J Clin Neurol.
2005 Oct;1(2):107-120. 10.3988/jcn.2005.1.2.107.
Neuroprotective Therapy in Parkinson's Disease: Current Status and New Directions from Experimental and Genetic Clues
- Affiliations
-
- 1Committees on Neurobiology and Molecular Medicine, Departments of Neurology and Neurobiology, Pharmacology, & Physiology, The University of Chicago, USA. unkang@uchicago.edu
- KMID: 1808481
- DOI: http://doi.org/10.3988/jcn.2005.1.2.107
Abstract
- Despite successful treatment of Parkinson's disease (PD) with a wide variety of symptomatic therapy, the disease continues to progress and drug-resistance symptoms become the predominant factors producing the disability of PD patients. Neuroprotective therapies have been tested, but clinically effective drugs have not been found yet. New insights gained from studies of genetic forms of PD point to the common pathogenic mechanisms that have been suspected in sporadic forms of the disease and may provide new approaches for the future neuroprotective therapies.
Keyword
MeSH Terms
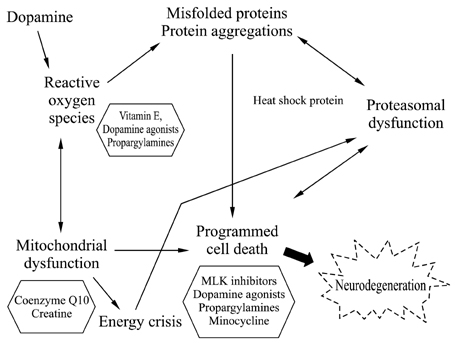
Figure
Reference
-
1. Tanner CM, Goldman SM. Epidemiology of Parkinson's disease. Neurol Clin. 1996. 14:317–335.
Article2. Fearnley JM, Lees AJ. Ageing and Parkinson's disease: substantia nigra regional selectivity. Brain. 1991. 114(Pt 5):2283–2301.
Article3. Gasser T. Genetics of Parkinson's disease. Curr Opin Neurol. 2005. 18(4):363–369.
Article4. Tanner CM, Aston DA. Epidemiology of Parkinson's disease and akinetic syndromes. Curr Opin Neurol. 2000. 13(4):427–430.
Article5. Javitch JA, D'Amato RJ, Strittmatter SM, Snyder SH. Parkinsonism-induced neurotoxin, N-methyl-4-phenyl-1,2,3,6-tetrahydropyridine: uptake of the metabolite N-methyl-4-phenylpyridine by dopamine neurons explains selective toxicity. Proc Natl Acad Sci USA. 1985. 82:2173–2177.
Article6. Bezard E, Gross CE, Fournier MC, Dovero S, Bloch B, Jaber M. Absence of MPTP-induced neuronal death in mice lacking the dopamine transporter. Exp Neurol. 1999. 155(2):268–273.
Article7. Liu Y, Roghani A, Edwards RH. Gene transfer of a reserpine-sensitive mechanism of resistance to N-methyl-4-phenylpyridinium. Proc Natl Acad Sci U S A. 1992. 89(19):9074–9078.
Article8. Ramsay RR, Singer TP. Energy-dependent uptake of N-methyl-4-phenylpyridinium, the neurotoxic metabolite of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine, by mitochondria. J Biol Chem. 1986. 261(17):7585–7587.
Article9. Klaidman LK, Adams JD Jr, Leung AC, Kim SS, Cadenas E. Redox cycling of MPP+: evidence for a new mechanism involving hydride transfer with xanthine oxidase, aldehyde dehydrogenase, and lipoamide dehydrogenase. Free Radic Biol Med. 1993. 15(2):169–179.
Article10. Miller GW, Gainetdinov RR, Levey AI, Caron MG. Dopamine transporters and neuronal injury. Trends Pharmacol Sci. 1999. 20(10):424–429.
Article11. Nicklas WJ, Youngster SK, Kindt MV, Heikkila RE. MPTP, MPP+ and mitochondrial function. Life Sci. 1987. 40(8):721–729.12. Nakamura K, Bindokas VP, Marks JD, Wright DA, Frim DM, Miller RJ, et al. The selective toxicity of 1-methyl-4-phenylpyridinium to dopaminergic neurons: the role of mitochondrial complex I and reactive oxygen species revisited. Mol Pharmacol. 2000. 58(2):271–278.
