J Korean Med Sci.
2013 Feb;28(2):220-226. 10.3346/jkms.2013.28.2.220.
Hematologic Recovery after Tandem High-Dose Chemotherapy and Autologous Stem Cell Transplantation in Children with High-Risk Solid Tumors
- Affiliations
-
- 1Department of Pediatrics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. kwsped@skku.edu
- 2Department of Laboratory Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- KMID: 1429184
- DOI: http://doi.org/10.3346/jkms.2013.28.2.220
Abstract
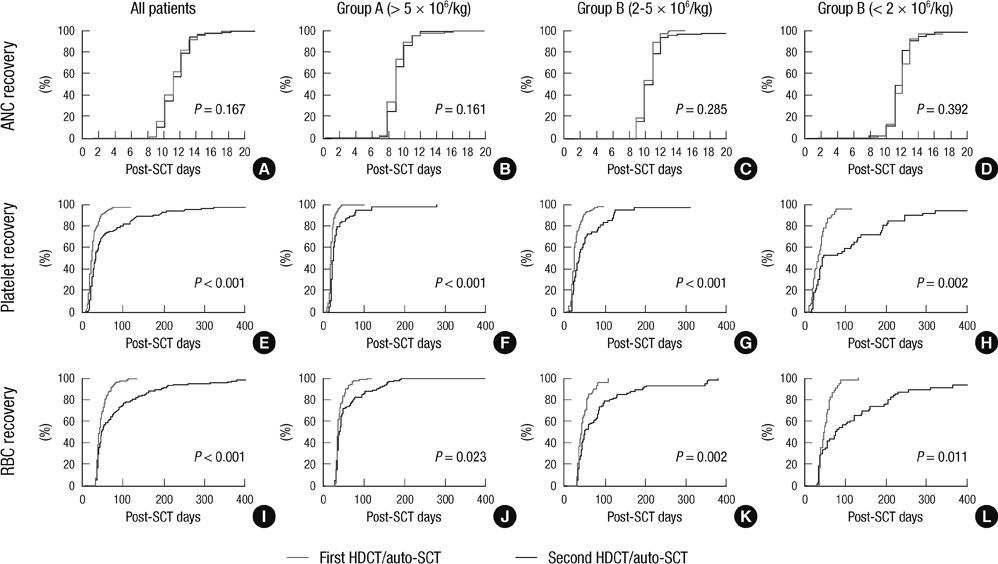
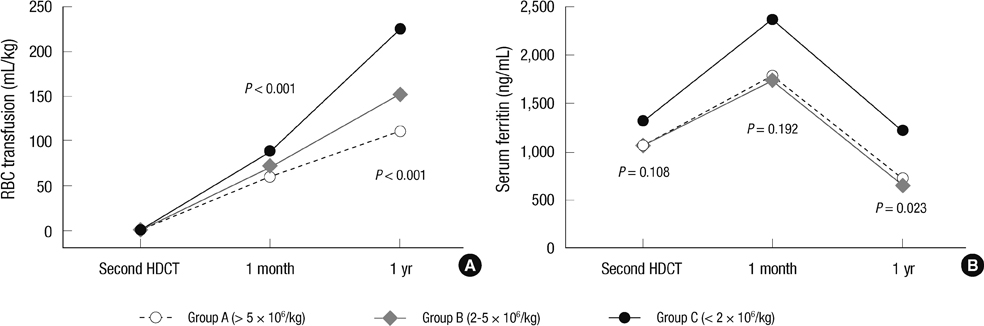
- Although the number of studies using tandem high-dose chemotherapy and autologous stem cell transplantation (HDCT/autoSCT) for the treatment of high-risk pediatric solid tumors has been increasing, documentation of hematologic recovery after tandem HDCT/autoSCT is very limited. For this reason, we retrospectively analyzed the hematologic recovery of 236 children with high-risk solid tumors who underwent tandem HDCT/autoSCT. The median numbers of CD34+ cells transplanted during the first and second HDCT/autoSCT were 4.3 x 10(6)/kg (range 0.6-220.2) and 4.1 x 10(6)/kg (range 0.9-157.6), respectively (P = 0.664). While there was no difference in neutrophil recovery between the first and second HDCT/autoSCT, platelet and RBC recoveries were significantly delayed in the second HDCT/autoSCT (P < 0.001 and P < 0.001, respectively). Delayed recovery in the second HDCT/autoSCT was more prominent when the number of transplanted CD34+ cells was lower, especially if it was < 2 x 10(6)/kg. A lower CD34+ cell count was also associated with increased RBC transfusion requirements and a higher serum ferritin level after tandem HDCT/autoSCT. More CD34+ cells need to be transplanted during the second HDCT/autoSCT in order to achieve the same hematologic recovery as the first HDCT/autoSCT.
