J Korean Med Sci.
2007 Feb;22(1):57-62. 10.3346/jkms.2007.22.1.57.
Sodium-activated Potassium Current in Guinea pig Gastric Myocytes
- Affiliations
-
- 1Department of Physiology, Chungbuk National University, College of Medicine, Cheongju, Korea. physiokyc@chungbuk.ac.kr
- 2Department of Biochemistry, Chungbuk National University, College of Medicine, Cheongju, Korea.
- 3Department of Physiology & Biophysics, Seoul National University, College of Medicine, Seoul, Korea.
- 4Department of Physiology, Sungkyunkwan University, School of Medicine, Suwon, Korea, Korea.
- 5Department of Physiology, Nagoya City University Medical School, Nagoya, Japan.
- 6Department of Physiology, College of Medicine, Kwandong University, Gangneung, Korea.
- 7Department of Physiology, Medical School, Shanghai Jiaotong University, Shanghai, China.
- KMID: 1713230
- DOI: http://doi.org/10.3346/jkms.2007.22.1.57
Abstract
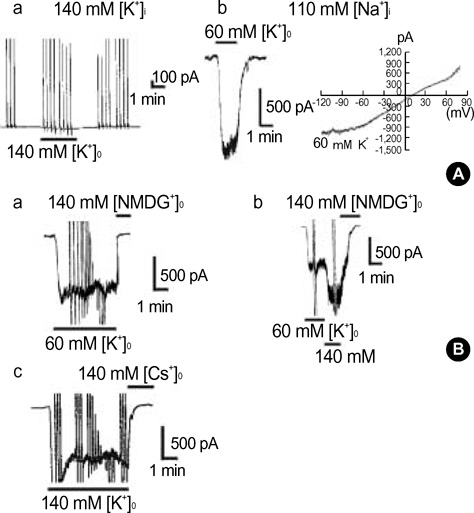
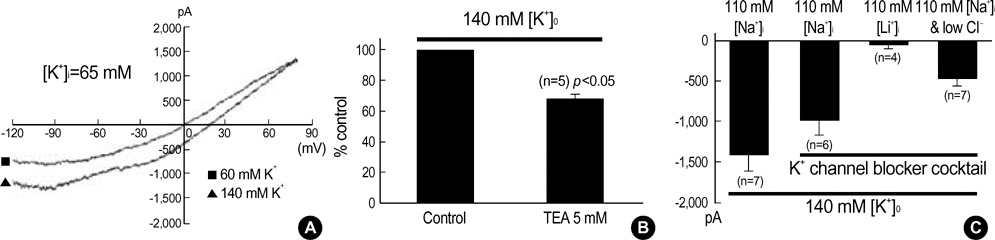
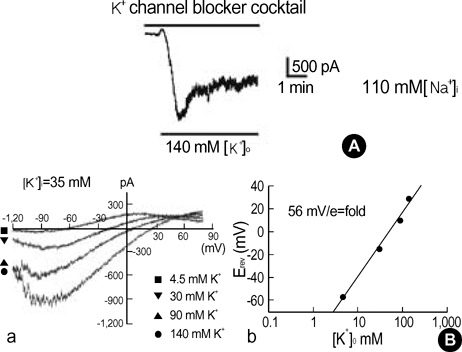
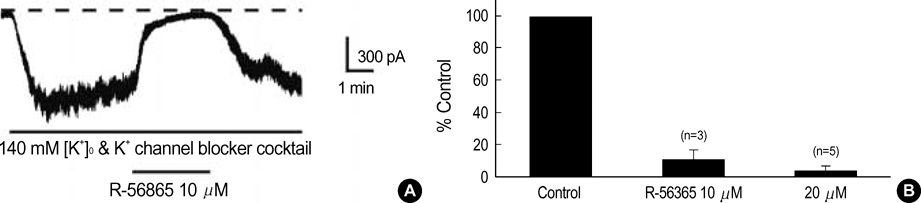
- This study was designed to identify and characterize Na+ -activated K+ current (I(K(Na))) in guinea pig gastric myocytes under whole-cell patch clamp. After whole-cell configuration was established under 110 mM intracellular Na+ concentration ([Na+]i) at holding potential of -60 mV, a large inward current was produced by external 60 mM K+([K+] degree). This inward current was not affected by removal of external Ca2+. K+ channel blockers had little effects on the current (p>0.05). Only TEA (5 mM) inhibited steady-state current to 68+/-2.7% of the control (p<0.05). In the presence of K+ channel blocker cocktail (mixture of Ba2+, glibenclamide, 4-AP, apamin, quinidine and TEA), a large inward current was activated. However, the amplitude of the steadystate current produced under [K+]degree (140 mM) was significantly smaller when Na+ in pipette solution was replaced with K+ - and Li+ in the presence of K+ channel blocker cocktail than under 110 mM [Na+]i. In the presence of K+ channel blocker cocktail under low Cl- pipette solution, this current was still activated and seemed K+ -selective, since reversal potentials (E(rev)) of various concentrations of [K+]degree-induced current in current/voltage (I/V) relationship were nearly identical to expected values. R-56865 (10-20 microgram), a blocker of IK(Na), completely and reversibly inhibited this current. The characteristics of the current coincide with those of IK(Na) of other cells. Our results indicate the presence of IK(Na) in guinea pig gastric myocytes.
