Korean J Lab Med.
2009 Dec;29(6):589-593. 10.3343/kjlm.2009.29.6.589.
Evaluation of Two Commercial HLA-B27 Real-Time PCR Kits
- Affiliations
-
- 1Greencross Reference Laboratory, Yongin, Korea. ssea74@yahoo.co.kr
- KMID: 1781615
- DOI: http://doi.org/10.3343/kjlm.2009.29.6.589
Abstract
- BACKGROUND
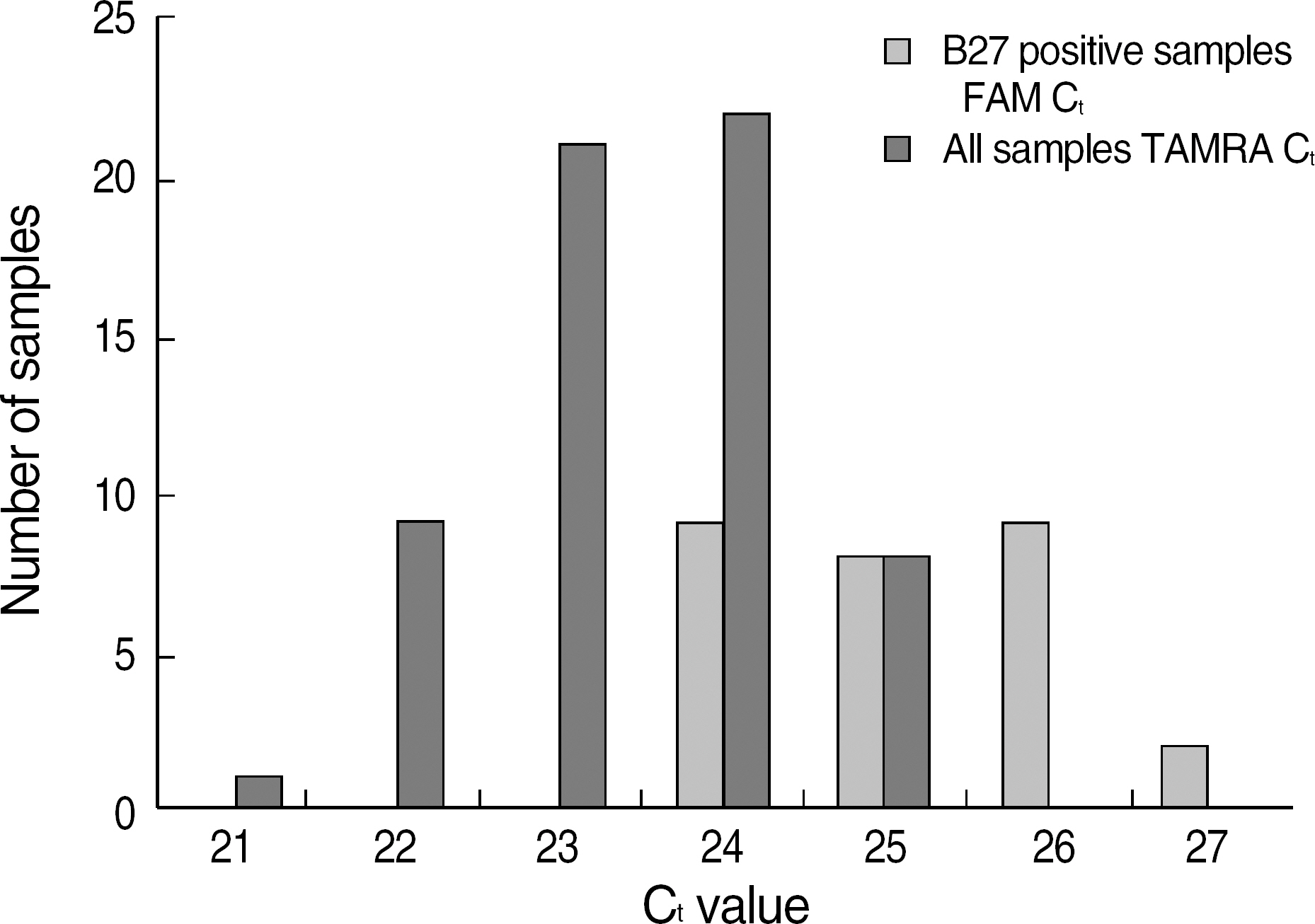
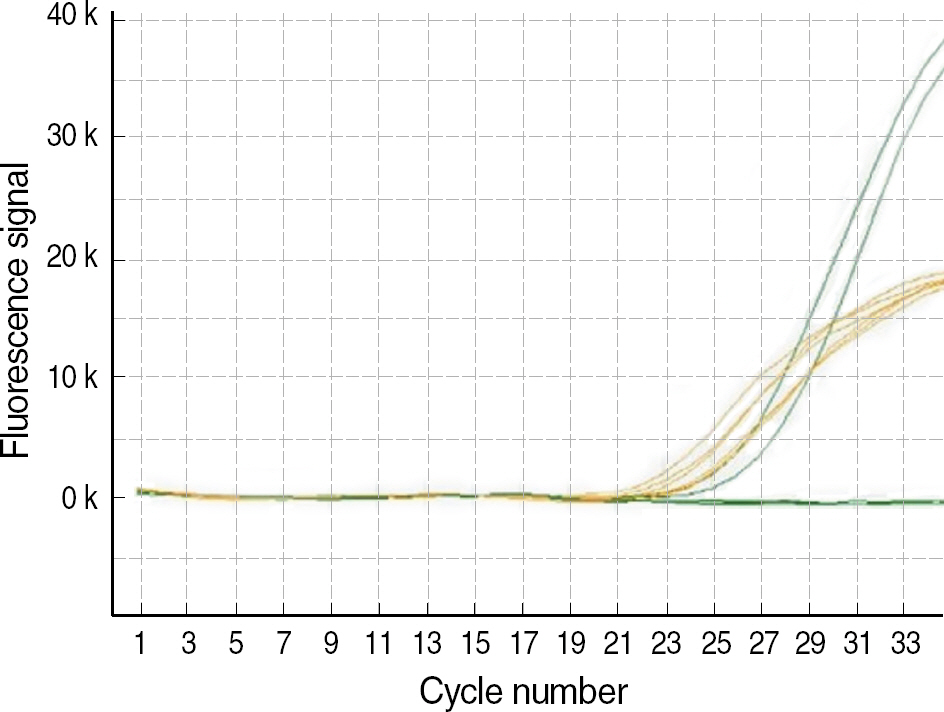
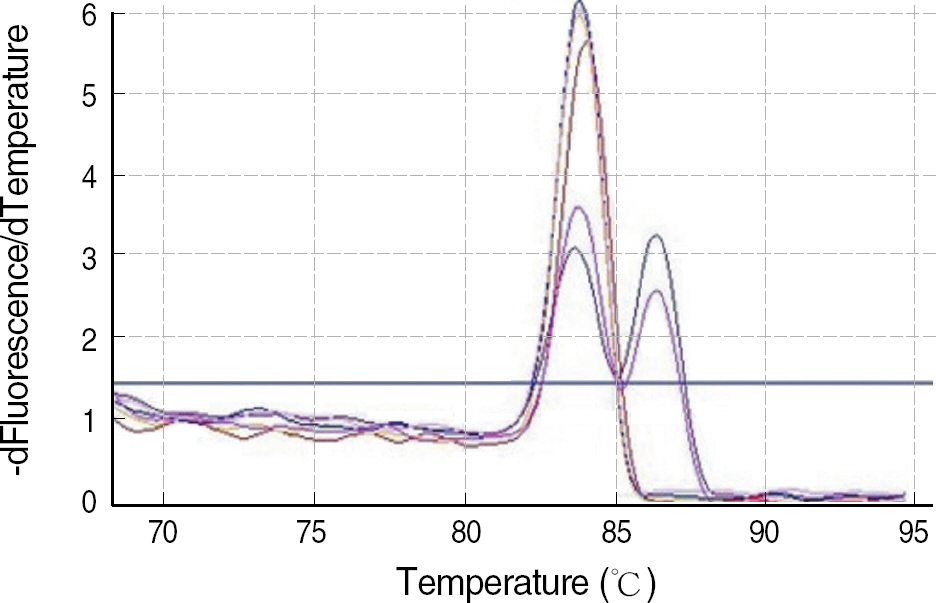
The standard PCR with sequence-specific primers (SSP) is a widely used method of HLA-B27 typing in clinical practice. The aim of our study was to evaluate 2 Korean HLA-B27 kits with different real-time PCR chemistries. METHODS: To validate the accuracy of real-time PCR kits, we selected 28 HLA-B27-positive samples and 33 HLA-B27-negative samples with a wide range of different HLA-B specificities typed by standard PCR-SSP. The 2 real-time PCR kits used were the AccuPower(R) HLA-B27 real-time PCR kit (Bioneer, Korea) with TaqMan probes and the Real-Q(TM) HLA-B*27 detection kit (BioSewoom, Korea) with SYBR Green I dye for melting curve analysis. RESULTS: All 61 samples typed by PCR-SSP demonstrated a perfect concordance with the 2 real-time PCR assays. It was possible to clearly discriminate between HLA-B27-positive and -negative samples in both real-time assays. CONCLUSIONS: In summary, both real-time PCR assays for HLA-B27 were fast, reliable, well-adapted for routine laboratory testing, and attractive alternatives to the conventional PCR-SSP method.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
Flow Cytometric Human Leukocyte Antigen-B27 Typing with Stored Samples for Batch Testing
Bo Young Seo, Dong Il Won
Ann Lab Med. 2013;33(3):174-183. doi: 10.3343/alm.2013.33.3.174.
