Korean J Gastroenterol.
2011 Mar;57(3):173-179. 10.4166/kjg.2011.57.3.173.
Durability of Sustained Virologic Response in Chronic Hepatitis C: Analysis of Factors Related to Relapse after Sustained Virologic Response with Peginterferon Plus Ribavirin Combination Therapy
- Affiliations
-
- 1Department of Internal Medicine, WHO Collaborating Center on Viral Hepatitis, The Catholic University of Korea College of Medicine, Seoul, Korea. yoonsk@catholic.ac.kr
- 2Department of Internal Medicine, Soon Chun Hyang University College of Medicine, Seoul, Korea.
- KMID: 1128356
- DOI: http://doi.org/10.4166/kjg.2011.57.3.173
Abstract
- BACKGROUND/AIMS
Pegylated interferon plus ribavirin combination therapy has been the standard of therapy for patients with chronic hepatitis C. Although previous studies have reported long term durability after the sustained virologic response (SVR) with standard therapy for chronic hepatitis C, it is still unclear in Korea. The aim of this study was to evaluate the relapse rate and related factors after SVR to pegylated interferon therapy in Korean patients with chronic hepatitis C.
METHODS
A total of 119 chronic hepatitis C patients were treated with pegylated interferon plus ribavirin, and 73 patients achieved SVR (61.3%). Among 73 patients who achieved SVR, 68 patients (genotype 1, n=40; genotype non-1, n=28) were evaluated for virological response after SVR.
RESULTS
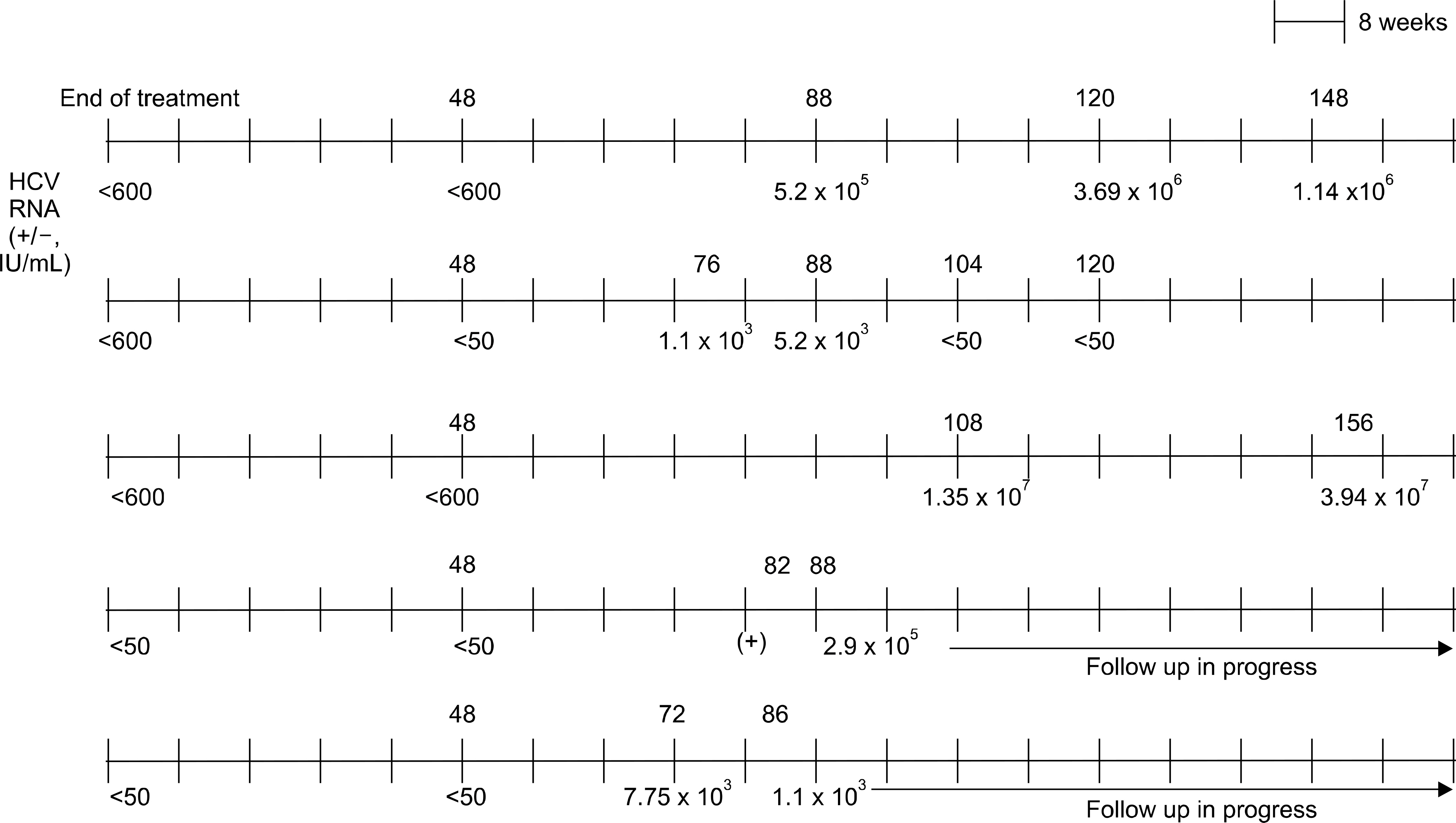
SVR rate in genotype 1 and genotype non-1 were 52.5%, and 65.1%, respectively. Relapse after SVR occurred in 5 patients (7.4%) with genotype 1, and the median time to relapse from SVR was 10 months. Univariate analysis revealed that the dose reduction of pegylated interferon (p=0.005) and cirrhosis (p=0.03) were significantly associated with relapse.
