Korean J Ophthalmol.
2013 Aug;27(4):268-275. 10.3341/kjo.2013.27.4.268.
Foveal Thickness between Stratus and Spectralis Optical Coherence Tomography in Retinal Diseases
- Affiliations
-
- 1Department of Ophthalmology, Seoul National University Bundang Hospital, Seoul National University College of Medicine, Seongnam, Korea. sejoon1@snu.ac.kr
- 2Department of Ophthalmology, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea.
- KMID: 1705423
- DOI: http://doi.org/10.3341/kjo.2013.27.4.268
Abstract
- PURPOSE
To compare the foveal thickness (FT) parameters measured by Stratus optical coherence tomography (OCT) and Spectralis OCT in various retinal diseases and to construct conversion formulas between the two types of OCT devices.
METHODS
We examined 366 consecutive patients (475 eyes) with retinal diseases and 13 normal controls (13 eyes). The patients were categorized into eight retinal disease groups. The mean amount and distribution of foveal thickness differences (FTD) measured by Stratus and Spectralis OCT were determined, and conversion formulas were constructed for Stratus OCT FT from Spectralis OCT FT for each retinal disease group.
RESULTS
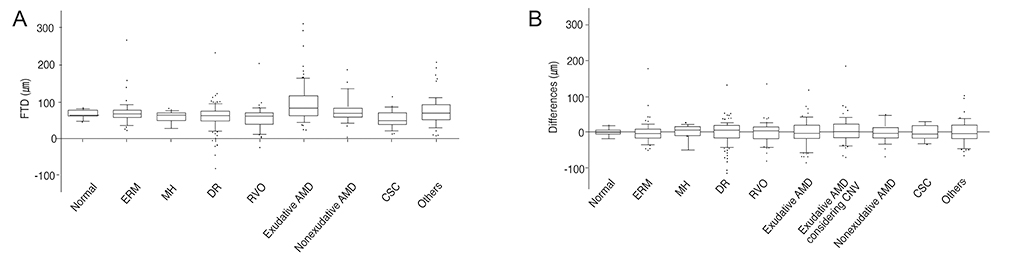
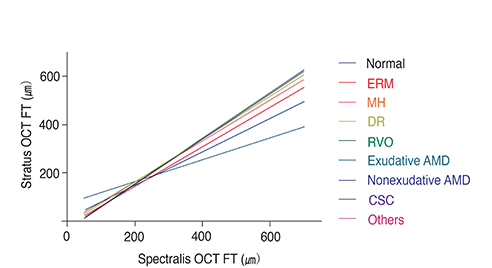
Among retinal diseases, the mean FTD was significantly larger in exudative age-related macular degeneration (AMD) patients (mean +/- SD, 94.0 +/- 55.0 microm) compared to normal subjects (66.2 +/- 11.7 microm; p < 0.0001). The proportion of eyes with a mean FTD outside 1.96 standard deviations of normal subject FTD was greatest in the exudative AMD (50.0%) group and smallest in the macular hole (18.2%) group. The predicted FTs obtained through the conversion formulas showed lower variance than the actual FTD values, especially in the exudative AMD group. The prediction line for exudative AMD deviated most from that of normal subjects.
CONCLUSIONS
FTD shows diverse values and variances among various retinal diseases, especially in exudative AMD, which indicates that Stratus OCT FT cannot be predicted from Spectralis OCT FT by FTD value alone. We constructed statistically significant conversion formulas, which provided more reliable methods to predict Stratus OCT-measured FT from Spectralis OCT measurements for different retinal disease groups.
MeSH Terms
Figure
Reference
-
1. Leung CK, Cheung CY, Weinreb RN, et al. Comparison of macular thickness measurements between time domain and spectral domain optical coherence tomography. Invest Ophthalmol Vis Sci. 2008; 49:4893–4897.2. Huang D, Swanson EA, Lin CP, et al. Optical coherence tomography. Science. 1991; 254:1178–1181.3. Hee MR, Izatt JA, Swanson EA, et al. Optical coherence tomography of the human retina. Arch Ophthalmol. 1995; 113:325–332.4. Puliafito CA, Hee MR, Lin CP, et al. Imaging of macular diseases with optical coherence tomography. Ophthalmology. 1995; 102:217–229.5. Thomas D, Duguid G. Optical coherence tomography: a review of the principles and contemporary uses in retinal investigation. Eye (Lond). 2004; 18:561–570.6. Forte R, Cennamo GL, Finelli ML, de Crecchio G. Comparison of time domain Stratus OCT and spectral domain SLO/OCT for assessment of macular thickness and volume. Eye (Lond). 2009; 23:2071–2078.7. Paunescu LA, Schuman JS, Price LL, et al. Reproducibility of nerve fiber thickness, macular thickness, and optic nerve head measurements using Stratus OCT. Invest Ophthalmol Vis Sci. 2004; 45:1716–1724.8. Gurses-Ozden R, Teng C, Vessani R, et al. Macular and retinal nerve fiber layer thickness measurement reproducibility using optical coherence tomography (OCT-3). J Glaucoma. 2004; 13:238–244.9. Kakinoki M, Sawada O, Sawada T, et al. Comparison of macular thickness between Cirrus HD-OCT and Stratus OCT. Ophthalmic Surg Lasers Imaging. 2009; 40:135–140.10. Joeres S, Tsong JW, Updike PG, et al. Reproducibility of quantitative optical coherence tomography subanalysis in neovascular age-related macular degeneration. Invest Ophthalmol Vis Sci. 2007; 48:4300–4307.11. Drexler W, Sattmann H, Hermann B, et al. Enhanced visualization of macular pathology with the use of ultrahigh-resolution optical coherence tomography. Arch Ophthalmol. 