high K+-evoked contractions. Spontaneous changes in [Ca2+]i and contractions were inhibited by forskolin (1 micrometer) without changing the resting [Ca2+]i. Forskoln (10 micrometer) inhibited muscle tension more strongly than [Ca2+]i stimulated by high K+, and thus shifted the [Ca2+]i-tension relationship to the lower-right. In histamine-stimulated contractions, forskolin (1 micrometer) inhibited both [Ca2+]i and muscle tension without changing the [Ca2+]i-tension relationship. In alpha-toxin-permeabilized tissues, forskolin (10 micrometer) inhibited the 0.3 micrometer Ca2+-evoked contractions in the presence of 0.1 mM GTP, but showed no effect on the Ca2+-tension relationship. We conclude that forskolin inhibits smooth muscle contractions by the following two mechanisms: a decrease in Ca2+ sensitivity of contractile elements in high K+-stimulated muscle and a decrease in [Ca2+]i in histamine-stimulated muscle.
Keyword
MeSH Terms
Figure
Reference
-
Adelstein RS., Conti MA., Hathaway DR., Klee CB. Phosphorylation of smooth muscle myosin light chain kinase by the catalytic subunit of adenosine 3':5'-monophosphate-dependent protein kinase. J Biol Chem. 253:8347–8350. 1978.Ahn HY., Kang SE., Chang KC., Karaki H. Dibutyryl cyclic AMP and forskolin inhibit phosphatidyl-inositol hydrolysis, Ca2+ influx and contraction in vascular smooth muscle. Jpn J Pharmacol. 59:263–265. 1992.Andersson R., Nilsson K. Role of cyclic nucleotides: metabolism and mechanical activity in smooth muscle. In Biochemistry of Smooth Muscle (edited by Stephens NL), 263−291. 1977.Bhalla RC., Webb RC., Singh D., Brock T. Role of cyclic AMP in rat aortic microsomal phosphorylation and calcium uptake. Am J Physiol. 234:H508–H514. 1978.
ArticleBulbring E., Tomita T. Catecholamine action on smooth muscle. Pharmacol Rev. 39:49–96. 1987.Cooper DM. Molecular and cellular requirements for the regulation adenylate cyclases by calcium. Biochem Soc Trans. 31:912–915. 2003.Eckly-Michel A., Martin V., Lugnier C. Involvement of cyclic nucleotide-dependent protein kinases in cyclic AMP-mediated vasorelaxation. Br J Pharmacol. 122:158–164. 1997.
ArticleHall IP., Donaldson J., Hill SJ. Inhibition od histamine-stimulated inositol phospholipids hydrolysis by agents which increase cyclic AMP levels in bovine tracheal smooth muscle. Br J Pharmacol. 97:603–613. 1989.Ise S., Nishimura J., Hirano K., Hara N., Kanaide H. Theophylline attenuates Ca2+ sensitivity and modulates BK channels in porcine tracheal smooth muscle. Br J Pharmacol. 140:939–947. 2003.Karaki H. Localization and sensitivity in vascular smooth muscle. Trends Pharmacol Sci. 10:320–325. 1989.Karaki H., Weiss GB. Calcium channels in smooth muscle. Gastroenterology. 87:960–970. 1984.
ArticleKwon SC., Ozaki H., Hori M., Karaki H. Isoproterenol changes the relationship between cytosolic Ca2+ and contraction in guineapig taneia caecum. Jpn J Pharmacol. 61:57–64. 1993.Kwon SC., Ozaki H., Karaki H. NO donor sodium nitroprusside inhibits excitation-contraction coupling in guinea pig taenia coli. Am J Physiol. 279:G1235–G1241. 2000a.
ArticleKwon SC., Park KY., Ahn DS., Lee HY., Kang BS. The effect of NO donor on contraction, cytosolic Ca2+ level and ionic currents in guinea-pig ileal smooth muscle. Korean J Physiol Pharmacol. 4:33–40. 2000b.Kushida M., Takeuchi T., Fujita A., Hata F. Dependence of Ca2+- induced contraction in a-toxin-permeabilized preparations of rat femoral artery. J Pharmacol Sci. 93:171–179. 2003.Lincoln TM., Cornwell TL. Towards an understanding of the mechanism of action of cyclic AMP and cyclic GMP in smooth muscle relaxation. Blood Vessels. 28:129–137. 1991.