Article13. Forno LS, DeLanney LE, Irwin I, Langston JW. Similarities and differences between MPTP-induced Parkinsonsim and Parkinson's disease. Neuropathologic considerations. Adv Neurol. 1993. 60:600–608.14. Vila M, Wu DC, Przedborski S. Engineered modeling and the secrets of Parkinson's disease. Trends Neurosci. 2001. 24:11 Suppl. S49–S55.
Article15. Przedborski S, Vila M. The 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model: a tool to explore the pathogenesis of Parkinson's disease. Ann N Y Acad Sci. 2003. 991:189–198.16. Manning-Bog AB, McCormack AL, Li J, Uversky VN, Fink AL, Di Monte DA. The herbicide paraquat causes up-regulation and aggregation of alpha-synuclein in mice: paraquat and alpha-synuclein. J Biol Chem. 2002. 277(3):1641–1644.
Article17. McCormack AL, Thiruchelvam M, Manning-Bog AB, Thiffault C, Langston JW, Cory-Slechta DA, et al. Environmental Risk Factors and Parkinson's Disease: Selective Degeneration of Nigral Dopaminergic Neurons Caused by the Herbicide Paraquat. Neurobiol Dis. 2002. 10(2):119–127.
Article18. Shimizu K, Ohtaki K, Matsubara K, Aoyama K, Uezono T, Saito O, et al. Carrier-mediated processes in blood-brain barrier penetration and neural uptake of paraquat. Brain Res. 2001. 906(1-2):135–142.
Article19. Barrientos A, Moraes CT. Titrating the effects of mitochondrial complex I impairment in the cell physiology. J Biol Chem. 1999. 274(23):16188–16197.
Article20. Betarbet R, Sherer TB, MacKenzie G, Garcia-Osuna M, Panov AV, Greenamyre JT. Chronic systemic pesticide exposure reproduces features of Parkinson's disease. Nat Neurosci. 2000. 3(12):1301–1306.
Article21. Hoglinger GU, Feger J, Prigent A, Michel PP, Parain K, Champy P, et al. Chronic systemic complex I inhibition induces a hypokinetic multisystem degeneration in rats. J Neurochem. 2003. 84(3):491–502.
Article22. Ferrante RJ, Schulz JB, Kowall NW, Beal MF. Systemic administration of rotenone produces selective damage in the striatum and globus pallidus, but not in the substantia nigra. Brain Res. 1997. 753:157–162.
Article23. Ross GW, Abbott RD, Petrovitch H, Morens DM, Grandinetti A, Tung KH, et al. Association of coffee and caffeine intake with the risk of Parkinson disease. Jama. 2000. 283(20):2674–2679.
Article24. Tanner CM, Goldman SM, Aston DA, Ottman R, Ellenberg J, Mayeux R, et al. Smoking and Parkinson's disease in twins. Neurology. 2002. 58(4):581–588.
Article25. Chen H, Zhang SM, Hernan MA, Schwarzschild MA, Willett WC, Colditz GA, et al. Nonsteroidal anti-inflammatory drugs and the risk of Parkinson disease. Arch Neurol. 2003. 60(8):1059–1064.
Article26. Etminan M, Gill SS, Samii A. Intake of vitamin E, vitamin C, and carotenoids and the risk of Parkinson's disease: a meta-analysis. Lancet Neurology. 2005. 4(6):362–365.
Article27. Chen H, Zhang SM, Schwarzschild MA, Hernan MA, Ascherio A. Physical activity and the risk of Parkinson disease. Neurology. 2005. 64(4):664–669.
Article28. Polymeropoulos MH, Lavedan C, Leroy E, Ide SE, Dehejia A, Dutra A, et al. Mutation in the alpha-synuclein gene identified in families with Parkinson's disease. Science. 1997. 276(5321):2045–2047.