Keyword
MeSH Terms
-
Adolescent
Antigens, CD34/metabolism
Antineoplastic Combined Chemotherapy Protocols/*therapeutic use
Blood Cell Count
Blood Platelets/cytology
Child
Child, Preschool
Combined Modality Therapy
Erythrocytes/cytology
Female
Ferritins/blood
Humans
Infant
Male
Neoplasms/*drug therapy
Neutrophils/cytology
Retrospective Studies
*Stem Cell Transplantation
Stem Cells/cytology/metabolism
Transplantation, Autologous
Young Adult
Antigens, CD34
Ferritins
Figure
Reference
-
1. Matthay KK, Villablanca JG, Seeger RC, Stram DO, Harris RE, Ramsay NK, Swift P, Shimada H, Black CT, Brodeur GM, et al. Children's Cancer Group. Treatment of high-risk neuroblastoma with intensive chemotherapy, radiotherapy, autologous bone marrow transplantation, and 13-cis-retinoic acid. N Engl J Med. 1999. 341:1165–1173.2. Berthold F, Boos J, Burdach S, Erttmann R, Henze G, Hermann J, Klingebiel T, Kremens B, Schilling FH, Schrappe M, et al. Myeloablative megatherapy with autologous stem-cell rescue versus oral maintenance chemotherapy as consolidation treatment in patients with high-risk neuroblastoma: a randomized controlled trial. Lancet Oncol. 2005. 6:649–658.3. Pritchard J, Cotterill SJ, Germond SM, Imeson J, de Kraker J, Jones DR. High dose melphalan in the treatment of advanced neuroblastoma: results of a randomised trial (ENSG-1) by the European Neuroblastoma Study Group. Pediatr Blood Cancer. 2005. 44:348–357.4. George RE, Li S, Medeiros-Nancarrow C, Neuberg D, Marcus K, Shamberger RC, Pulsipher M, Grupp SA, Diller L. High-risk neuroblastoma treated with tandem autologous peripheral-blood stem cell-supported transplantation: long-term survival update. J Clin Oncol. 2006. 24:2891–2896.5. Kletzel M, Katzenstein HM, Haut PR, Yu AL, Morgan E, Reynolds M, Geissler G, Marymount MH, Liu D, Kalapurakal JA, et al. Treatment of high-risk neuroblastoma with triple-tandem high-dose therapy and stem-cell rescue: results of the Chicago Pilot II Study. J Clin Oncol. 2002. 20:2284–2292.6. Gajjar A, Chintagumpala M, Ashley D, Kellie S, Kun LE, Merchant TE, Woo S, Wheeler G, Ahern V, Krasin MJ, et al. Risk-adapted craniospinal radiotherapy followed by high-dose chemotherapy and stem-cell rescue in children with newly diagnosed medulloblastoma (St Jude Medulloblastoma-96): long-term results from a prospective, multicentre trial. Lancet Oncol. 2006. 7:813–820.7. Sung KW, Lee SH, Yoo KH, Jung HL, Cho EJ, Koo HH, Lee SK, Kim J, Lim DH, Suh YL, et al. Tandem high-dose chemotherapy and autologous stem cell rescue in patients over 1 year of age with stage 4 neuroblastoma. Bone Marrow Transplant. 2007. 40:37–45.8. Sung KW, Yoo KH, Cho EJ, Koo HH, Lim DH, Shin HJ, Ahn SD, Ra YS, Choi ES, Ghim TT. High-dose chemotherapy and autologous stem cell rescue in children with newly diagnosed high-risk or relapsed medulloblastoma or supratentorial primitive neuroectodermal tumor. Pediatr Blood Cancer. 2007. 48:408–415.9. Banfi A, Bianchi G, Galotto M, Cancedda R, Quarto R. Bone marrow stromal damage after chemo/radiotherapy: occurrence, consequences and possibilities of treatment. Leuk Lymphoma. 2001. 42:863–870.10. Chueh HW, Sung KW, Lee SH, Yoo KH, Koo HH, Kim JY, Cho EJ. Iron chelation treatment with deferasirox prior to high-dose chemotherapy and autologous stem cell transplantation may reduce the risk of hepatic veno-occlusive disease in children with high-risk solid tumors. Pediatr Blood Cancer. 2011. 