Korean J Pediatr Infect Dis.
2012 Dec;19(3):89-110.
Meningococcal Disease and Quadrivalent MenACWY-CRM Vaccine (Menveo(R))
- Affiliations
-
- 1Novartis Vaccines and Diagnostics Inc. USA. Theodore.tsai@novartis.com
Abstract
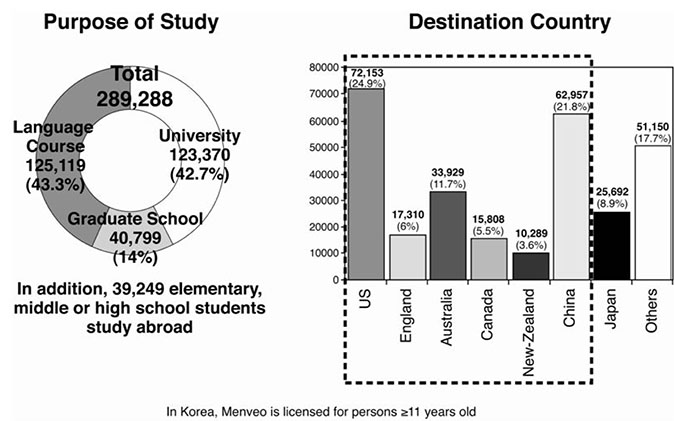
- Meningococcal Disease, manifesting as meningitis and septicemia, is a life-threatening bacterial infection that results in significant morbidity and mortality, particularly in childhood. Its epidemic potential and limited opportunities for clinical intervention due to its rapid course present unique public health and clinical challenges. Incidence is highest in infants and young children, with a secondary peak of risk in adolescents. Approximately 10% of cases are fatal and survivors can be left with serious and permanent sequelae including amputations, hearing loss and cognitive impairment. Transmission is only from human-to-human, by infected respiratory tract secretions or saliva and therefore crowding poses a tremendously elevated risk for disease development. Military recruits and university students are at high risk due to the high carriage rate in adolescents, their behavior patterns and close contact. Menveo(R) (Novartis Vaccines and Diagnostics), a novel quadrivalent meningococcal conjugate vaccine directed against meningococcal serogroups A, C, W-135 and Y, has been shown to be immunogenic and well tolerated in all age groups and was recently licensed for use in Korea. Recent cases and deaths among military recruits drew public attention to their elevated risk and the Korean government has recommended vaccination of all new military recruits. Many Korean students seek to attend school, university, or language institutes in countries where routine meningococcal vaccination is required - clinicians should be aware of such requirements to ensure that students are vaccinated prior to arrival in the destination country.
MeSH Terms
-
Academies and Institutes
Adolescent
Amputation
Bacterial Infections
Child
Crowding
Dietary Sucrose
Hearing Loss
Humans
Incidence
Infant
Korea
Meningitis
Meningococcal Vaccines
Military Personnel
Neisseria meningitidis
Public Health
Respiratory System
Saliva
Sepsis
Survivors
Vaccination
Vaccines
Dietary Sucrose
Meningococcal Vaccines
Vaccines
Figure
Reference
-
1. McIntyre PB, O'Brien KL, Greenwood B, van de Beek D. Effect of vaccines on bacterial meningitis worldwide. Lancet. 2012. 380:1703–1711.
Article2. Stephens DS, Greenwood B, Brandtzaeg P. Epidemic meningitis, meningococcemia, and Neisseria meningitidis. Lancet. 2007. 369:2196–2210.3. Centers for Disease Control and Prevention. Outbreak of meningococcal disease associated with an elementary school -- Oklahoma, March 2010. MMWR Morb Mortal Wkly Rep. 2012. 61:217–221.4. Krause G, Blackmore C, Wiersma S, Lesneski C, Gauch L, Hopkins RS. Mass vaccination campaign following community outbreak of meningococcal disease. Emerg Infect Dis. 2002. 8:1398–1403.
Article5. Nolan P. The Rhode Island meningitis vaccine experience --mass vaccination campaigns, politics and health policy. Med Health R I. 2004. 87:65–67.6. Thompson MJ, Ninis N, Perera R, Mayon-White R, Phillips C, Bailey L. Clinical recognition of meningococcal disease in children and adolescents. Lancet. 2006. 367:397–403.7. Fitzwater SP, Watt JP, Levine OS, Santosham M. Haemophilus influenzae type b conjugate vaccines: considerations for vaccination schedules and implications for developing countries. Hum Vaccin. 2010. 6:810–818.