Article29. Kruger R, Kuhn W, Muller T, Woitalla D, Graeber M, Kosel S, et al. Ala30Pro mutation in the gene encoding alpha-synuclein in Parkinson's disease. Nat Genet. 1998. 18(2):106–108.30. Zarranz JJ, Alegre J, Gomez-Esteban JC, Lezcano E, Ros R, Ampuero I, et al. The new mutation, E46K, of alpha-synuclein causes Parkinson and Lewy body dementia. Ann Neurol. 2004. 55(2):164–173.
Article31. Singleton AB, Farrer M, Johnson J, Singleton A, Hague S, Kachergus J, et al. alpha-Synuclein locus triplication causes Parkinson's disease. Science. 2003. 302(5646):841.
Article32. Stefanis L, Larsen KE, Rideout HJ, Sulzer D, Greene LA. Expression of A53T mutant but not wild-type alpha-synuclein in PC12 cells induces alterations of the ubiquitin-dependent degradation system, loss of dopamine release, and autophagic cell death. J Neurosci. 2001. 21(24):9549–9560.
Article33. Tanaka Y, Engelender S, Igarashi S, Rao RK, Wanner T, Tanzi RE, et al. Inducible expression of mutant alpha-synuclein decreases proteasome activity and increases sensitivity to mitochondria-dependent apoptosis. Hum Mol Genet. 2001. 10(9):919–926.
Article34. Petrucelli L, O'Farrell C, Lockhart PJ, Baptista M, Kehoe K, Vink L, et al. Parkin protects against the toxicity associated with mutant alpha-synuclein: proteasome dysfunction selectively affects catecholaminergic neurons. Neuron. 2002. 36(6):1007–1019.
Article35. Masliah E, Rockenstein E, Veinbergs I, Mallory M, Hashimoto M, Takeda A, et al. Dopaminergic loss and inclusion body formation in alpha-synuclein mice: implications for neurodegenerative disorders. Science. 2000. 287(5456):1265–1269.
Article36. Lee MK, Stirling W, Xu Y, Xu X, Qui D, Mandir AS, et al. Human alpha-synuclein-harboring familial Parkinson's disease-linked Ala-53 --> Thr mutation causes neurodegenerative disease with alpha-synuclein aggregation in transgenic mice. Proc Natl Acad Sci U S A. 2002. 99(13):8968–8973.
Article37. Conway KA, Lee SJ, Rochet JC, Ding TT, Williamson RE, Lansbury PT Jr. Acceleration of oligomerization, not fibrillization, is a shared property of both alpha-synuclein mutations linked to early-onset Parkinson's disease: implications for pathogenesis and therapy. Proc Natl Acad Sci U S A. 2000. 97(2):571–576.
Article38. Xu J, Kao SY, Lee FJ, Song W, Jin LW, Yankner BA. Dopamine-dependent neurotoxicity of alpha-synuclein: a mechanism for selective neurodegeneration in Parkinson disease. Nat Med. 2002. 8(6):600–606.
Article39. Conway KA, Rochet JC, Bieganski RM, Lansbury PT Jr. Kinetic stabilization of the alpha-synuclein protofibril by a dopamine-alpha-synuclein adduct. Science. 2001. 294(5545):1346–1349.
Article40. Matsumine H, Saito M, Shimoda-Matsubayashi S, Tanaka H, Ishikawa A, Nakagawa-Hattori Y, et al. Localization of a gene for an autosomal recessive form of juvenile Parkinsonism to chromosome 6q25.2-27. Am J Hum Genet. 1997. 60(3):588–596.41. Kitada T, Asakawa S, Hattori N, Matsumine H, Yamamura Y, Minoshima S, et al. Mutations in the parkin gene cause autosomal recessive juvenile parkinsonism. Nature. 1998. 392:605–608.
Article42. Shimura H, Hattori N, Kubo S, Mizuno Y, Asakawa S, Minoshima S, et al. Familial Parkinson disease gene product, parkin, is a ubiquitin-protein ligase. Nat Genet. 2000. 25(3):302–305.
Article43. Zhang Y, Gao J, Chung KK, Huang H, Dawson VL, Dawson TM. Parkin functions as an E2-dependent ubiquitin-protein ligase and promotes the degradation of the synaptic vesicle-associated protein, CDCrel-1. Proc Natl Acad Sci U S A. 2000. 97(24):13354–13359.
