Ann Surg Treat Res.
2014 Feb;86(2):68-75. 10.4174/astr.2014.86.2.68.
Antichemosensitizing effect of resveratrol in cotreatment with oxaliplatin in HCT116 colon cancer cell
- Affiliations
-
- 1Department of Surgery, Dankook University School of Medicine, Cheonan, Korea. dkpark@dankook.ac.kr
- KMID: 1908500
- DOI: http://doi.org/10.4174/astr.2014.86.2.68
Abstract
- PURPOSE
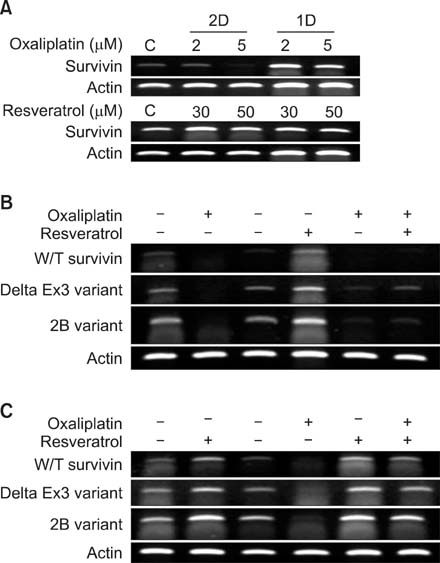
Although oxaliplatin is one of the most widely used chemotherapeutic agents for the treatment of advanced stages of colorectal cancers in clinic, cancer cells often develop oxaliplatin drug resistance. Thus, overcoming oxaliplatin drug resistance is a major issue in the successful treatment for advanced stages of colorectal malignancy. In order to maximize oxaliplatin therapy, we examined whether resveratrol, a natural phytochemical known to have chemopreventive effects on cancers, can have a chemosensitizing effect upon cotreatment with oxaliplatin. Survivin, a small inhibitor of apoptosis protein (IAP), expression is examined using HCT116 colon cancer cells.
METHODS
In order to examine resveratrol chemosensitizing effect upon oxaliplatin cotreatment, survivin transcripts and protein expression, cell proliferation, and apoptotic responses were evaluated using HCT116 cells. Reverse transcription polymerase chain reaction (RT-PCR), Western blot, crystal violet staining analyses were performed. For survivin specific inhibition, YM155 molecule was used.
RESULTS
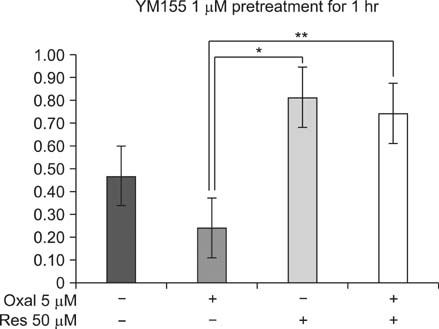
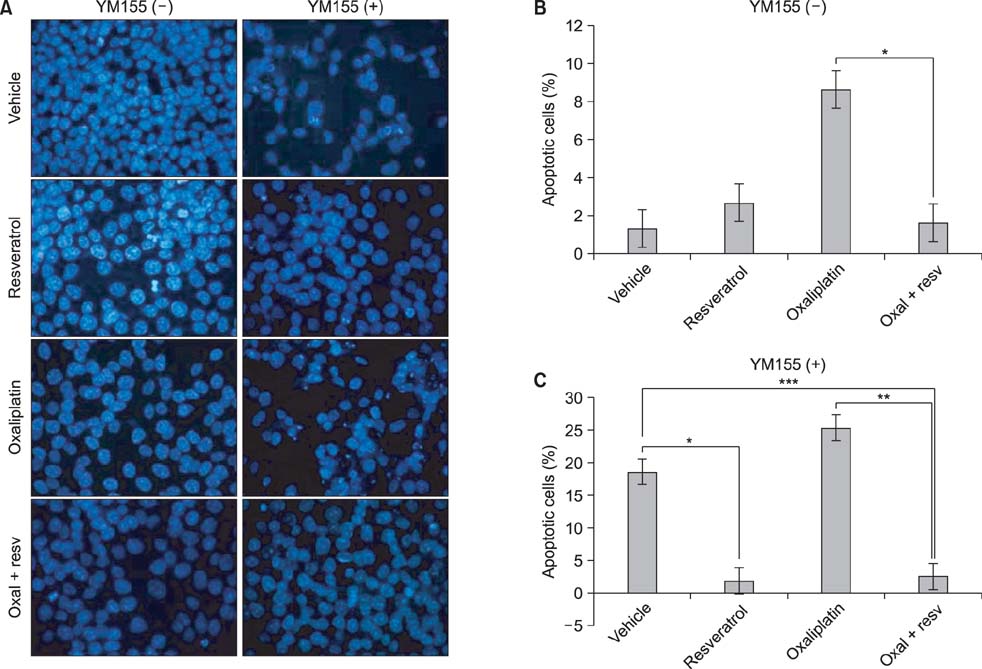
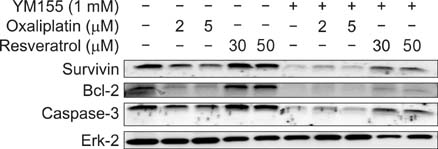
Although oxaliplatin significantly suppressed survivin transcripts and protein expression level in HCT116 cells, resveratrol cotreatment induced restoration of survivin expression level of both transcripts and protein. Apoptotic induction by oxaliplatin only treatment was nullified upon resveratrol cotreatment. Induction of survivin restoration upon resveratrol cotreatment also occurred when survivin specific inhibitor, YM155, was used. In addition to survivin restoration, resveratrol cotreatment also induced restoration of Bcl-2/caspase-3 expression suppressed by oxaliplatin only treatment.
CONCLUSION
Resveratrol has an antichemosensitizing effect upon cotreatment with oxaliplatin in HCT colon cancer cells. This antichemosensitizing effect of resveratrol can be cell-type specific. However, clinical use of resveratrol cotreatment with oxaliplatin should be approached cautiously.
MeSH Terms
Figure
Reference
-
