Yonsei Med J.
2006 Oct;47(5):688-697. 10.3349/ymj.2006.47.5.688.
[6]-Gingerol Induces Cell Cycle Arrest and Cell Death of Mutant p53-expressing Pancreatic Cancer Cells
- Affiliations
-
- 1Brain Korea 21 Project for Medical Science, Yonsei University College of Medicine, Seoul, Korea.
- 2Internal Medicine and Institute of Gastroenterology, Yonsei University College of Medicine, Seoul, Korea. sysong@yumc.yonsei.ac.kr
- KMID: 1381250
- DOI: http://doi.org/10.3349/ymj.2006.47.5.688
Abstract
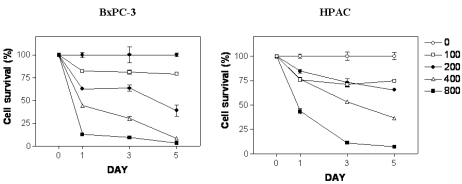
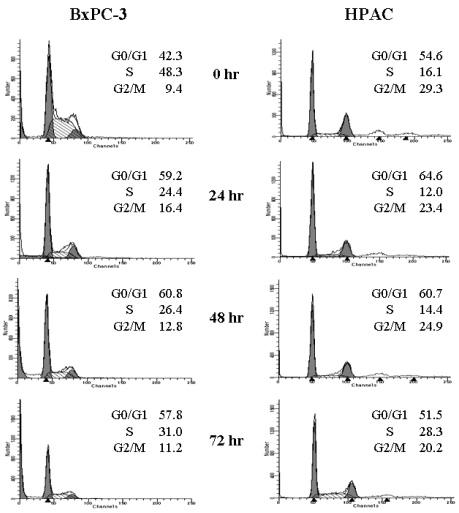
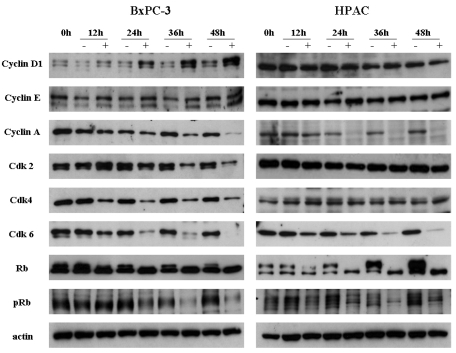
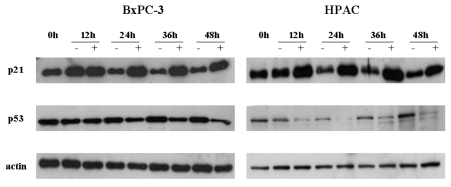
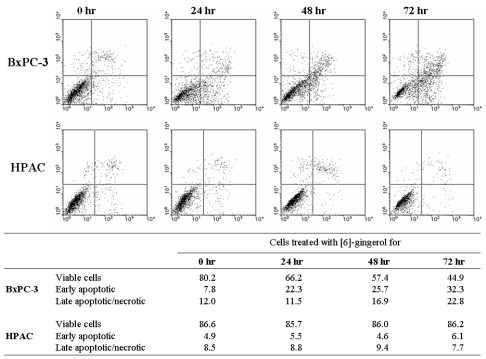
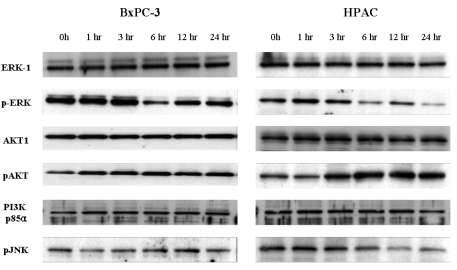
- [6]-Gingerol, a major phenolic compound derived from ginger, has anti-bacterial, anti-inflammatory and anti-tumor activities. While several molecular mechanisms have been described to underlie its effects on cells in vitro and in vivo, the underlying mechanisms by which [6]-gingerol exerts anti-tumorigenic effects are largely unknown. The purpose of this study was to investigate the action of [6]-gingerol on two human pancreatic cancer cell lines, HPAC expressing wild- type (wt) p53 and BxPC-3 expressing mutated p53. We found that [6]-gingerol inhibited the cell growth through cell cycle arrest at G1 phase in both cell lines. Western blot analyses indicated that [6]-gingerol decreased both Cyclin A and Cyclin-dependent kinase (Cdk) expression. These events led to reduction in Rb phosphorylation followed by blocking of S phase entry. p53 expression was decreased by [6]-gingerol treatment in both cell lines suggesting that the induction of Cyclin-dependent kinase inhibitor, p21(cip1), was p53-independent. [6]-Gingerol induced mostly apoptotic death in the mutant p53-expressing cells, while no signs of early apoptosis were detected in wild type p53-expressing cells and this was related to the increased phosphorylation of AKT. These results suggest that [6]-gingerol can circumvent the resistance of mutant p53- expressing cells towards chemotherapy by inducing apoptotic cell death while it exerts cytostatic effect on wild type p53- expressing cells by inducing temporal growth arrest.