Keyword
MeSH Terms
Figure
Cited by 1 articles
-
H2 Receptor-Mediated Relaxation of Circular Smooth Muscle in Human Gastric Corpus: the Role of Nitric Oxide (NO)
Sang Eok Lee, Dae Hoon Kim, Young Chul Kim, Joung-Ho Han, Woong Choi, Chan Hyung Kim, Hye Won Jeong, Seon-Mee Park, Sei Jin Yun, Song-Yi Choi, Rohyun Sung, Young Ho Kim, Ra Young Yoo, Park Hee Sun, Heon Kim, Young-Jin Song, Wen-Xie Xu, Hyo-Yung Yun, Sang Jin Lee
Korean J Physiol Pharmacol. 2014;18(5):425-430. doi: 10.4196/kjpp.2014.18.5.425.
Reference
-
1. Amberg GC, Koh SD, Imaizumi Y, Ohya S, Sanders KM. A-type potassium currents in smooth muscle. Am J Physiol Cell Physiol. 2003. 284:C583–C595.
Article2. Farrugia G. Ionic conductances in gastrointestinal smooth muscle and interstitial cells of Cajal. Annu Rev Physiol. 1999. 61:45–84.3. Garcia ML, Galvez A, Garcia-Calvo M, King VF, Vazquez J, Kaczorowski GJ. Use of toxins to study potassium channels. J Bioenerg Biomembr. 1991. 23:615–646.
Article4. Horowitz B, Ward SM, Sanders KM. Cellular and molecular basis for electrical rhythmicity in gastrointestinal tract. Annu Rev Physiol. 1999. 61:19–43.5. Latorre R, Oberhauser A, Labarca P, Alvarez O. Varieties of calcium-activated potassium channels. Annu Rev Physiol. 1989. 51:385–399.
Article6. Mitra R, Morad M. Ca2+ and Ca2+-activated K+ currents in mammalian gastric smooth muscle cells. Science. 1989. 229:269–272.7. Sim JH, Yang DK, Kim YC, Park SJ, Kang TM, So I, Kim KW. ATP-sensitive K+ channels composed of Kir6.1 and SUR2B subunits in guinea pig gastric myocytes. Am J Physiol Gastrointest Liver Physiol. 2002. 282:G137–G144.8. Blatz AL, Magleby KL. Calcium-activated potassium channels. Trends Neurosci. 1987. 10:463–467.
Article9. Koh SD, Bradley KK, Rae MG, Keef KD, Horowitz B, Sanders KM. Basal activation of ATP-sensitive potassium channels in murine colonic smooth muscle cell. Biophys J. 1998. 75:1793–1800.
Article10. Cho SY, Beckett EA, Baker SA, Han I, Park KJ, Monaghan K, Ward SM, Sanders KM, Koh SD. A pH-sensitive potassium conductance (TASK) and its function in the murine gastrointestinal tract. J Physiol. 2005. 565:243–259.11. Kameyama M, Kakei M, Sato R, Shibasaki T, Matsuda H, Irisawa H. Intracellular Na+ activates a K+ channel in mammalian cardiac cells. Nature. 1984. 309:354–356.12. Egan TM, Dagan D, Kupper J, Levitan IB. Properties and rundown of sodium-activated potassium channels in rat olfactory bulb neurons. J Neurosci. 1992. 12:1964–1976.