Article8. Tan TQ. Pediatric invasive pneumococcal disease in the United States in the era of pneumococcal conjugate vaccines. Clin Microbiol Rev. 2012. 25:409–419.
Article9. Yazdankhah SP, Caugant DA. Neisseria meningitidis: an overview of the carriage state. J Med Microbiol. 2004. 53:821–832.10. van Deuren M, Brandtzaeg P, van der Meer JW. Update on meningococcal disease with emphasis on pathogenesis and clinical management. Clin Microbiol Rev. 2000. 13:144–166.
Article11. Davila S, Wright VJ, Khor CC, Sim KS, Binder A, Breunis WB, et al. Genome-wide association study identifies variants in the CFH region associated with host susceptibility to meningococcal disease. Nat Genet. 2010. 42:772–776.
Article12. Owen EP, Leisegang F, Whitelaw A, Simpson J, Baker S, Würzner R, et al. Complement 2013-02-14component C5 and C6 mutation screening indicated in meningococcal disease in South Africa. S Afr Med J. 2012. 102:525–527.
Article13. Helminen M, Seitsonen S, Jarva H, Meri S, Järvelä IE. A novel mutation W388X underlying properdin deficiency in a Finnish family. Scand J Immunol. 2012. 75:445–448.
Article14. Ram S, Lewis LA, Rice PA. Infections of people with complement deficiencies and patients who have undergone splenectomy. Clin Microbiol Rev. 2010. 23:740–780.
Article15. Hermans PW, Hazelzet JA. Plasminogen activator inhibitor type 1 gene polymorphism and sepsis. Clin Infect Dis. 2005. 41:S453–S458.
Article16. Bradley DT, Bourke TW, Fairley DJ, Borrow R, Shields MD, Young IS, et al. Genetic susceptibility to invasive meningococcal disease: MBL2 structural polymorphisms revisited in a large case-control study and a systematic review. Int J Immunogenet. 2012. 39:328–337.
Article17. Girard MP, Preziosi MP, Aguado MT, Kieny MP. A review of vaccine research and development: meningococcal disease. Vaccine. 2006. 24:4692–4700.
Article18. Erickson L, De Wals P. Complications and sequelae of meningococcal disease in Quebec, Canada, 1990-1994. Clin Infect Dis. 1998. 26:1159–1164.
Article19. Viner RM, Booy R, Johnson H, Edmunds JW, Hudson L, Bedford H, et al. Outcomes of invasive meningococcal serogroup B disease in children and adolescents (MOSAIC): a case-control study. Lancet Neurol. 2012. 11:774–783.
Article20. van de Beek D, Brouwer MC, Thwaites GE, Tunkel AR. Advances in treatment of bacterial meningitis. Lancet. 2012. 380:1693–1702.
Article21. Dalton T, Cegielski P, Akksilp S, Asencios L, Campos Caoili J, Cho SN, et al. Prevalence of and risk factors for resistance to second-line drugs in people with multidrug-resistant tuberculosis in eight countries: a prospective cohort study. Lancet. 2012. 380:1406–1417.
Article22. World Health Organization. Meningococcal vaccines: WHO position paper, November 2011. Wkly Epidemiol Rec. 2011. 86:521–539.23. Agrawal S, Nadel S. Acute bacterial meningitis in infants and children. epidemiology and management. Paediatr Drugs. 2011. 13:385–400.24. Goldschneider I, Gotschlich EC, Artenstein MS. Human immunity to the meningococcus. I. The role of humoral antibodies. J Exp Med. 1969. 129:1307–1326.25. Harrison LH, Pass MA, Mendelsohn AB, Egri M, Rosenstein NE, Bustamante A, et al. Invasive meningococcal disease in adolescents and young adults. JAMA. 2001. 286:694–699.
Article26. Halperin SA, Bettinger JA, Greenwood B, Harrison LH, Jelfs J, Ladhani SN, et al. The changing and dynamic epidemiology of meningococcal disease. Vaccine. 2012. 30:Suppl 2. B26–B36.