Article44. Goldberg MS, Fleming SM, Palacino JJ, Cepeda C, Lam HA, Bhatnagar A, et al. Parkin-deficient mice exhibit nigrostriatal deficits but not loss of dopaminergic neurons. J Biol Chem. 2003. 278(44):43628–43635.
Article45. Palacino JJ, Sagi D, Goldberg MS, Krauss S, Motz C, Wacker M, et al. Mitochondrial dysfunction and oxidative damage in parkin-deficient mice. J Biol Chem. 2004. 279(18):18614–18622.
Article46. Leroy E, Boyer R, Auburger G, Leube B, Ulm G, Mezey E, et al. The ubiquitin pathway in Parkinson's disease. Nature. 1998. 395(6701):451–452.
Article47. Lincoln S, Vaughan J, Wood N, Baker M, Adamson J, Gwinn-Hardy K, et al. Low frequency of pathogenic mutations in the ubiquitin carboxy-terminal hydrolase gene in familial Parkinson's disease. Neuroreport. 1999. 10(2):427–429.
Article48. Maraganore DM, Lesnick TG, Elbaz A, Chartier-Harlin MC, Gasser T, Kruger R, et al. UCHL1 is a Parkinson's disease susceptibility gene. Ann Neurol. 2004. 55(4):512–521.
Article49. Larsen CN, Price JS, Wilkinson KD. Substrate binding and catalysis by ubiquitin C-terminal hydrolases: identification of two active site residues. Biochemistry. 1996. 35(21):6735–6744.
Article50. Larsen CN, Krantz BA, Wilkinson KD. Substrate specificity of deubiquitinating enzymes: ubiquitin C-terminal hydrolases. Biochemistry. 1998. 37(10):3358–3368.
Article51. Saigoh K, Wang YL, Suh JG, Yamanishi T, Sakai Y, Kiyosawa H, et al. Intragenic deletion in the gene encoding ubiquitin carboxy-terminal hydrolase in gad mice. Nat Genet. 1999. 23(1):47–51.
Article52. Bonifati V, Rizzu P, van Baren MJ, Schaap O, Breedveld GJ, Krieger E, et al. Mutations in the DJ-1 gene associated with autosomal recessive early-onset parkinsonism. Science. 2003. 299(5604):256–259.
Article53. Wilson MA, Collins JL, Hod Y, Ringe D, Petsko GA. The 1.1-A resolution crystal structure of DJ-1, the protein mutated in autosomal recessive early onset Parkinson's disease. Proc Natl Acad Sci U S A. 2003. 100(16):9256–9261.
Article54. Mitsumoto A, Nakagawa Y, Takeuchi A, Okawa K, Iwamatsu A, Takanezawa Y. Oxidized forms of peroxiredoxins and DJ-1 on two-dimensional gels increased in response to sublethal levels of paraquat. Free Radic Res. 2001. 35(3):301–310.
Article55. Shendelman S, Jonason A, Martinat C, Leete T, Abeliovich A. DJ-1 is a redox-dependent molecular chaperone that inhibits alpha-synuclein aggregate formation. PLoS Biol. 2004. 2(11):e362.56. Chen L, Cagniard B, Mathews T, Jones S, Koh HC, Ding Y, et al. Age-dependent motor deficits and dopaminergic dysfunction in DJ-1 null mice. J Biol Chem. 2005. 280(22):21418–21426.
Article57. Goldberg MS, Pisani A, Haburcak M, Vortherms TA, Kitada T, Costa C, et al. Nigrostriatal Dopaminergic Deficits and Hypokinesia Caused by Inactivation of the Familial Parkinsonism-Linked Gene DJ-1. Neuron. 2005. 45(4):489–496.
Article58. Valente EM, Abou-Sleiman PM, Caputo V, Muqit MM, Harvey K, Gispert S, et al. Hereditary early-onset Parkinson's disease caused by mutations in PINK1. Science. 2004. 304(5674):1158–1160.
Article59. Beilina A, Van Der Brug M, Ahmad R, Kesavapany S, Miller DW, Petsko GA, et al. Mutations in PTEN-induced putative kinase 1 associated with recessive parkinsonism have differential effects on protein stability. Proc Natl Acad Sci U S A. 2005. 102(16):5703–5708.