Keyword
MeSH Terms
-
Tumor Suppressor Protein p53/*genetics/metabolism
Proto-Oncogene Proteins c-akt/genetics/metabolism
Pancreatic Neoplasms/*drug therapy
Mutation
Humans
Gene Expression Regulation, Neoplastic/drug effects
Fatty Alcohols/*pharmacology/therapeutic use
Drug Resistance, Neoplasm
Cell Proliferation/drug effects
Cell Line, Tumor
Cell Cycle/*drug effects
Apoptosis/*drug effects
Antineoplastic Agents/*pharmacology/therapeutic use
Figure
Cited by 1 articles
-
Prognostic Implications of Cyclin B1, p34cdc2, p27Kip1 and p53 Expression in Gastric Cancer
Dong-Hoon Kim
Yonsei Med J. 2007;48(4):694-700. doi: 10.3349/ymj.2007.48.4.694.
Reference
-
1. Ahmad N, Feyes DK, Nieminen AL, Agarwal R, Mukhtar H. Green tea constituent epigallocatechin-3-gallate and induction of apoptosis and cell cycle arrest in human carcinoma cells. J Natl Cancer Inst. 1997; 89:1881–1886. PMID: 9414176.
Article2. Liu XJ, Yang L, Mao YQ, Wang Q, Huang MH, Wang YP, et al. Effects of the tyrosine protein kinase inhibitor genistein on the proliferation, activation of cultured rat hepatic stellate cells. World J Gastroenterol. 2002; 8:739–745. PMID: 12174389.
Article3. Pan MH, Chen WJ, Lin-Shiau SY, Ho CT, Lin JK. Tangeretin induces cell-cycle G1 arrest through inhibiting Cyclin-dependent kinases 2 and 4 activities as well as elevating Cdk inhibitors p21 and p27 in human colorectal carcinoma cells. Carcinogenesis. 2002; 23:1677–1684. PMID: 12376477.
Article4. Agarwal R. Cell signaling and regulators of cell cycle as molecular targets for prostate cancer prevention by dietary agents. Biochem Pharmacol. 2000; 60:1051–1059. PMID: 11007941.
Article5. Bhatia N, Agarwal C, Agarwal R. Differential responses of skin cancer-chemopreventive agents silibinin, quercetin, and epigallocatechin 3-gallate on mitogenic signaling and cell cycle regulators in human epidermoid carcinoma A431 cells. Nutr Cancer. 2001; 39:292–299. PMID: 11759294.
Article6. Surh Y. Molecular mechanisms of chemopreventive effects of selected dietary and medicinal phenolic substances. Mutat Res. 1999; 428:305–327. PMID: 10518003.
Article7. Surh YJ, Lee E, Lee JM. Chemoprotective properties of some pungent ingredients present in red pepper and ginger. Mutat Res. 1998; 402:259–267. PMID: 9675305.