Article27. Harrison LH, Trotter CL, Ramsay ME. Global epidemiology of meningococcal disease. Vaccine. 2009. 27:Suppl 2. B51–B63.
Article28. du Plessis M, Moodley C, Mothibeli KM, Fali A, Klugman KP, von Gottberg A, et al. Population snapshot of invasive serogroup B meningococci in South Africa from 2005 to 2008. J Clin Microbiol. 2012. 50:2577–2584.
Article29. Pollard AJ. Global epidemiology of meningococcal disease and vaccine efficacy. Pediatr Infect Dis J. 2004. 23:S274–S279.
Article30. Bröker M, Jacobsson S, Kuusi M, Pace D, Simões MJ, Skoczynska A, et al. Meningococcal serogroup Y emergence in Europe: Update 2011. Hum Vaccin Immunother. 2012. 8:[Epub ahead of print].31. Vyse A, Wolter JM, Chen J, Ng T, Soriano-Gabarro M. Meningococcal disease in Asia: an under-recognized public health burden. Epidemiol Infect. 2011. 139:967–985.
Article32. Kilic A, Urwin R, Li H, Saracli MA, Stratton CW, Tang YW. Clonal Spread of Serogroup W135 Meningococcal Disease in Turkey. J Clin Microbiol. 2006. 44:222–224.
Article33. Kriz P, Wieffer H, Holl K, Rosenlund M, Budhia S, Vyse A. Changing epidemiology of meningococcal disease in Europe from the mid-20th to the early 21st century. Expert Rev Vaccines. 2011. 10:1477–1486.
Article34. Cho HK, Lee H, Kang JH, Kim KN, Kim DS, Kim YK. The causative organisms of bacterial meningitis in Korean children in 1996-2005. J Korean Med Sci. 2010. 25:895–899.
Article35. Kim SA, Kim DW, Dong BQ, Kim JS, Anh DD, Kilgore PE. An expanded age range for meningococcal meningitis: molecular diagnostic evidence from population-based surveillance in Asia. BMC Infect Dis. 2012. 12:310.
Article36. Lee SO, Ryu SH, Park SJ, Ryu J, Woo JH, Kim YS. Meningococcal disease in the republic of Korea army: incidence and serogroups determined by PCR. J Korean Med Sci. 2003. 18:163–166.
Article37. Park HS, Lee DH, Seo PW, Kim SH, Choi TY, Kim JH. Carrier rate, serogroup and vaccination effect of Neisseria meningitidis on army trainees in Korea. Chungbuk Med J. 1995. 5:45–57.38. Holers VM. The spectrum of complement alternative pathway-mediated diseases. Immunol Rev. 2008. 223:300–316.
Article39. Zuber J, Fakhouri F, Roumenina LT, Loirat C, Frémeaux-Bacchi V. Use of eculizumab for atypical haemolytic uraemic syndrome and C3 glomerulopathies. Nat Rev Nephrol. 2012. 8:643–657.
Article40. Békondi C, Bernede C, Passone N, Minssart P, Kamalo C, Mbolidi D, et al. Primary and opportunistic pathogens associated with meningitis in adults in Bangui, Central African Republic, in relation to human immunodeficiency virus serostatus. Int J Infect Dis. 2006. 10:387–395.
Article41. Bruce MG, Rosenstein NE, Capparella JM, Shutt KA, Perkins BA, Collins M. Risk factors for meningococcal disease in college students. JAMA. 2001. 286:688–693.
Article42. Brundage JF, Ryan MA, Feighner BH, Erdtmann FJ. Meningococcal disease among United States military service members in relation to routine uses of vaccines with different serogroup-specific components, 1964-1998. Clin Infect Dis. 2002. 35:1376–1381.
Article43. MacLennan J, Kafatos G, Neal K, Andrews N, Cameron JC, Roberts R, et al. Social behavior and meningococcal carriage in British teenagers. Emerg Infect Dis. 2006. 12:950–957.
Article44. Dull PM, McIntosh ED. Meningococcal vaccine development - from glycoconjugates against MenACWY to proteins against MenB - potential for broad protection against meningococcal disease. Vaccine. 2012. 30:Suppl 2. B18–B25.
Article45. Lee LH, Blake MS. Effect of increased CRM197 carrier protein dose on meningococcal C bactericidal antibody response. Clin Vaccine Immunol. 2012. 19:551–556.