Article60. Zimprich A, Biskup S, Leitner P, Lichtner P, Farrer M, Lincoln S, et al. Mutations in LRRK2 cause autosomal-dominant parkinsonism with pleomorphic pathology. Neuron. 2004. 44(4):601–607.
Article61. Karamohamed S, DeStefano AL, Wilk JB, Shoemaker CM, Golbe LI, Mark MH, et al. A haplotype at the PARK3 locus influences onset age for Parkinson's disease: the GenePD study. Neurology. 2003. 61(11):1557–1561.
Article62. Zhang J, Song Y, Chen H, Fan D. The tau gene haplotype h1 confers a susceptibility to Parkinson's disease. Eur Neurol. 2005. 53(1):15–21.
Article63. Oliveira SA, Li YJ, Noureddine MA, Zuchner S, Qin X, Pericak-Vance MA, et al. Identification of Risk and Age-at-Onset Genes on Chromosome 1p in Parkinson Disease. Am J Hum Genet. 2005. 77(2):252–264.
Article64. Sun XM, Butterworth M, MacFarlane M, Dubiel W, Ciechanover A, Cohen GM. Caspase activation inhibits proteasome function during apoptosis. Mol Cell. 2004. 14(1):81–93.
Article65. Ellgaard L, Helenius A. Quality control in the endoplasmic reticulum. Nat Rev Mol Cell Biol. 2003. 4(3):181–191.
Article66. Sullivan PG, Dragicevic NB, Deng JH, Bai Y, Dimayuga E, Ding Q, et al. Proteasome inhibition alters neural mitochondrial homeostasis and mitochondria turnover. J Biol Chem. 2004. 279(20):20699–20707.
Article67. LaVoie MJ, Hastings TG. Peroxynitrite- and nitrite-induced oxidation of dopamine: implications for nitric oxide in dopaminergic cell loss. J Neurochem. 1999. 73(6):2546–2554.
Article68. Junn E, Mouradian MM. Apoptotic signaling in dopamine-induced cell death: the role of oxidative stress, p38 mitogen-activated protein kinase, cytochrome c and caspases. J Neurochem. 2001. 78(2):374–383.
Article69. Luo Y, Umegaki H, Wang X, Abe R, Roth GS. Dopamine induces apoptosis through an oxidation-involved SAPK/JNK activation pathway. J Biol Chem. 1998. 273(6):3756–3764.
Article70. Nakamura K, Wright DA, Wiatr T, Kowlessur D, Milstien S, Lei XG, et al. Preferential resistance of dopaminergic neurons to the toxicity of glutathione depletion is independent of cellular glutathione peroxidase and Is mediated by tetrahydrobiopterin. J Neurochem. 2000. 74(6):2305–2315.
Article71. Nakamura K, Bindokas VP, Kowlessur D, Elas M, Milstien S, Marks JD, et al. Tetrahydrobiopterin scavenges superoxide in dopaminergic neurons. J Biol Chem. 2001. 276:34402–34407.
Article72. Jellinger KA. Post mortem studies in Parkinson's disease--is it possible to detect brain areas for specific symptoms? J Neural Transm Suppl. 1999. 56:1–29.73. Greene JG, Dingledine R, Greenamyre JT. Gene expression profiling of rat midbrain dopamine neurons: implications for selective vulnerability in parkinsonism. Neurobiol Dis. 2005. 18(1):19–31.
Article74. Kweon GR, Marks JD, Krencik R, Leung EH, Schumacker PT, Hyland K, et al. Distinct mechanisms of neurodegeneration induced by chronic complex I inhibition in dopaminergic and non-dopaminergic cells. J Biol Chem. 2004. 279(50):51783–51792.
Article75. Grimm J, Mueller A, Hefti F, Rosenthal A. Molecular basis for catecholaminergic neuron diversity. Proc Natl Acad Sci U S A. 2004. 101(38):13891–13896.
Article76. Grunblatt E, Mandel S, Jacob-Hirsch J, Zeligson S, Amariglo N, Rechavi G, et al. Gene expression profiling of parkinsonian substantia nigra pars compacta; alterations in ubiquitin-proteasome, heat shock protein, iron and oxidative stress regulated proteins, cell adhesion/cellular matrix and vesicle trafficking genes. J Neural Transm. 2004. 111(12):1543–1573.