Reference
-
1.Gonzalez S., Martinez-Borra J., Lopez-Larrea C. Immunogenetics, HLA-B27 and spondyloarthropathies. Curr Opin Rheumatol. 1999. 11:257–64.2.Whang DH., Yang YS., Hong HK. Allele and haplotype frequencies of human leukocytes antigen-A, -B, and -DR loci in Koreans: DNA typing of 1,500 cord blood units. Korean J Lab Med. 2008. 28:465–74. (황동희, 양윤선, 홍혜경. 한국인에서 Human Leukocyte Antigen-A,-B, -DR 대립유전자와일배체형빈도: 제대혈 1,500단위의DNA 형별검 사. 대한진단검사의학회지 2008;28:465-74.).3.Roh EY., Kim HS., Kim SM., Lim YM., Han BY., Park MH. HLA-A, -B, -DR allele frequencies and haplotypic associations in Koreans defined by generic-level DNA typing. Korean J Lab Med. 2003. 23:420–30. (노은연, 김현수, 김선미, 인영미, 한복연, 박명희. DNA 형별검사에 의한 한국인의 HLA-A, -B, -DR 형별및일배체형의분포. 대한진단검사의학회지 2003;23:420-30.).4.Khan MA., Mathieu A., Sorrentino R., Akkoc N. The pathogenetic role of HLA-B27 and its subtypes. Autoimmun Rev. 2007. 6:183–9.
Article5.Park KS., Kang SY., Lee WI. HLA-B27 subtypes in Korean patients with ankylosing spondylitis. Korean J Lab Med. 2008. 28:46–52. (박경선, 강소영, 이우인. 한국인 강직성 척추염 환자의 HLA-B27 아형 연구.대한진단검사의학회지 2008;28:46-52.).
Article6.Lee SH., Choi IA., Lee YA., Park EK., Kim YH., Kim KS, et al. Human leukocyte antigen-B∗2705 is the predominant subtype in the Korean population with ankylosing spondylitis, unlike in other Asians. Rheumatol Int. 2008. 29:43–6.
Article7.Dunky A., Neumuller J., Hubner C., Fischer GF., Bayer PM., Wagner E, et al. HLA-B27 determination using serological methods. A comparison of enzyme immunoassay and a microlymphocytotoxic test with flow cytometry and a molecular biological assay. Rheumatol Int. 1996. 16:95–100.
Article8.Levering WH., Wind H., Sintnicolaas K., Hooijkaas H., Gratama JW. Flow cytometric HLA-B27 screening: cross-reactivity patterns of commercially available anti-HLA-B27 monoclonal antibodies with other HLA-B antigens. Cytometry B Clin Cytom. 2003. 54:28–38.
Article9.Bon MA., van Oeveren-Dybicz A., van den Bergh FA. Genotyping of HLA-B27 by real-time PCR without hybridization probes. Clin Chem. 2000. 46:1000–2.
Article10.Faner R., Casamitjana N., Colobran R., Ribera A., Pujol-Borrell R., Palou E, et al. HLA-B27 genotyping by fluorescent resonance emission transfer (FRET) probes in real-time PCR. Hum Immunol. 2004. 65:826–38.
Article11.Sylvain K., Aurelie H., Marc M., Christophe R. Rapid screening for HLA-B27 by a TaqMan-PCR assay using sequence-specific primers and a minor groove binder probe, a novel type of TaqMan trade mark probe. J Immunol Methods. 2004. 287:179–86.12.Tiemann C., Vogel A., Dufaux B., Zimmer M., Krone JR., Hagedorn HJ. Rapid DNA typing of HLA-B27 allele by real-time PCR using lightcycler technology. Clin Lab. 2001. 47:131–4.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Utility of In-House PCR for HLA-B27 Typing: Comparison of Concordance Rate between PCR Kit and In-House PCR
- Association Between Psoriasis and HLA - B27 Antigen
- Comparison of HLA-B27 typing methods -PCR-SSP, microlymphocytotoxicity, and flow cytometry
- HLA-B27 Subtypes in Korean Patients with Ankylosing Spondylitis
- Three cases of psoriatic spondylitis showing HLA-B27 positive