1. Jung KW, Won YJ, Kong HJ, Oh CM, Seo HG, Lee JS. Cancer statistics in Korea: incidence, mortality, survival and prevalence in 2010. Cancer Res Treat. 2013; 45:1–14.2. Raymond E, Faivre S, Chaney S, Woynarowski J, Cvitkovic E. Cellular and molecular pharmacology of oxaliplatin. Mol Cancer Ther. 2002; 1:227–235.3. Fujie Y, Yamamoto H, Ngan CY, Takagi A, Hayashi T, Suzuki R, et al. Oxaliplatin, a potent inhibitor of survivin, enhances paclitaxel-induced apoptosis and mitotic catastrophe in colon cancer cells. Jpn J Clin Oncol. 2005; 35:453–463.4. Sohn WJ, Lee JW, Park DG. Change in expression of survivin caused by using oxaliplatin in HCT116 colon cancer cells. J Korean Soc Coloproctol. 2010; 26:246–253.5. Higgins CF. Multiple molecular mechanisms for multidrug resistance transporters. Nature. 2007; 446:749–757.6. Gupta SC, Kim JH, Prasad S, Aggarwal BB. Regulation of survival, proliferation, invasion, angiogenesis, and metastasis of tumor cells through modulation of inflammatory pathways by nutraceuticals. Cancer Metastasis Rev. 2010; 29:405–434.7. Fulda S, Debatin KM. Sensitization for anticancer drug-induced apoptosis by the chemopreventive agent resveratrol. Oncogene. 2004; 23:6702–6711.8. Zhao W, Bao P, Qi H, You H. Resveratrol down-regulates survivin and induces apoptosis in human multidrug-resistant SPC-A-1/CDDP cells. Oncol Rep. 2010; 23:279–286.9. Aggarwal BB, Bhardwaj A, Aggarwal RS, Seeram NP, Shishodia S, Takada Y. Role of resveratrol in prevention and therapy of cancer: preclinical and clinical studies. Anticancer Res. 2004; 24(5A):2783–2840.10. Jazirehi AR, Bonavida B. Resveratrol modifies the expression of apoptotic regulatory proteins and sensitizes non-Hodgkin's lymphoma and multiple myeloma cell lines to paclitaxel-induced apoptosis. Mol Cancer Ther. 2004; 3:71–84.11. Rezk YA, Balulad SS, Keller RS, Bennett JA. Use of resveratrol to improve the effectiveness of cisplatin and doxorubicin: study in human gynecologic cancer cell lines and in rodent heart. Am J Obstet Gynecol. 2006; 194:e23–e26.12. Harikumar KB, Aggarwal BB. Resveratrol: a multitargeted agent for age-associated chronic diseases. Cell Cycle. 2008; 7:1020–1035.13. Longley DB, Wilson TR, McEwan M, Allen WL, McDermott U, Galligan L, et al. c-FLIP inhibits chemotherapy-induced colorectal cancer cell death. Oncogene. 2006; 25:838–848.14. Sasaki H, Sheng Y, Kotsuji F, Tsang BK. Down-regulation of X-linked inhibitor of apoptosis protein induces apoptosis in chemoresistant human ovarian cancer cells. Cancer Res. 2000; 60:5659–5666.15. Altieri DC. Validating survivin as a cancer therapeutic target. Nat Rev Cancer. 