Article46. Andrews N, Borrow R, Miller E. Validation of serological correlate of protection for meningococcal C conjugate vaccine by using efficacy estimates from postlicensure surveillance in England. Clin Diagn Lab Immunol. 2003. 10:780–786.
Article47. Campbell H, Andrews N, Borrow R, Trotter C, Miller E. Updated postlicensure surveillance of the meningococcal C conjugate vaccine in England and Wales: effectiveness, validation of serological correlates of protection, and modeling predictions of the duration of herd immunity. Clin Vaccine Immunol. 2010. 17:840–847.
Article48. De Wals P, Deceuninck G, Lefebvre B, Boulianne N, De Serres G. Effectiveness of serogroup C meningococcal conjugate vaccine: a 7-year follow-up in Quebec, Canada. Pediatr Infect Dis J. 2011. 30:566–569.49. Macneil JR, Cohn AC, Zell ER, Schmink S, Miller E, Clark T, et al. Early estimate of the effectiveness of quadrivalent meningococcal conjugate vaccine. Pediatr Infect Dis J. 2011. 30:451–455.
Article50. Novak RT, Kambou JL, Diomandé FV, Tarbangdo TF, Ouédraogo-Traoré R, Sangaré L, et al. Serogroup A meningococcal conjugate accination in Burkina Faso: analysis of national surveillance data. Lancet Infect Dis. 2012. 12:757–764.
Article51. Centers for Disease Control and Prevention. Licensure of a meningococcal conjugate vaccine (Menveo) and guidance for use - Advisory Committee on Immunization Practices (ACIP), 2010. MMWR Morb Mortal Wkly Rep. 2010. 59:273.52. Klein NP, Reisinger KS, Johnston W, Odrljin T, Gill CJ, Bedell L, et al. Safety and immunogenicity of a novel quadrivalent meningococcal CRM-conjugate vaccine given concomitantly with routine vaccinations in infants. Pediatr Infect Dis J. 2012. 31:64–71.
Article53. Klein NP, Shepard J, Bedell L, Odrljin T, Dull P. Immunogenicity and safety of a quadrivalent meningococcal conjugate vaccine administered concomitantly with measles, mumps, rubella, varicella vaccine in healthy toddlers. Vaccine. 2012. 30:3929–3936.
Article54. Black S, Klein NP, Shah J, Bedell L, Karsten A, Dull PM. Immunogenicity and tolerability of a quadrivalent meningococcal glycoconjugate vaccine in children 2-10 years of age. Vaccine. 2010. 28:657–663.
Article55. Halperin SA, Gupta A, Jeanfreau R, Klein NP, Reisinger K, Walter E, et al. Comparison of the safety and immunogenicity of an investigational and a licensed quadrivalent meningococcal conjugate vaccine in children 2-10 years of age. Vaccine. 2010. 28:7865–7872.
Article56. Jackson LA, Baxter R, Reisinger K, Karsten A, Shah J, Bedell L, et al. Phase III comparison of an investigational quadrivalent meningococcal conjugate vaccine with the licensed meningococcal ACWY conjugate vaccine in adolescents. Clin Infect Dis. 2009. 49:e1–e10.
Article57. Jackson LA, Jacobson RM, Reisinger KS, Anemona A, Danzig LE, Dull PM. A randomized trial to determine the tolerability and immunogenicity of a quadrivalent meningococcal glycoconjugate vaccine in healthy adolescents. Pediatr Infect Dis J. 2009. 28:86–91.
Article58. Reisinger KS, Baxter R, Block SL, Shah J, Bedell L, Dull PM. Quadrivalent meningococcal vaccination of adults: phase III comparison of an investigational conjugate vaccine, MenACWY-CRM, with the licensed vaccine, Menactra. Clin Vaccine Immunol. 2009. 16:1810–1815.
Article59. Gill CJ, Baxter R, Anemona A, Ciavarro G, Dull P. Persistence of immune responses after a single dose of Novartis meningococcal serogroup A, C, W-135 and Y CRM-197 conjugate vaccine (Menveo®) or Menactra® among healthy adolescents. Hum Vaccin. 2010. 6:881–887.
