Korean J Physiol Pharmacol.
2008 Apr;12(2):73-77. 10.4196/kjpp.2008.12.2.73.
Testosterone Relaxes Rabbit Seminal Vesicle by Calcium Channel Inhibition
- Affiliations
-
- 1Department of Physiology, College of Medicine, Chung-Ang University, Seoul 156-756, Korea. heeyun@cau.ac.kr
- 2Department of Urology, College of Medicine, Chung-Ang University, Seoul 156-756, Korea.
- 3Department of Neurology, Seoul Medical Center, Seoul 135-740, Korea.
- KMID: 1486146
- DOI: http://doi.org/10.4196/kjpp.2008.12.2.73
Abstract
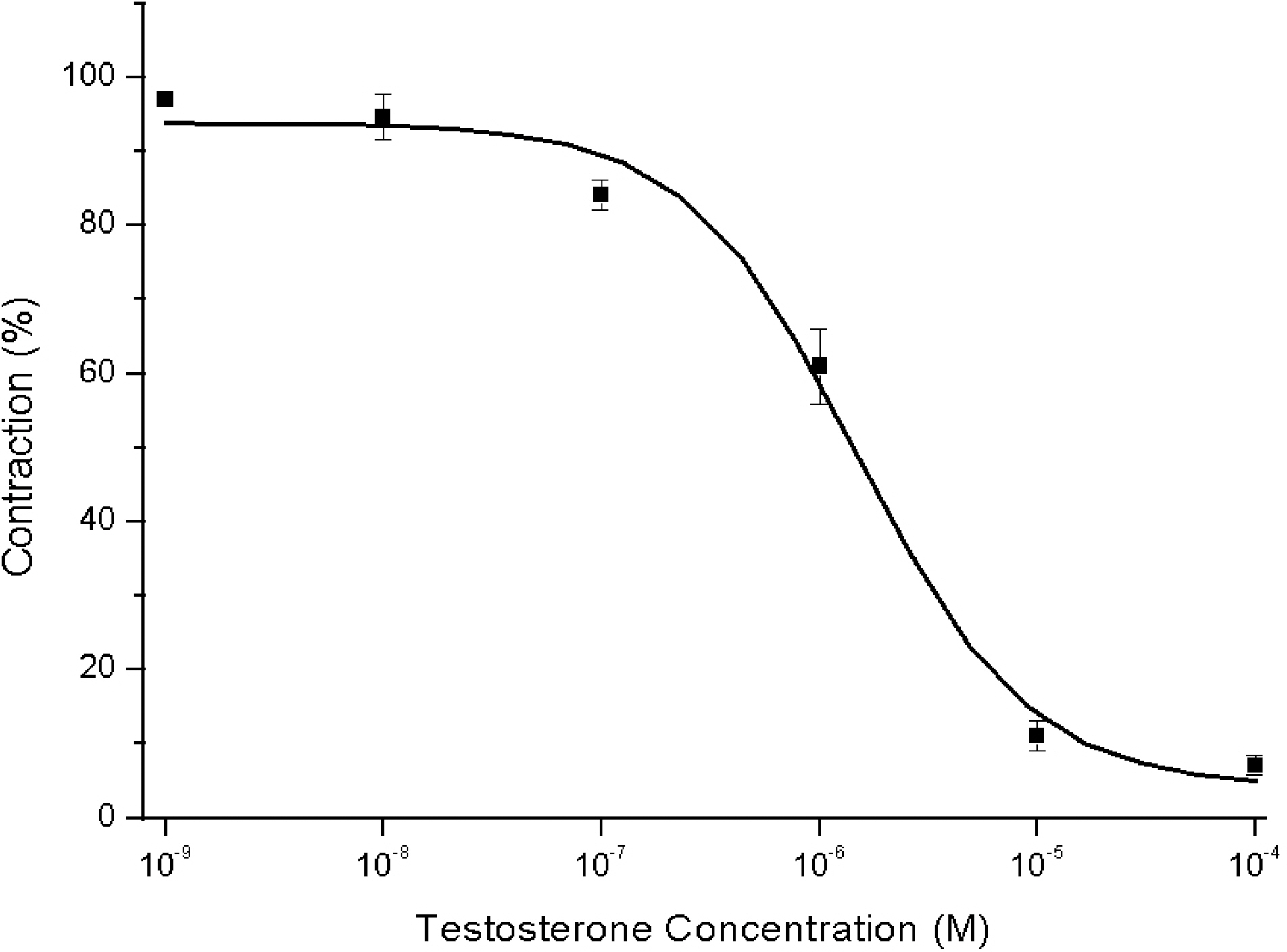
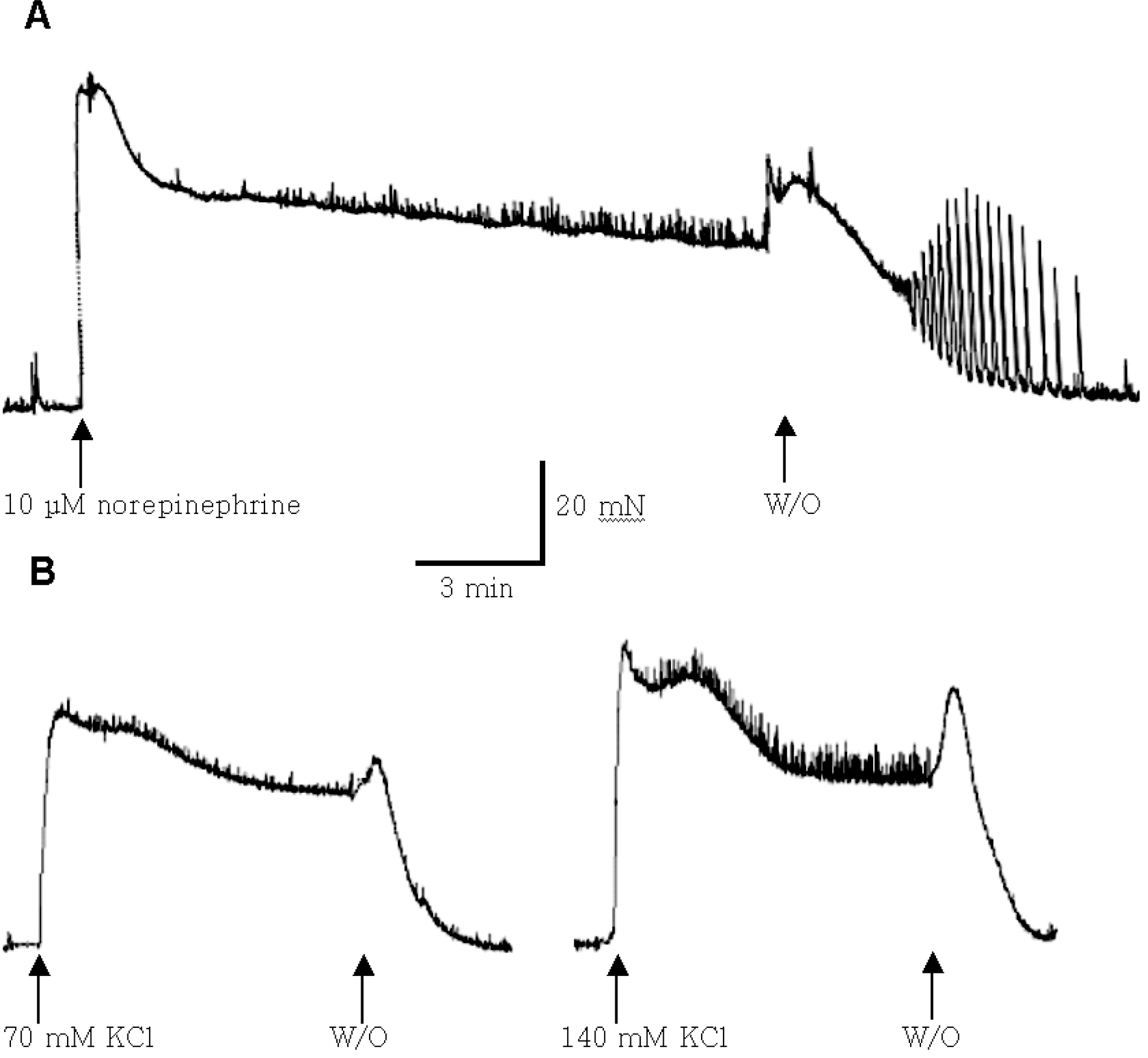
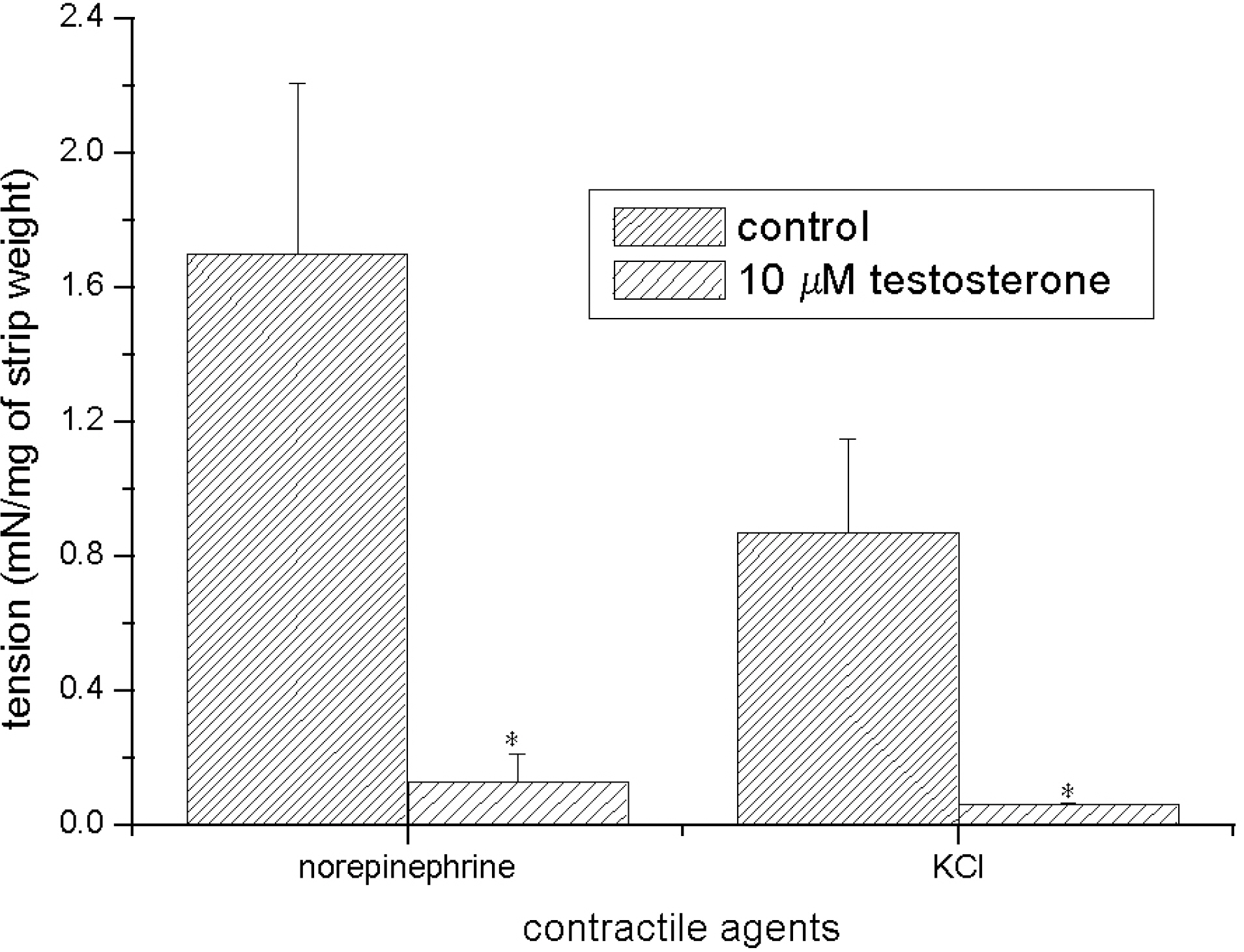
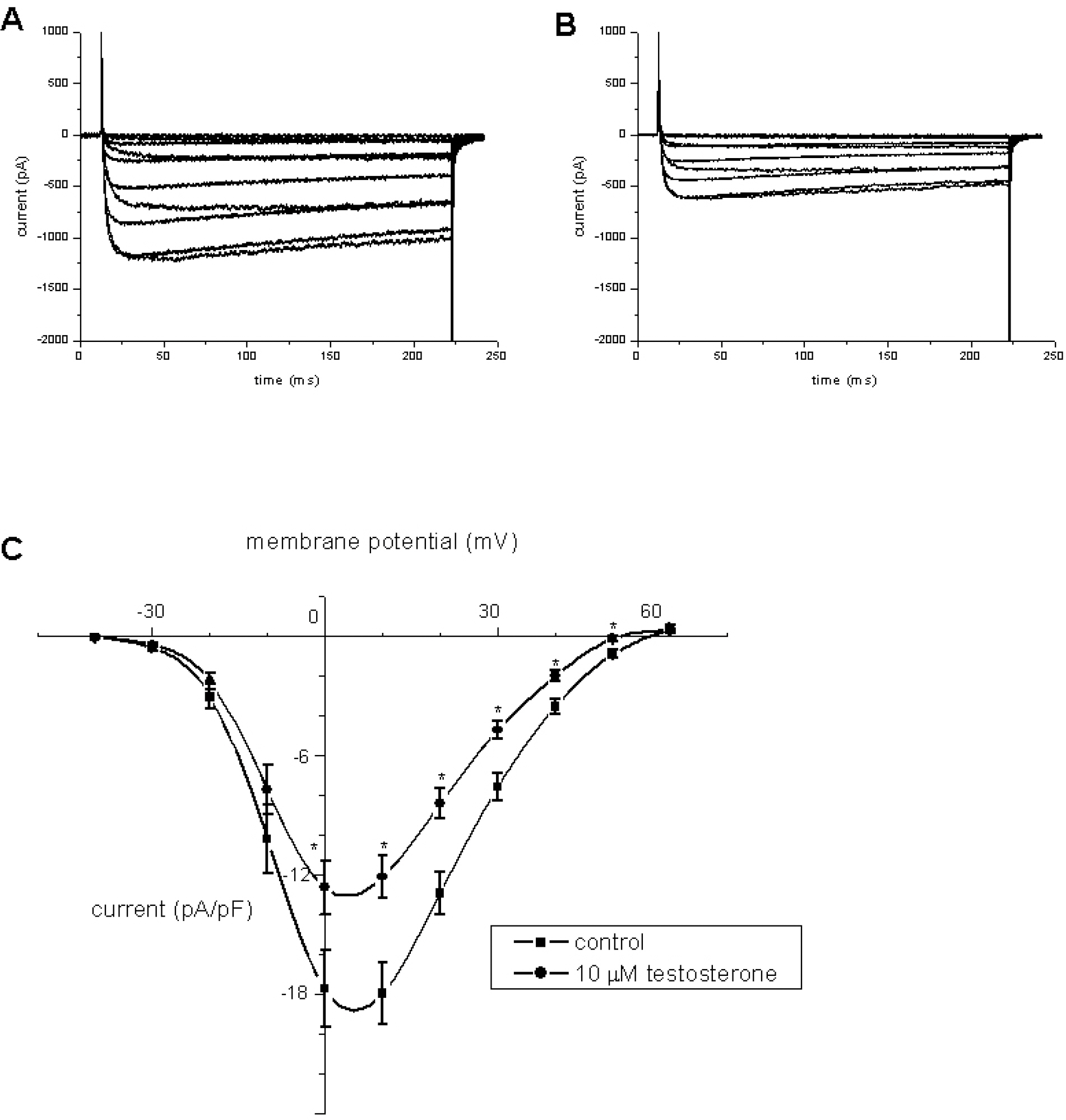