ArticleMorales S., Camello PJ., Mawe GM., Pozo MJ. Cyclic AMP-mediated inhibition of gallbladder contractility: role of K+ channel activation and Ca2+ handling. Br J Pharmacol. 143:994–1005. 2004.Murray KJ. Cyclic AMP and mechanism of vasodilation. Pharmacol Ther. 47:329–354. 1990.Murthy KS. Signaling for contraction and relaxation in smooth muscle of the gut. Annu Rev Physiol. 68:345–374. 2006.
ArticleNishikori K., Maeno H. Close relationship between adenosine3': 5'-monophosphate-dependent endogenous phosphorylation of a specific protein and stimulation of calcium uptake in rat uterine microsomes. J Biol Chem. 254:6099–6106. 1979.Nishimura J., van Breemen C. Direct regulation of smooth muscle contractile elements by second messengers. Biochem Biophys Res Commun. 163:929–935. 1989.
ArticleOzaki H., Blondfield DP., Hori M., Sanders KM., Publicover NG. Cyclic AMP-mediated regulation of excitation-contraction coupling in canine gastric smooth muscle. J Physiol. 447:351–372. 1992.
ArticleOzaki H., Hori M., Kim YS., Kwon SC., Ahn DS., Nakazawa H., Kobaysahi M., Karaki H. Inhibitory mechanism of xestospongin-C on contraction and ion channels in the intestinal smooth muscle. Br J Pharmacol. 137:1207–1212. 2002.
ArticlePfitzer G., Hofmann F., DiSalvo J., Ruegg JC. cGMP and cAMP inhibit tension development in skinned coronary arteries. Pflugers Arch. 401:277–280. 1984.
ArticlePorter M., Evans MC., Miner AS., Berg KM., Ward KR., Ratz PH. Convergence of Ca2+-desensitizing mechanisms activated by forskolin and phenylephrine, but not 8-bromo-cGMP. Am J Physiol. 290:C1552–C1559. 2006.Ratz PH. Receptor activation induces short-term modulation of arterial sontractions: memory in vascular smooth muscle. Am J Physiol. 269:C417–C423. 1999.Rembold CM., Chen XL. Mechanisms responsible for forskolin-induced relaxation of rat tail artery. Hypertension. 31:872–877. 1998.
ArticleRembold CM., O'Connor MJ., Clarkson M., Wardle RL., Murphy RA. HSP20 phosphorylation in nitroglycerin- and forskolin-induced sustained reductions in swine carotid artery. J Appl Physiol. 91:1460–1466. 2001.Seamon KB., Daly JW. Forskolin: its biological and chemical properties. Adv Cyclic Nucleotide Protein Phosphoryl Res. 20:1–150. 1986.Wellman GC., Santana LF., Bonev AD., Nelson MT. Role of phospholamban in the modulation of arterial Ca2+ sparks and Ca2+-activated K+ channels by cAMP. Am J Physiol. 281:C1029–C1037. 2001.Woodrum DA., Brophy CM., Wingard CJ., Beall A., Rasmussen H. Phosphorylation events associated with cyclic nucleotide-dependent inhibition of smooth muscle contraction. Am J Physiol. 277:H931–H939. 1999.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The effect of carbon monoxide on contraction, cytosolic Ca2+ level and ionic currents in guinea pig ileal smooth muscle
- The effect of NO donor on contraction, cytosolic Ca2+ level and ionic currents in guinea-pig ileal smooth muscle
- Effects of ryanodine on the intracellular Na+ activity and tension and action potentials of rat and guinea pig cardiac ventricular muscles
- Effects of Ketamine on Intracellular Ca2+ Pooling in Guinea Pig Trachea
- Investigation into the Effects of Mosapride on Motility of Guinea Pig Stomach, Ileum, and Colon