Korean J Physiol Pharmacol.
2013 Jun;17(3):197-201. 10.4196/kjpp.2013.17.3.197.
Anti-Oxidative Activity of Pectin and Its Stabilizing Effect on Retinyl Palmitate
- Affiliations
-
- 1College of Pharmacy, Chung-Ang University, Seoul 156-756, Korea. jaehwi@cau.ac.kr
- 2Department of Pharmacology, College of Medicine, Chung-Ang University, Seoul 156-756, Korea. jhjeong3@cau.ac.kr
- KMID: 1429298
- DOI: http://doi.org/10.4196/kjpp.2013.17.3.197
Abstract
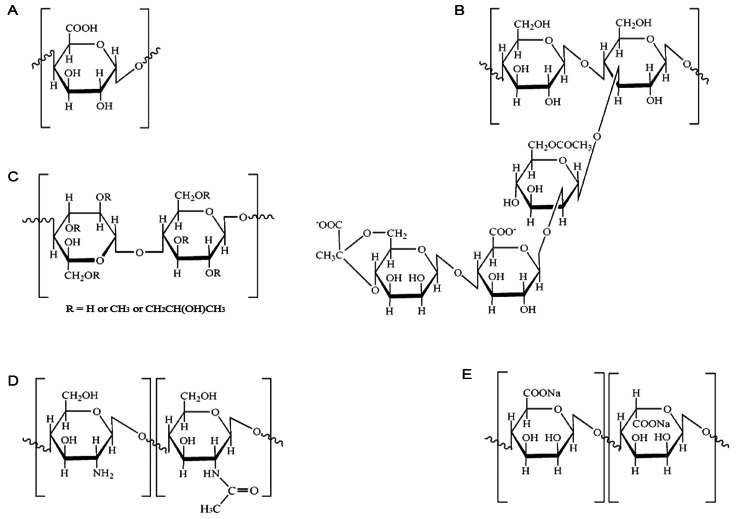
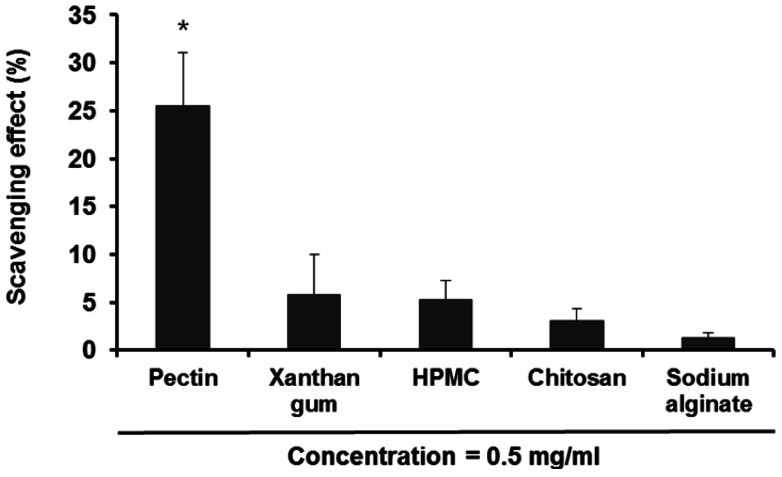
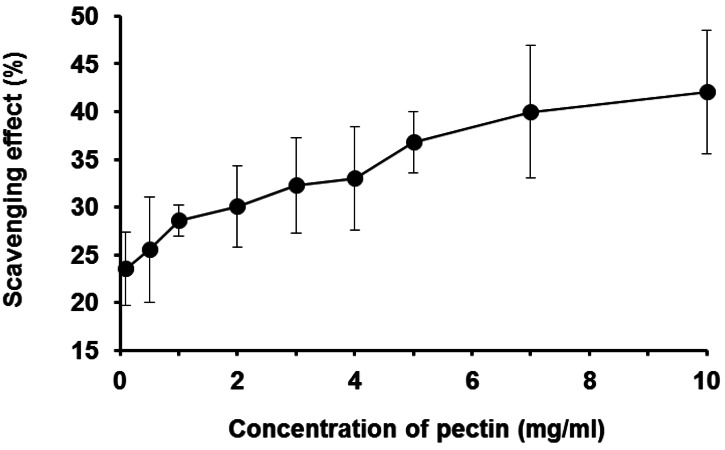
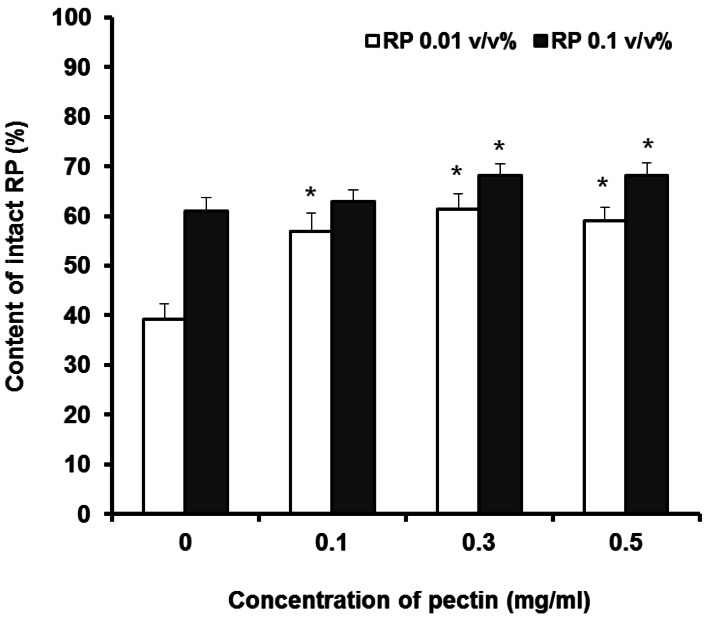
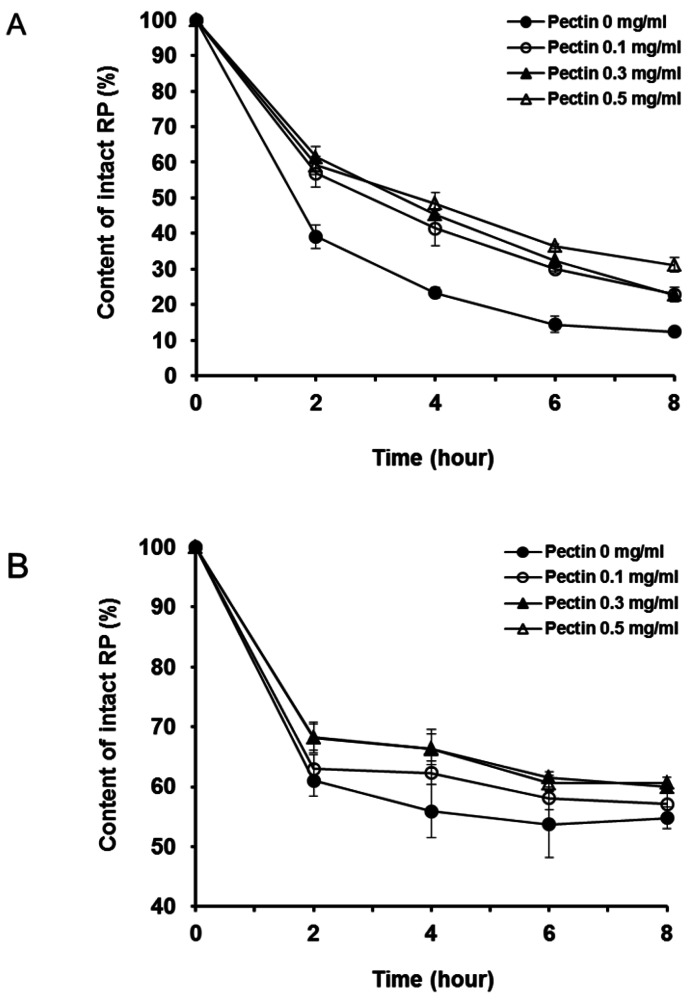
- The purpose of this study was to examine the anti-oxidative activity of pectin and other polysaccharides in order to develop a cosmeceutical base having anti-oxidative effects towards retinyl palmitate (RP). The anti-oxidative stabilizing effects of pectin and other polysaccharides on RP were evaluated by DPPH assay and then the stabilizing effect of pectin on RP was examined as a function of time. Among the polysaccharides we examined, pectin exhibited a considerably higher anti-oxidative activity, with an approximately 5-fold greater DPPH radical scavenging effect compared to other polysaccharides. The DPPH radical scavenging effect of pectin increased gradually with increasing concentrations of pectin. At two different RP concentrations, 0.01 and 0.1% in ethanol, addition of pectin improved the stability of RP in a concentration dependent manner. The stabilizing effect of pectin on RP was more effective for the lower concentration of RP (0.01%, v/v). Further, degradation of RP was reduced following the addition of pectin as measured over 8 hours. From the results obtained, it can be suggested that pectin may be a promising ingredient for cosmeceutical bases designed to stabilize RP or other pharmacological agents subject to degradation by oxidation.
Figure
Cited by 1 articles
-
Pectin Micro- and Nano-capsules of Retinyl Palmitate as Cosmeceutical Carriers for Stabilized Skin Transport
Jieun Ro, Yeongseok Kim, Hyeongmin Kim, Kyunghee Park, Kwon-Eun Lee, Prakash Khadka, Gyiae Yun, Juhyun Park, Suk Tai Chang, Jonghwi Lee, Ji Hoon Jeong, Jaehwi Lee
Korean J Physiol Pharmacol. 2015;19(1):59-64. doi: 10.4196/kjpp.2015.19.1.59.