CONCLUSIONS
These results suggested that the relapse could occur even after SVR achievement in Korean patients with chronic hepatitis C, and the dose reduction of pegylated interferon during treatment or having cirrhosis may increased the risk for relapse.
MeSH Terms
-
Adult
Aged
Antiviral Agents/*therapeutic use
Drug Therapy, Combination
Female
Genotype
Hepatitis C, Chronic/complications/*drug therapy
Humans
Interferon Alfa-2a/*therapeutic use
Interferon Alfa-2b/*therapeutic use
Liver Cirrhosis/complications
Logistic Models
Male
Middle Aged
Polyethylene Glycols/*therapeutic use
RNA, Viral/blood
Recurrence
Ribavirin/*therapeutic use
Risk Factors
Figure
Reference
-
References
1. Armstrong GL, Wasley A, Simard EP, McQuillan GM, Kuhnert WL, Alter MJ. The prevalence of hepatitis C virus infection in the United States, 1999 through 2002. Ann Intern Med. 2006; 144:705–714.
Article2. Suh DJ, Jeong SH. Current status of hepatitis C virus infection in Korea. Intervirology. 2006; 49:70–75.
Article3. McHutchison JG, Gordon SC, Schiff ER, et al. Interferon alfa-2b alone or in combination with ribavirin as initial treatment for chronic hepatitis C. Hepatitis Interventional Therapy Group. N Engl J Med. 1998; 339:1485–1492.4. Poynard T, Marcellin P, Lee SS, et al. Randomised trial of interferon alpha2b plus ribavirin for 48 weeks or for 24 weeks versus interferon alpha2b plus placebo for 48 weeks for treatment of chronic infection with hepatitis C virus. International Hepatitis Interventional Therapy Group (IHIT). Lancet. 1998; 352:1426–1432.5. Fried MW, Shiffman ML, Reddy KR, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med. 2002; 347:975–982.
Article6. Desmond CP, Roberts SK, Dudley F, et al. Sustained virological response rates and durability of the response to interferon-based therapies in hepatitis C patients treated in the clinical setting. J Viral Hepat. 2006; 13:311–315.
Article7. Formann E, Steindl-Munda P, Hofer H, et al. Longterm follow-up of chronic hepatitis C patients with sustained virological response to various forms of interferon-based anti-viral therapy. Aliment Pharmacol Ther. 2006; 23:507–511.
Article8. Khokhar N. Late relapse in chronic hepatitis C after sustained viral response to interferon and ribavirin. J Gastroenterol Hepatol. 2004; 19:471–472.
Article9. Reichard O, Glaumann H, Frydén A, et al. Two-year biochemical, virological, and histological follow-up in patients with chronic hepatitis C responding in a sustained fashion to interferon al-fa-2b treatment. Hepatology. 1995; 21:918–922.
Article10. Tsuda N, Yuki N, Mochizuki K, et al. Longterm clinical and virological outcomes of chronic hepatitis C after successful interferon therapy. J Med Virol. 2004; 74:406–413.
Article11. Kim CH, Park BD, Lee JW, et al. Durability of a sustained virologic response in combination therapy with interferon/peginterferon and ribavirin for chronic hepatitis C. Korean J Hepatol. 2009; 15:70–79.
Article12. Rodriguez-Torres M, Jeffers LJ, Sheikh MY, et al. Peginterferon alfa-2a and ribavirin in Latino and non-Latino whites with hepatitis C. N Engl J Med. 2009; 360:257–267.
Article13. Missiha S, Heathcote J, Arenovich T, Khan K. Canadian Pegasys Expanded Access Group. Impact of asian race on response to combination therapy with peginterferon alfa-2a and ribavirin in chronic hepatitis C. Am J Gastroenterol. 2007; 102:2181–2188.
Article14. Pham TN, MacParland SA, Mulrooney PM, Cooksley H, Naoumov NV, Michalak TI. Hepatitis C virus persistence after spontaneous or treatmentinduced resolution of hepatitis C. J Virol. 2004; 78:5867–5874.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Durability of a sustained virologic response in patients with chronic hepatitis C treated with peginterferon and ribavirin
- The Combination Therapy with Peg-interferon Alfa and Ribavirin for Chronic Hepatitis C in Korea as an Initial Treatment
- Impact of adherence to peginterferon-ribavirin combination therapy in chronic hepatitis C patients on achieving a sustained virologic response
- Antiviral Therapy in Patients after Treatment for Hepatitis C-Related Hepatocellular Carcinoma
- Durability of a sustained virologic response in combination therapy with interferon/peginterferon and ribavirin for chronic hepatitis C