2003; 3:46–54.16. Sah NK, Khan Z, Khan GJ, Bisen PS. Structural, functional and therapeutic biology of survivin. Cancer Lett. 2006; 244:164–171.17. Mita AC, Mita MM, Nawrocki ST, Giles FJ. Survivin: key regulator of mitosis and apoptosis and novel target for cancer therapeutics. Clin Cancer Res. 2008; 14:5000–5005.18. Yamamoto H, Ngan CY, Monden M. Cancer cells survive with survivin. Cancer Sci. 2008; 99:1709–1714.19. Baur JA, Sinclair DA. Therapeutic potential of resveratrol: the in vivo evidence. Nat Rev Drug Discov. 2006; 5:493–506.20. Jang M, Cai L, Udeani GO, Slowing KV, Thomas CF, Beecher CW, et al. Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science. 1997; 275:218–220.21. Shakibaei M, Harikumar KB, Aggarwal BB. Resveratrol addiction: to die or not to die. Mol Nutr Food Res. 2009; 53:115–128.22. Harikumar KB, Kunnumakkara AB, Sethi G, Diagaradjane P, Anand P, Pandey MK, et al. Resveratrol, a multitargeted agent, can enhance antitumor activity of gemcitabine in vitro and in orthotopic mouse model of human pancreatic cancer. Int J Cancer. 2010; 127:257–268.23. Csaki C, Keshishzadeh N, Fischer K, Shakibaei M. Regulation of inflammation signalling by resveratrol in human chondrocytes in vitro. Biochem Pharmacol. 2008; 75:677–687.24. Velculescu VE, Madden SL, Zhang L, Lash AE, Yu J, Rago C, et al. Analysis of human transcriptomes. Nat Genet. 1999; 23:387–388.25. Kim PJ, Plescia J, Clevers H, Fearon ER, Altieri DC. Survivin and molecular pathogenesis of colorectal cancer. Lancet. 2003; 362:205–209.26. Altieri DC. Survivin, cancer networks and pathway-directed drug discovery. Nat Rev Cancer. 2008; 8:61–70.27. Altieri DC. Coupling apoptosis resistance to the cellular stress response: the IAP-Hsp90 connection in cancer. Cell Cycle. 2004; 3:255–256.28. Fortugno P, Beltrami E, Plescia J, Fontana J, Pradhan D, Marchisio PC, et al. Regulation of survivin function by Hsp90. Proc Natl Acad Sci U S A. 2003; 100:13791–13796.29. Rödel F, Frey B, Leitmann W, Capalbo G, Weiss C, Rodel C. Survivin antisense oligonucleotides effectively radiosensitize colorectal cancer cells in both tissue culture and murine xenograft models. Int J Radiat Oncol Biol Phys. 2008; 71:247–255.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Change in Expression of Survivin Caused by Using Oxaliplatin in HCT116 Colon Cancer Cells
- YM155, specific survivin inhibitor, can enhance artesunate-induced cytotoxicity in HCT116 colon cancer cells
- The Changes of Expression of Survivin by Butyrate in HCT116 Colon Cancer Cells
- The Nedd8-activating enzyme inhibitor MLN4924 suppresses colon cancer cell growth via triggering autophagy
- Cucurbitacin E’s Anti-Cancer Effects on HCT116 Human Colon Cancer Cells by Controlling Expression and Phosphorylation Levels of Caspase-9, eIF-2α, and ATF-4