Article13. Dryer SE. Na+-activated K+ channels: a new family of large conductance ion channels. Trends Neurosci. 1994. 17:155–160.14. Bertrand D, Bader CR, Berheim L, Haimann C. KNa, A sodium activated potassium current. Pflügers Arch. 1989. 414:S76–S79.15. Dryer SE. Na+-activated K+ channels and voltage-evoked ionic currents in brain stem and parasympathetic neurons of the chick. J Physiol. 1989. 435:513–532.16. Hainmann C, Bernheim L, Bertrand D, Bader CR. Potassium current activated by intracellular sodium in quail trigeminal ganglion neurons. J Gen Physiol. 1990. 95:961–979.17. Malysz J, Richardson D, Farraway L, Christen M-O, Huizinga JD. Generation of slow wave type action potential in the mouse small intestine involves a non-L-type calcium channel. Can J Physiol Pharmacol. 1994. 73:1502–1511.18. Tomita T, Hata T. Effects of removal of Na+ and Cl- on spontaneous electrical activity, slow wave, in the circular muscle of the guineapig gastric antrum. Jpn J Physiol. 2000. 50:469–477.19. Benham CD, Bolton TB, Lang RJ. Acetylcholine activates an inward currents in single mammalian smooth muscle cells. Nature. 1985. 316:345–347.20. Holm AJ, Rich A, Miller ST, Strege P, Ou Y, Gibbons SJ, Sarr MG, Szurszewski JH, Rae JL, Farrugia G. Sodium current in human jejunal circular smooth muscle cells. Gastroenterology. 2002. 122:178–187.
Article21. Aickin CC. Investigation of factors affecting the intracellular sodium activity in the smooth muscle of guinea-pig ureter. J Physiol. 1987. 385:483–505.
Article22. Lee CO, Fozzard H. Activities of potassium and sodium ions in rabbit heart muscle. J Gen Physiol. 1975. 65:695–708.
Article23. Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ. Improved pathch-clamp techniques for high resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 1981. 391:85–100.24. Bhattacharjee A, Joiner WJ, Wu M, Yang Y, Sigworth FJ, Kaczmarek LK. Slick (Slo2.1), a rapid-gating sodium-activated potassium channel inhibited by ATP. J Neurosci. 2003. 23:11681–11691.25. Dryer SE. Na+-activated K+ channels and voltage-dependent K+ current in spinal neurons of the frog embryo. J Physiol. 1991. 462:349–372.26. Bhattacharjee A, Gan Li, Kaczmarek LK. Localization of the slack potassium channel in the rat central nervous system. J Comp Neurol. 2002. 454:241–254.
Article27. Bhattacharjee A, Kaczmarek LK. For K+ channels, Na+ is the new Ca2+. Trends Neurosci. 2005. 28:422–428.28. Wang Z, Kimitsu T, Noma T. Conductance properties of the Na+-activated K+ channel in guinea-pig ventricular cells. J Physiol. 1991. 433:241–257.29. Leyssens A, Caemeliet E. Block of the transient inward current by R56865 in guinea-pig ventricular myocytes. Eur J Pharmacol. 1991. 196:43–51.
Article30. Li Y, Sato T, Arita M. Bepridil blunts the shortening of action potential duration caused by metabolic inhibition via blockade of ATP-sensitive K+ channels and Na+-activated K+ channels. J Pharmacol Exp Ther. 1999. 291:562–568.31. Pike MM, Kitakaze M, Marban E. 23Na-NMR measurements of intracellular sodium in intact perfused ferret hearts during ischemia and reperfusion. Am J Physiol. 1990. 259:H767–H773.32. van Echteld CJ, Kirkels JH, Eijgelshoven MH, van der Meer P, Ruigrok TJC. Intracellular sodium during ischemia and calcium-free perfusion: a 23Na NMR study. J Mol Cell Cardiol. 1991. 23:297–307.33. Glitch HG. Characteristics of active Na transport in intact cardia cells. Am J Physiol. 1979. 236:H189–H199.34. Rose CR, Konnerth A. NMDA receptor-mediated Na+ signals in spines and dendrites. J Neurosci. 2001. 21:4207–4214.35. Kim U, McCormick DA. Functional and ionic properties of a slow afterhyperpolarization in ferret perigeniculate neurons in vitro. J Neurophysiol. 1998. 80:1222–1235.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effects of Na-Ca Exchange Mechanism on the Action Potential and Membrane Currents in the Single Cells of the Guinea-Pig and the Rabbit Heart
- Modulation of outward potassium currents by nitric oxide in longitudinal smooth muscle cells of guinea-pig ileum
- Effects of ryanodine on the intracellular Na+ activity and tension and action potentials of rat and guinea pig cardiac ventricular muscles
- Depolarizing Mechanisms of Acetylcholine in Gastric Smooth Muscle
- Ca2+-activated K+ Current in Freshly Isolated c-Kit Positive Cells in Guinea-pig Stomach