Reference
-
1. Boehnlein J, Sakr A, Lichtin JL, Bronaugh RL. Characterization of esterase and alcohol dehydrogenase activity in skin. Metabolism of retinyl palmitate to retinol (vitamin A) during percutaneous absorption. Pharm Res. 1994; 11:1155–1159. PMID: 7971717.3. Serri R, Iorizzo M. Cosmeceuticals: focus on topical retinoids in photoaging. Clin Dermatol. 2008; 26:633–635. PMID: 18940544.
Article4. Guenin EP, Zatz JL. Skin permeation of retinyl palmitate from vescicles. J Soc Cosmet Chem. 1995; 46:261–270.5. Sorg O, Tran C, Carraux P, Grand D, Hügin A, Didierjean L, Saurat JH. Spectral properties of topical retinoids prevent DNA damage and apoptosis after acute UV-B exposure in hairless mice. Photochem Photobiol. 2005; 81:830–836. PMID: 15760277.
Article6. Antille C, Tran C, Sorg O, Carraux P, Didierjean L, Saurat JH. Vitamin A exerts a photoprotective action in skin by absorbing ultraviolet B radiation. J Invest Dermatol. 2003; 121:1163–1167. PMID: 14708621.
Article7. Idson B. Vitamins in cosmetics, an update I. overview and vitamin A. Drug Cosmet Ind. 1990; 146:26–91.8. Ihara H, Hashizume N, Hirase N, Suzue R. Esterification makes retinol more labile to photolysis. J Nutr Sci Vitaminol (Tokyo). 1999; 45:353–358. PMID: 10524354.
Article9. Carlotti ME, Rossatto V, Gallarate M, Trotta M, Debernardi F. Vitamin A palmitate photostability and stability over time. J Cosmet Sci. 2004; 55:233–252. PMID: 15264052.
Article10. Carlotti ME, Rossatto V, Gallarate M. Vitamin A and vitamin A palmitate stability over time and under UVA and UVB radiation. Int J Pharm. 2002; 240:85–94. PMID: 12062504.
Article11. Ihara H, Hashizume N, Hirase N, Suzue R. Esterification makes retinol more labile to photolysis. J Nutr Sci Vitaminol (Tokyo). 1999; 45:353–358. PMID: 10524354.
Article12. Sane A, Limtrakul J. Formation of retinyl palmitate-loaded poly(l-lactide) nanoparticles using rapid expansion of supercritical solutions into liquid solvents (RESOLV). J Supercrit Fluid. 2009; 51:230–237.
Article13. Carlotti ME, Rossatto V, Gallarate M, Trotta M, Debernardi F. Vitamin A palmitate photostability and stability over time. J Cosmet Sci. 2004; 55:233–252. PMID: 15264052.
Article14. Maxwell EG, Belshaw NJ, Waldron KW, Morris VJ. Pectin -An emerging new bioactive food polysaccharide. Trends Food Sci Tech. 2011; 24:64–73.15. Wong TW, Colombo G, Sonvico F. Pectin matrix as oral drug delivery vehicle for colon cancer treatment. AAPS PharmSciTech. 2011; 12:201–214. PMID: 21194013.
Article16. Yamaguchi T, Takamura H, Matoba T, Terao J. HPLC method for evaluation of the free radical-scavenging activity of foods by using 1,1-diphenyl-2-picrylhydrazyl. Biosci Biotechnol Biochem. 1998; 62:1201–1204. PMID: 9692204.
Article17. Rice Evans CA, Miller NJ, Paganga G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997; 2:152–159.18. Rice-Evans CA, Miller NJ, Paganga G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic Biol Med. 1996; 20:933–956. PMID: 8743980.
Article19. Denès JM, Baron A, Renard CM, Péan C, Drilleau JF. Different action patterns for apple pectin methylesterase at pH 7.0 and 4.5. Carbohydr Res. 2000; 327:385–393. PMID: 10990023.
Article20. García-Ochoa F, Santos VE, Casas JA, Gómez E. Xanthangum: production, recovery, and properties. Biotechnol Adv. 2000; 18:549–579. PMID: 14538095.21. Fatimi A, Tassin JF, Quillard S, Axelos MA, Weiss P. The rheological properties of silated hydroxypropylmethylcellulose tissue engineering matrices. Biomaterials. 2008; 29:533–543. PMID: 17996292.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Enhanced In Vitro Skin Deposition Properties of Retinyl Palmitate through Its Stabilization by Pectin
- Pectin Micro- and Nano-capsules of Retinyl Palmitate as Cosmeceutical Carriers for Stabilized Skin Transport
- Suppressive effects of pectin on colitis-associated colon carcinogenesis
- A Case of Ileus in a Patient with Schizophrenia Under Paliperidone Palmitate Treatment
- Differential Effects of Palmitate and Docosahexaenoic acid on ATP-sensitive K+ Channel Activity of Pancreatic beta-cells