58:441–447.11. Siena S, Schiavo R, Pedrazzoli P, Carlo-Stella C. Therapeutic relevance off CD34 cell dose in blood cell transplantation for cancer therapy. J Clin Oncol. 2000. 18:1360–1377.12. Klaus J, Herrmann D, Breitkreutz I, Hegenbart U, Mazitschek U, Egerer G, Cremer FW, Lowenthal RM, Huesing J, Fruehauf S, et al. Effect of CD34 cell dose on hematopoietic reconstitution and outcome in 508 patients with multiple myeloma undergoing autologous peripheral blood stem cell transplantation. Eur J Haematol. 2007. 78:21–28.13. Mavroudis D, Read E, Cottler-Fox M, Couriel D, Molldrem J, Carter C, Yu M, Dunbar C, Barrett J. CD34+ cell dose predicts survival, posttransplant morbidity, and rate of hematologic recovery after allogeneic marrow transplants for hematologic malignancies. Blood. 1996. 88:3223–3229.14. DiPersio JF, Micallef IN, Stiff PJ, Bolwell BJ, Maziarz RT, Jacobsen E, Nademanee A, McCarty J, Bridger G, Calandra G. 3101 Investigators. Phase III prospective randomized double-blind placebo-controlled trial of plerixafor plus granulocyte colony-stimulating factor compared with placebo plus granulocyte colony-stimulating factor for autologous stem-cell mobilization and transplantation for patients with non-Hodgkin's lymphoma. J Clin Oncol. 2009. 27:4767–4773.15. Nademanee AP, Dipersio JF, Maziarz RT, Stadtmauer EA, Micallef IN, Stiff PJ, Hsu FJ, Bridger G, Bolwell BJ. Plerixafor plus granulocyte colony-stimulating factor versus placebo plus granulocyte colony-stimulating factor for mobilization of CD34+ hematopoietic stem cells in patients with multiple myeloma and low peripheral blood CD34+ cell count: results of a subset analysis of a randomized trial. Biol Blood Marrow Transplant. 2012. 18:1564–1572.16. Shander A, Cappellini MD, Goodnough LT. Iron overload and toxicity: the hidden risk of multiple blood transfusions. Vox Sang. 2009. 97:185–197.17. Fraga CG, Oteiza PI. Iron toxicity and antioxidant nutrients. Toxicology. 2002. 180:23–32.18. Borgna-Pignatti C, Cappellini MD, De Stefano P, Del Vecchio GC, Forni GL, Gamberini MR, Ghilardi R, Origa R, Piga A, Romeo MA, et al. Survival and complications in thalassemia. Ann N Y Acad Sci. 2005. 1054:40–47.19. Harmatz P, Butensky E, Quirolo K, Williams R, Ferrell L, Moyer T, Golden D, Neumayr L, Vichinsky E. Severity of iron overload in patients with sickle cell disease receiving chronic red blood cell transfusion therapy. Blood. 2000. 96:76–79.20. Fenaux P, Rose C. Impact of iron overload in myelodysplastic syndromes. Blood Rev. 2009. 23:S15–S19.21. Bae SJ, Kang C, Sung KW, Chueh HW, Son MH, Lee SH, Yoo KH, Koo HH. Iron overload during follow-up after tandem high-dose chemotherapy and autologous stem cell transplantation in patients with high-risk neuroblastoma. J Korean Med Sci. 2012. 27:363–369.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Factors affecting hematologic recovery and infection in high-dose chemotherapy and autologous stem cell transplantation in patients with high-risk solid tumor
- Tandem High-dose Chemotherapy and Autologous Stem Cell Transplantation in Children with Brain Tumors : Review of Single Center Experience
- Evaluation of Cardiac Function in Children with Solid Tumors, who Underwent Tandem High-Dose Chemotherapy and Autologous Stem Cell Transplantation
- A Case of Autologous Cord Blood Stem Cell Transplantationin Stage IV Neuroblastoma
- High-dose chemotherapy and autologous stem cell transplantation for pediatric brain tumors