Article77. Sechi G, Deledda MG, Bua G, Satta WM, Deiana GA, Pes GM, et al. Reduced intravenous glutathione in the treatment of early Parkinson's disease. Prog Neuropsychopharmacol Biol Psychiatry. 1996. 20(7):1159–1170.
Article78. Parkinson Study G. DATATOP: a multicenter controlled clinical trial in early Parkinson's disease. Parkinson Study Group. Arch Neurol. 1989. 46(10):1052–1060.79. Parkinson Study G. Effect of deprenyl on the progression of disability in early Parkinson's disease. N Engl J Med. 1989. 321(20):1364–1371.80. Parkinson Study G. Effects of tocopherol and deprenyl on the progression of disability in early Parkinson's disease. N Engl J Med. 1993. 328:176–183.81. Schapira AH, Cooper JM, Dexter D, Jenner P, Clark JB, Marsden CD. Mitochondrial complex I deficiency in Parkinson's disease. Lancet. 1989. 1(8649):1269.
Article82. Mann VM, Cooper JM, Krige D, Daniel SE, Schapira AHV, Marsden CD. Brain, skeletal muscle and platelet homogenate mitochondrial function in Parkinson's disease. Brain. 1992. 115:333–342.
Article83. Beal MF, Matthews RT, Tieleman A, Shults CW. Coenzyme Q10 attenuates the 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) induced loss of striatal dopamine and dopaminergic axons in aged. Brain Res. 1998. 783:109–114.
Article84. Matthews RT, Ferrante RJ, Klivenyi P, Yang L, Klein AM, Mueller G, et al. Creatine and cyclocreatine attenuate MPTP neurotoxicity. Exp Neurol. 1999. 157(1):142–149.
Article85. Shults CW, Haas RH, Passov D, Beal MF. Coenzyme Q10 levels correlate with the activities of complexes I and II/III in mitochondria from parkinsonian and nonparkinsonian subjects. Ann Neurol. 1997. 42(2):261–264.
Article86. Shults CW, Oakes D, Kieburtz K, Beal MF, Haas R, Plumb S, et al. Effects of coenzyme Q10 in early Parkinson disease: evidence of slowing of the functional decline. Arch Neurol. 2002. 59(10):1541–1550.87. Tabrizi SJ, Blamire AM, Manners DN, Rajagopalan B, Styles P, Schapira AH, et al. High-dose creatine therapy for Huntington disease: a 2-year clinical and MRS study. Neurology. 2005. 64(9):1655–1656.
Article88. Elm JJ, Goetz CG, Ravina B, Shannon K, Wooten GF, Tanner CM, et al. A responsive outcome for Parkinson's disease neuroprotection futility studies. Ann Neurol. 2005. 57(2):197–203.
Article89. Silva RM, Kuan CY, Rakic P, Burke RE. Mixed lineage kinase-c-jun N-terminal kinase signaling pathway: a new therapeutic target in Parkinson's disease. Mov Disord. 2005. 20(6):653–664.
Article90. Saporito MS, Brown EM, Miller MS, Carswell S. CEP-1347/KT-7515, an inhibitor of c-jun N-terminal kinase activation, attenuates the 1-methyl-4-phenyl tetrahydropyridine-mediated loss of nigrostriatal dopaminergic neurons In vivo. J Pharmacol Exp Ther. 1999. 288(2):421–427.91. Teismann P, Tieu K, Choi DK, Wu DC, Naini A, Hunot S, et al. Cyclooxygenase-2 is instrumental in Parkinson's disease neurodegeneration. Proc Natl Acad Sci U S A. 2003. 100(9):5473–5478.
Article92. Schwid SR. Parkinson Study Group. CEP-1347 in Parkinson's disease: a pilot study. Mov Disord. 2002. 17:S91.94. Ravina BM, Fagan SC, Hart RG, Hovinga CA, Murphy DD, Dawson TM, et al. Neuroprotective agents for clinical trials in Parkinson's disease: a systematic assessment. Neurology. 2003. 60(8):1234–1240.