Article8. Suzuki F, Kobayashi M, Komatsu Y, Kato A, Pollard RB. Keishi-ka-kei-to, a traditional Chinese herbal medicine, inhibits pulmonary metastasis of B16 melanoma. Anticancer Res. 1997; 17:873–878. PMID: 9137420.9. Park KK, Chun KS, Lee JM, Lee SS, Surh YJ. Inhibitory effects of [6]-gingerol, a major pungent principle of ginger, on phorbol ester-induced inflammation, epidermal ornithine decarboxylase activity and skin tumor promotion in ICR mice. Cancer Lett. 1998; 129:139–144. PMID: 9719454.
Article10. Yoshimi N, Wang A, Morishita Y, Tanaka T, Sugie S, Kawai K, et al. Modifying effects of fungal and herb metabolites on azoxymethane-induced intestinal carcinogenesis in rats. Jpn J Cancer Res. 1992; 83:1273–1278. PMID: 1483942.
Article11. Bode AM, Ma WY, Surh YJ, Dong Z. Inhibition of epidermal growth factor-induced cell transformation and activator protein 1 activation by [6]-gingerol. Cancer Res. 2001; 61:850–853. PMID: 11221868.12. Lee E, Surh YJ. Induction of apoptosis in HL-60 cells by pungent vanilloids, [6]-gingerol and [6]-paradol. Cancer Lett. 1998; 134:163–168. PMID: 10025876.
Article13. Mahady GB, Pendland SL, Yun GS, Lu ZZ, Stoia A. Ginger (Zingiber officinale Roscoe) and the gingerols inhibit the growth of Cag A+ strains of Helicobacter pylori. Anticancer Res. 2003; 23:3699–3702. PMID: 14666666.14. Oya N. Chemoradiotherapy for pancreatic cancer: current status and perspectives. Int J Clin Oncol. 2004; 9:451–457. PMID: 15616875.
Article15. Ridwelski K, Meyer F. Current options for palliative treatment in patients with pancreatic cancer. Dig Dis. 2001; 19:63–75. PMID: 11385253.
Article16. Nio Y, Dong M, Uegaki K, Hirahara N, Minari Y, Sasaki S, et al. p53 expression affects the efficacy of adjuvant chemotherapy after resection of invasive ductal carcinoma of the pancreas. Anticancer Res. 1998; 18:3773–3779. PMID: 9854494.17. King TC, Estalilla OC, Safran H. Role of p53 and p16 gene alterations in determining response to concurrent paclitaxel and radiation in solid tumor. Semin Radiat Oncol. 1999; 9(2):Suppl 1. 4–11. PMID: 10210535.18. Xu ZW, Friess H, Buchler MW, Solioz M. Overexpression of Bax sensitizes human pancreatic cancer cells to apoptosis induced by chemotherapeutic agents. Cancer Chemother Pharmacol. 2002; 49:504–510. PMID: 12107556.
Article19. Song SY, Meszoely IM, Coffey RJ, Pietenpol JA, Leach SD. K-Ras-independent effects of the farnesyl transferase inhibitor L-744,832 on Cyclin B1/Cdc2 kinase activity, G2/M cell cycle progression and apoptosis in human pancreatic ductal adenocarcinoma cells. Neoplasia. 2000; 2:261–272. PMID: 10935512.20. Wallace-Brodeur RR, Lowe SW. Clinical implications of p53 mutations. Cell Mol Life Sci. 1999; 55:64–75. PMID: 10065152.
Article21. Natsugoe S, Nakashima S, Matsumoto M, Xiangming C, Okumura H, Kijima F, et al. Expression of p21 WAF1/Cip1 in the p53-dependent pathway is related to prognosis in patients with advanced esophageal carcinoma. Clin Cancer Res. 1999; 5:2445–2449. PMID: 10499617.22. Chang F, Lee JT, Navolanic PM, Steelman LS, Shelton JG, Blalock WL, et al. Involvement of PI3K/Akt pathway in cell cycle progression, apoptosis, and neoplastic transformation: a target for cancer chemotherapy. Leukemia. 2003; 17:590–603. PMID: 12646949.