- Recent studies have documented that testosterone relaxes several smooth muscles by modulating K+channel activities. Smooth muscles of seminal vesicles play a fundamental role in ejaculation, which might involve testosterone. This study was aimed to assess the role of testosterone in seminal vesicular motility by studying its effects on contractile agents and on the ion channels of single vesicular myocytes in a rabbit model. The contractile responses of circular smooth muscle strips of rabbit seminal vesicles to norepinephrine (10 micrometer), a high concentration of KCl (70 mM), and testosterone (10 micrometer were observed. Single vesicular myocytes of rabbit were isolated using proteolytic enzymes including collagenase and papain. Inside-out, attached, and whole-cell configurations were examined using the patch clamp technique. The applications of 10 micrometernorepinephrine or 70 mM KCl induced tonic contractions, and 10 micrometertestosterone (pharmacological concentration) evoked dose-dependent relaxations of these precontracted strips. Various K+channel blockers, such as tetraethylammonium (TEA; 10 mM), iberiotoxin (0.1 micrometer), 4-aminopyridine (4-AP, 10 micrometer, or glibenclamide (10 micrometer rarely affected these relaxations. Single channel data (of inside-out and attached configurations) of BK channel activity were also hardly affected by testosterone (10 micrometer). On the other hand, however, testosterone reduced L-type Ca2+currents significantly, and found to induce acute relaxation of seminal vesicular smooth muscle and this was mediated, at least in part, by Ca2+current inhibition in rabbit.
Keyword
MeSH Terms
-
4-Aminopyridine
Calcium
Calcium Channels
Collagenases
Contracts
Ejaculation
Glyburide
Hand
Ion Channels
Male
Muscle Cells
Muscle, Smooth
Norepinephrine
Papain
Peptide Hydrolases
Peptides
Relaxation
Seminal Vesicles
Testosterone
Tetraethylammonium
4-Aminopyridine
Calcium
Calcium Channels
Collagenases
Glyburide
Ion Channels
Norepinephrine
Papain
Peptide Hydrolases
Peptides
Testosterone
Tetraethylammonium
Figure
Cited by 2 articles
-
The Relaxing Effect of α-Defensin 1 on the Adrenergic Responses of Rat Bladder
Shin Young Lee, Don Kyu Kim, Kyung Do Kim, Soon Chul Myung, Moo Yeol Lee
Korean J Physiol Pharmacol. 2011;15(3):143-147. doi: 10.4196/kjpp.2011.15.3.143.Spontaneous Contractions Augmented by Cholinergic and Adrenergic Systems in the Human Ureter
Hyun Woo Lee, Cheol Hee Baak, Moo Yeol Lee, Young Chul Kim
Korean J Physiol Pharmacol. 2011;15(1):37-41. doi: 10.4196/kjpp.2011.15.1.37.
Reference
-
Aumuller G., Riva A. Morphology and functions of the human seminal vesicle. Andrologia. 24:183–196. 1992.Bancroft J. The endocrinology of sexual arousal. J Endocrinol. 186:411–427. 2005.