2003; 121:695–706.12. Sayanagi K, Sharma S, Kaiser PK. Comparison of retinal thickness measurements between three-dimensional and radial scans on spectral-domain optical coherence tomography. Am J Ophthalmol. 2009; 148:431–438.13. Wojtkowski M, Bajraszewski T, Targowski P, Kowalczyk A. Real-time in vivo imaging by high-speed spectral optical coherence tomography. Opt Lett. 2003; 28:1745–1747.14. Alam S, Zawadzki RJ, Choi S, et al. Clinical application of rapid serial fourier-domain optical coherence tomography for macular imaging. Ophthalmology. 2006; 113:1425–1431.15. Srinivasan VJ, Wojtkowski M, Witkin AJ, et al. High-definition and 3-dimensional imaging of macular pathologies with high-speed ultrahigh-resolution optical coherence tomography. Ophthalmology. 2006; 113:2054.e1–2054.e14.16. Lalwani GA, Rosenfeld PJ, Fung AE, et al. A variable-dosing regimen with intravitreal ranibizumab for neovascular age-related macular degeneration: year 2 of the PrONTO Study. Am J Ophthalmol. 2009; 148:43–58.e1.17. Shimura M, Nakazawa T, Yasuda K, et al. Comparative therapy evaluation of intravitreal bevacizumab and triamcinolone acetonide on persistent diffuse diabetic macular edema. Am J Ophthalmol. 2008; 145:854–861.18. Soheilian M, Ramezani A, Obudi A, et al. Randomized trial of intravitreal bevacizumab alone or combined with triamcinolone versus macular photocoagulation in diabetic macular edema. Ophthalmology. 2009; 116:1142–1150.19. Grover S, Murthy RK, Brar VS, Chalam KV. Comparison of retinal thickness in normal eyes using Stratus and Spectralis optical coherence tomography. Invest Ophthalmol Vis Sci. 2010; 51:2644–2647.20. Han IC, Jaffe GJ. Comparison of spectral- and time-domain optical coherence tomography for retinal thickness measurements in healthy and diseased eyes. Am J Ophthalmol. 2009; 147:847–858. 858.e121. Lammer J, Scholda C, Prunte C, et al. Retinal thickness and volume measurements in diabetic macular edema: a comparison of four optical coherence tomography systems. Retina. 2011; 31:48–55.22. Ibrahim MA, Sepah YJ, Symons RC, et al. Spectral- and time-domain optical coherence tomography measurements of macular thickness in normal eyes and in eyes with diabetic macular edema. Eye (Lond). 2012; 26:454–462.23. Krebs I, Hagen S, Smretschnig E, et al. Conversion of Stratus optical coherence tomography (OCT) retinal thickness to Cirrus OCT values in age-related macular degeneration. Br J Ophthalmol. 2011; 95:1552–1554.24. Sayanagi K, Sharma S, Yamamoto T, Kaiser PK. Comparison of spectral-domain versus time-domain optical coherence tomography in management of age-related macular degeneration with ranibizumab. Ophthalmology. 2009; 116:947–955.25. Cukras C, Wang YD, Meyerle CB, et al. Optical coherence tomography-based decision making in exudative age-related macular degeneration: comparison of time- vs spectral-domain devices. Eye (Lond). 2010; 24:775–783.26. Menke MN, Dabov S, Knecht P, Sturm V. Reproducibility of retinal thickness measurements in patients with age-related macular degeneration using 3D Fourier-domain optical coherence tomography (OCT) (Topcon 3D-OCT 1000). Acta Ophthalmol. 2011; 89:346–351.27. Ray R, Stinnett SS, Jaffe GJ. Evaluation of image artifact produced by optical coherence tomography of retinal pathology. Am J Ophthalmol. 2005; 139:18–29.28. Leung CK, Chan WM, Chong KK, et al. Alignment artifacts in optical coherence tomography analyzed images. Ophthalmology. 2007; 114:263–270.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Comparison of Retinal Nerve Fiber Layer Thickness between Stratus and Spectralis OCT
- Comparison of Time Domain OCT and Spectrum Domain OCT for Retinal Nerve Fiber Layer Assessment
- Comparison of Macular Retinal Thickness among Four Optical Coherence Tomography Devices in Healthy Young Subjects
- Choroidal Thickness at the Outside of Fovea in Diabetic Retinopathy Using Spectral-Domain Optical Coherence Tomography
- Foveal Retinal Detachment Diagnosed by Optical Coherence Tomography after Successful Retinal Detachment Surgery