Article60. Jackson LA, Baxter R, Gill CJ, Odrljin T, Dull P. Persistence of bactericidal antibodies and boosting after vaccination with Menveo® in adolescents enrolled in phase II and III clinical studies. 2011. In : Presented at 49th annual meeting of IDSA;61. Jacobson RM, Jackson LA, Reisinger K, Izu A, Odrljin T, Dull PM. Antibody persistence and response to a booster dose of a quadrivalent conjugate vaccine for meningococcal disease in adolescents. Pediatr Infect Dis J. 2012. 10. 30. [Epub ahead of print].
Article62. Gasparini R, Conversano M, Bona G, Gabutti G, Anemona A, Dull PM, et al. Randomized trial on the safety, tolerability, and immunogenicity of MenACWY-CRM, an investigational quadrivalent meningococcal glycoconjugate vaccine, administered concomitantly with a combined tetanus, reduced diphtheria, and acellular pertussis vaccine in adolescents and young adults. Clin Vaccine Immunol. 2010. 17:537–544.
Article63. Arguedas A, Soley C, Loaiza C, Rincon G, Guevara S, Perez A, et al. Safety and immunogenicity of one dose of MenACWY-CRM, an investigational quadrivalent meningococcal glycoconjugate vaccine, when administered to adolescents concomitantly or sequentially with Tdap and HPV vaccines. Vaccine. 2010. 28:3171–3179.
Article64. Pina ML, Bassily E, Machmer A, Hou V, Reinhart A. Safety and immunogenicity of a quadrivalent meningococcal polysaccharide diphtheria toxoid conjugate vaccine in infants and toddlers: three multicenter phase 3 studies. Pediatr Infect Dis J. 2012. 31:1173–1183.
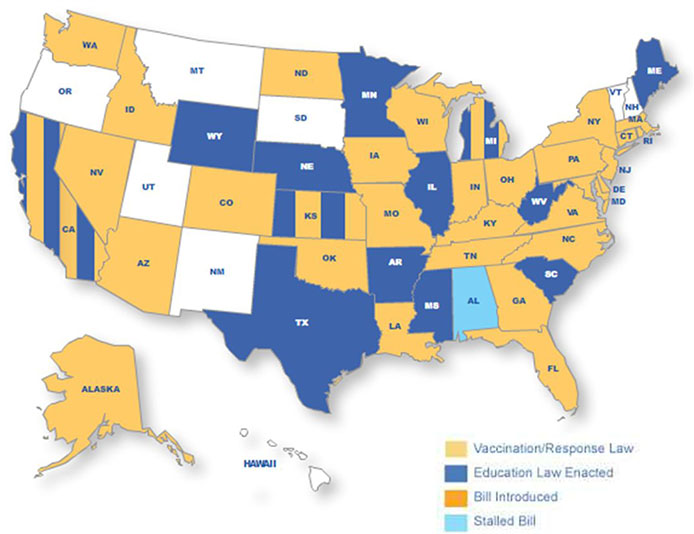
Article65. Lee HJ, Chung MH, Kim WJ, Hong YJ, Choi KM, Lee J, et al. Safety and immunogenicity of MenACWY-CRM vaccine administered to Korean adolescents and adults. 2012. In : Presented at 19th WONCA Asia Pacific Regional Conference;66. NATO Standardization Agency. National military strategies for vaccination of NATO forces. 2008. AMedP-23.67. Ministry of Hajj, Kingdon of Saudi Arabia. Saudi Ministry of Health Requirements. Accessed 18th Nov 2012. http://www.hajinformation.com/main/p3001.htm.68. Korean Educational Development Institute. accessed 11th May 2012. http://cesi.kedi.re.kr.69. The National Meningitis Association. Discover the NMA's Work State by State. accessed 18th Dec 2012. http://www.nmaus.org/awareness.70. Higher Education in Korea. accessed 11th May 2012. http://www.academyinfo.go.kr.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Immunogenicity of MenACWY-CRM in Korean Military Recruits: Influence of Tetanus-Diphtheria Toxoid Vaccination on the Vaccine Response to MenACWY-CRM
- One-year antibody persistence and safety of a 4-dose schedule of MenACWY-CRM in healthy infants from South Korea
- Comparison of Immune Responses to Two Quadrivalent Meningococcal Conjugate Vaccines (CRM197 and Diphtheria Toxoid) in Healthy Adults
- Meningococcal Vaccines
- A Case Study of Meningococcal Vaccination At a University Hospital