Article95. Parkinson Study G. Impact of deprenyl and tocopherol treatment on Parkinson's disease in DATATOP subjects not requiring levodopa. Parkinson Study Group. Ann Neurol. 1996. 39(1):29–36.96. Olanow CW, Calne D. Does selegiline monotherapy in Parkinson's disease act by symptomatic or protective mechanisms? Neurology. 1992. 42:4 Suppl 4. 13–26.97. Parkinson Study G. A controlled, randomized, delayed-start study of rasagiline in early Parkinson disease. Arch Neurol. 2004. 61(4):561–566.98. Youdim MB, Bar Am O, Yogev-Falach M, Weinreb O, Maruyama W, Naoi M, et al. Rasagiline: neurodegeneration, neuroprotection, and mitochondrial permeability transition. J Neurosci Res. 2005. 79(1-2):172–179.
Article99. Schapira AH, Olanow CW. Rationale for the use of dopamine agonists as neuroprotective agents in Parkinson's disease. Ann Neurol. 2003. 53:Suppl 3. S149–S157. discussion S157-9.
Article100. Halbig TD, Tse W, Olanow CW. Neuroprotective agents in Parkinson's disease: clinical evidence and caveats. Neurol Clin. 2004. 22:3 Suppl. S1–S17.101. Parkinson Study G. Dopamine transporter brain imaging to assess the effects of pramipexole vs levodopa on Parkinson disease progression. Jama. 2002. 287(13):1653–1661.102. Whone AL, Watts RL, Stoessl AJ, Davis M, Reske S, Nahmias C, et al. Slower progression of Parkinson's disease with ropinirole versus levodopa: The REAL-PET study. Ann Neurol. 2003. 54(1):93–101.
Article103. Fahn S, Oakes D, Shoulson I, Kieburtz K, Rudolph A, Lang A, et al. Levodopa and the progression of Parkinson's disease. N Engl J Med. 2004. 351(24):2498–2508.
Article104. Guttman M, Stewart D, Hussey D, Wilson A, Houle S, Kish S. Influence of L-dopa and pramipexole on striatal dopamine transporter in early PD. Neurology. 2001. 56(11):1559–1564.
Article105. Shoulson I, Penney J, McDermott M, Schwid S, Kayson E, Chase T, et al. A randomized, controlled trial of remacemide for motor fluctuations in Parkinson's disease. Neurology. 2001. 56(4):455–462.
Article106. Rascol O, Olanow CW, Brooks D, et al. A 2-year multicenter placebo-controlled, double blind parallel group study of the effect of riluzole in Parkinson's disease. Mov Disord. 2002. 17:39.107. Hilker R, Portman AT, Voges J, Staal MJ, Burghaus L, van Laar T, et al. Disease progression continues in patients with advanced Parkinson's disease and effective subthalamic nucleus stimulation. J Neurol Neurosurg Psychiatry. 2005. 76(9):1217–1221.
Article108. Auluck PK, Chan HY, Trojanowski JQ, Lee VM, Bonini NM. Chaperone suppression of alpha-synuclein toxicity in a Drosophila model for Parkinson's disease. Science. 2002. 295(5556):865–868.
Article109. Dong Z, Wolfer DP, Lipp HP, Bueler H. Hsp70 gene transfer by adeno-associated virus inhibits MPTP-induced nigrostriatal degeneration in the mouse model of Parkinson disease. Mol Ther. 2005. 11(1):80–88.
Article110. Auluck PK, Meulener MC, Bonini NM. Mechanisms of Suppression of {alpha}-Synuclein Neurotoxicity by Geldanamycin in Drosophila. J Biol Chem. 2005. 280(4):2873–2878.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Syndromic Approach to Parkinson's Disease: Role of Functional Imaging
- Promise of Neurorestoration and Mitochondrial Biogenesis in Parkinson's Disease with Multi Target Drugs: An Alternative to Stem Cell Therapy
- Hypothesis: Somatic Mosaicism and Parkinson Disease
- Nicotine Prevents MPTP-induced Dopaminergic Neurodegeneration
- Why Do We Need Multifunctional Neuroprotective and Neurorestorative Drugs for Parkinson's and Alzheimer's Diseases as Disease Modifying Agents