ArticleChou TM., Sudhir K., Hutchison SJ., Ko E., Amidon TM., Collins P., Chatterjee K. Testosterone induces dilation of canine coronary conductance and resistance arteries in vivo. Circulation. 94:2614–2619. 1996.
ArticleCohen PG. The association of premature ejaculation and hypogonadotrophic hypogonadism. J Sex Marital Ther. 23:208–211. 1997.Costarella CE., Stallone JN., Rutecki GW., Whittier FC. Testosterone causes direct relaxation of rat thoracic aorta. J Pharmacol Exp Ther. 277:34–39. 1996.Deenadayalu VP., White RE., Stallone JN., Gao X., Garcia AJ. Testosterone relaxes coronary arteries by opening the large-conductance, calcium-activated potassium channel. Am J Physiol. 281:H1720–H1727. 2001.
ArticleDing AQ., Stallone JN. Testosterone-induced relaxation of rat aorta is androgen structure specific and involves K+ channel activation. J Appl Physiol. 91:2742–2750. 2001.Er F., Michels G., Brandt MC., Khan I., Hasse H., Eicks M., Lindner M., Hoppe UC. Impact of testosterone on cardiac L-type calcium channels and Ca2+ sparks: acute actions antagonize chronic effects. Cell Calcium. 41:467–477. 2007.Hamill RW., Schroeder B. Hormonal regulation of adult sympathetic neurons: the effects of castration on neuropeptide Y, norepinephrine, and tyrosine hydroxylase activity. J Neurobiol. 21:731–742. 1990.
ArticleHib J., Ponzio R., Gomez DE., Vilar O. Contractile behaviour of rat seminal vesicle after gonadectomy, testosterone replacement and cyproterone treatment. Andrologia. 17:435–439. 1985.
ArticleKashiwagi B., Shibata Y., Ono Y., Suzuki K., Honma S., Yamanaka H. Effect of androgen concentration on seminal vesicle blood flow in rats-establishment of new highly sensitive simultaneous androgen measurement method. Urology. 66:218–223. 2005.
ArticleKim J., Cole D., Johnson A., Centenera V., Schenkman E., Durham J., Azzaro A., Mawhinney M. Androgen induced norepinephrine release from postganglionic neurons mediates accessory sex organ smooth muscle proliferation. Urology. 167:1897–1904. 2002.
ArticleMelvin JE., McNeill TH., Hamill RW. Biochemical and morphological effects of castration on the postorganizational development of the hypogastric ganglion. Dev Brain Res. 38:131–139. 1988.
ArticleMotofei IG. A bihormonal model of normal sexual stimulation; the etiology of premature ejaculation. Med Hypotheses. 57:93–95. 2001.
ArticlePerusquia M., Navarrete E., Jasso-Kamel J., Montano LM. Androgens induce relaxation of contractile activity in pregnant human myometrium at term: a nongenomic action on L-type calcium channels. Biol Reprod. 73:214–221. 2005.Scragg JL., Jones RD., Channer KS., Jones TH., Peers C. Testosterone is a potent inhibitor of L-type Ca2+ channels. Biochem Biophys Res Commun. 318:503–506. 2004.Yue P., Chatterjee K., Beale C., Poole-Wilson PA., Collins P. Testosterone relaxes rabbit coronary arteries and aorta. Circulation. 91:1154–1160. 1995.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Relaxation Responses of Isolated Rabbit Seminal Vesicle to Polyamines
- The Study of K Channels in Smooth Muscle Cells of Rabbit Seminal Vesicle
- Effects of Phytoestrogen on Potassium Channel Activities of Smooth Muscle Cells of Rabbit Seminal Vesicle
- The Role of beta-Adrenergic Receptor in the Seminal Vesicle Contraction
- Laparoscopic Excision of Congenital Seminal Vesicle Cyst Associated with Ipsilateral Renal Agenesis