Article23. Kim SO, Kundu JK, Shin YK, Park JH, Cho MH, Kim TY, et al. [6]-Gingerol inhibits COX-2 expression by blocking the activation of p38 MAP kinase and NF-kappaB in phorbol ester-stimulated mouse skin. Oncogene. 2005; 24:2558–2567. PMID: 15735738.24. Yang CS, Landau JM, Huang MT, Newmark HL. Inhibition of carcinogenesis by dietary polyphenolic compounds. Annu Rev Nutr. 2001; 21:381–406. PMID: 11375442.
Article25. Quelle DE, Ashmun RA, Shurtleff SA, Kato JY, Bar-Sagi D, Roussel MF, et al. Overexpression of mouse D-type Cyclins accelerates G1 phase in rodent fibroblasts. Genes Dev. 1993; 7:1559–1571. PMID: 8339933.
Article26. Kranenburg O, van der Eb AJ, Zantema A. Cyclin D1 is an essential mediator of apoptotic neuronal cell death. EMBO J. 1996; 15:46–54. PMID: 8598205.
Article27. Willis AC, Chen X. The promise and obstacle of p53 as a cancer therapeutic agent. Curr Mol Med. 2002; 2:329–345. PMID: 12108946.
Article28. Nelson WG, Kastan MB. DNA strand breaks: the DNA template alterations that trigger p53-dependent DNA damage response pathways. Mol Cell Biol. 1994; 14:1815–1823. PMID: 8114714.29. Levine AJ. p53, the cellular gatekeeper for growth and division. Cell. 1997; 88:323–331. PMID: 9039259.
Article30. Seeman S, Maurici D, Oliver M, de Formentel CC, Hainaut P. The tumor suppressor gene TP53: implications for cancer management and therpay. Crit Rev Clin Lab Sci. 2004; 41:551–583. PMID: 15603511.31. Sherr CJ, Roberts JM. CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev. 1999; 13:1501–1512. PMID: 10385618.
Article32. Coleman ML, Marshall CJ, Olson MF. Ras promotes p21(Waf1/Cip1) protein stability via a Cyclin D1-imposed block in proteasome-mediated degradation. EMBO J. 2003; 22:2036–2046. PMID: 12727871.
Article33. Olson MF, Paterson HF, Marshall CJ. Signals from Ras and Rho GTPases interact to regulate expression of p21Waf1/Cip1. Nature. 1998; 394:295–299. PMID: 9685162.
Article34. Liberto M, Cobrinik D. Growth factor-dependent induction of p21(CIP1) by the green tea polyphenol, epigallocatechin gallate. Cancer Lett. 2000; 154:151–161. PMID: 10806303.
Article35. Cho SG, Choi EJ. Apoptotic signaling pathways: caspases and stress-activated protein kinases. J Biochem Mol Biol. 2002; 35:24–27. PMID: 16248966.
Article36. Datta SR, Brunet A, Greenberg ME. Cellular survival: a play in three Akts. Genes Dev. 1999; 13:2905–2927. PMID: 10579998.
Article37. Li Q, Zhu GD. Targeting serine/threonine protein kinase B/Akt and cell-cycle checkpoint kinases for treating cancer. Curr Top Med Chem. 2002; 2:939–971. PMID: 12171565.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Molecular Aspects of Radiotherapy
- Effect of Adenovirus-p53 to Non-Small Cell Lung Cancer Cell Lines
- Transcription repression of a CCAAT-binding transcription factor CBF/HSP70 by p53
- p53 Prevents Immature Escaping from Cell Cycle G2 Checkpoint Arrest through Inhibiting cdk2-dependent NF-Y Phosphorylation
- Gemcitabine-induced Cell Death in Lung Cancer Cells: the Role of p53