Korean J Lab Med.
2010 Feb;30(1):1-8. 10.3343/kjlm.2010.30.1.1.
Prognostic Significance of TEL/AML1 Rearrangement and Its Additional Genetic Changes in Korean Childhood Precursor B-Acute Lymphoblastic Leukemia
- Affiliations
-
- 1Department of Laboratory Medicine, University of Ulsan College of Medicine and Asan Medical Center, Seoul, Korea. ejseo@amc.seoul.kr
- 2Department of Pediatrics, University of Ulsan College of Medicine and Asan Medical Center, Seoul, Korea.
- KMID: 1096811
- DOI: http://doi.org/10.3343/kjlm.2010.30.1.1
Abstract
- BACKGROUND
TEL (ETV6)/AML1 (RUNX1) rearrangement is observed in approximately 20-25% of childhood precursor B-ALL and is associated with a favorable outcome. Additional genetic changes, associated with TEL/AML1, are frequently found. We evaluated the prevalence and prognostic significance of TEL/AML1 rearrangement and additional genetic changes in the TEL and AML1 genes in Korean childhood precursor B-ALL.
METHODS
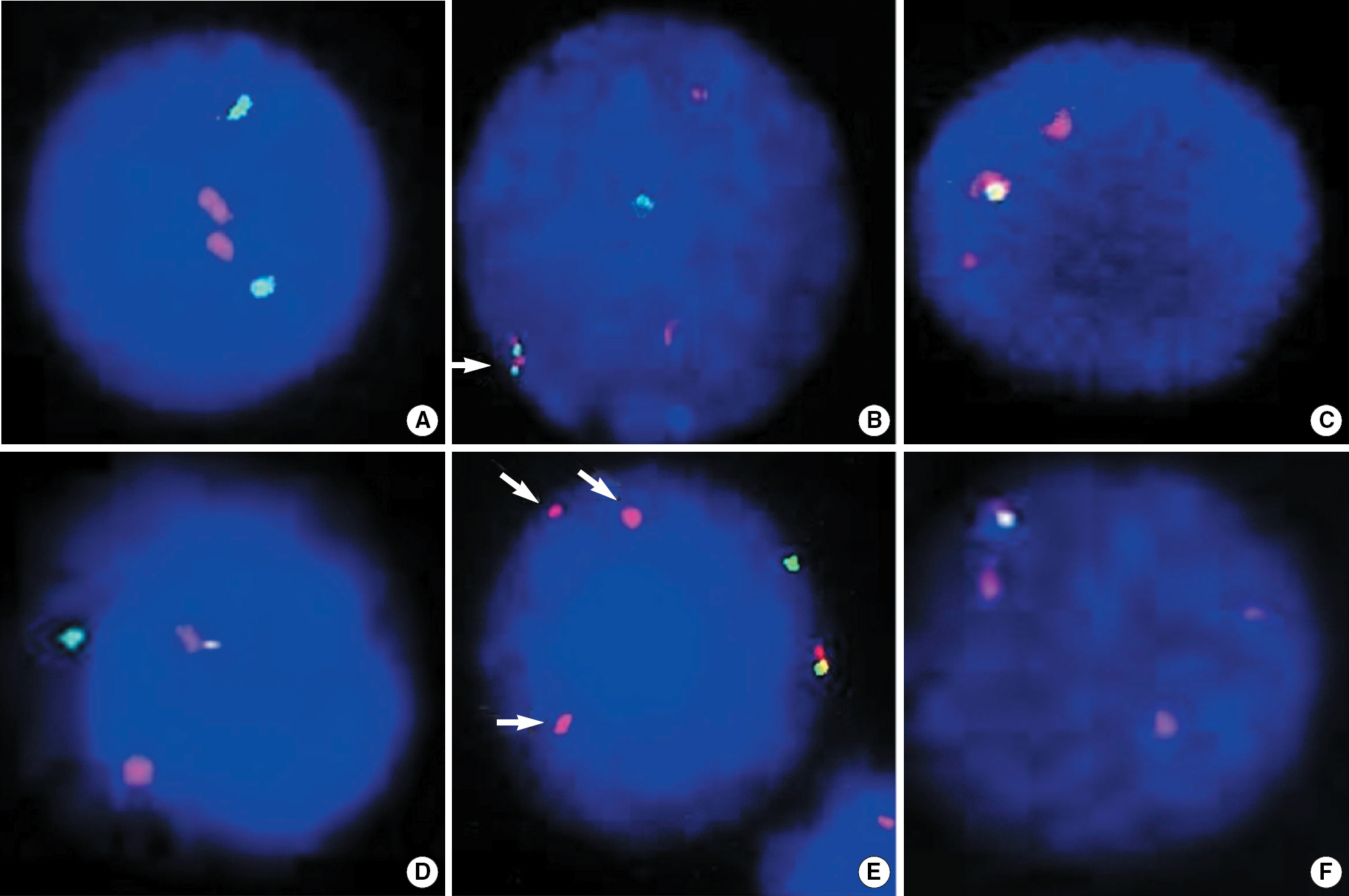
We performed FISH using LSITEL/AML1 ES probe (Vysis, USA) in 123 children diagnosed as having precursor B-ALL and assessed clinical relevance of the TEL/AML1 rearrangement and additional genetic abnormalities.
RESULTS
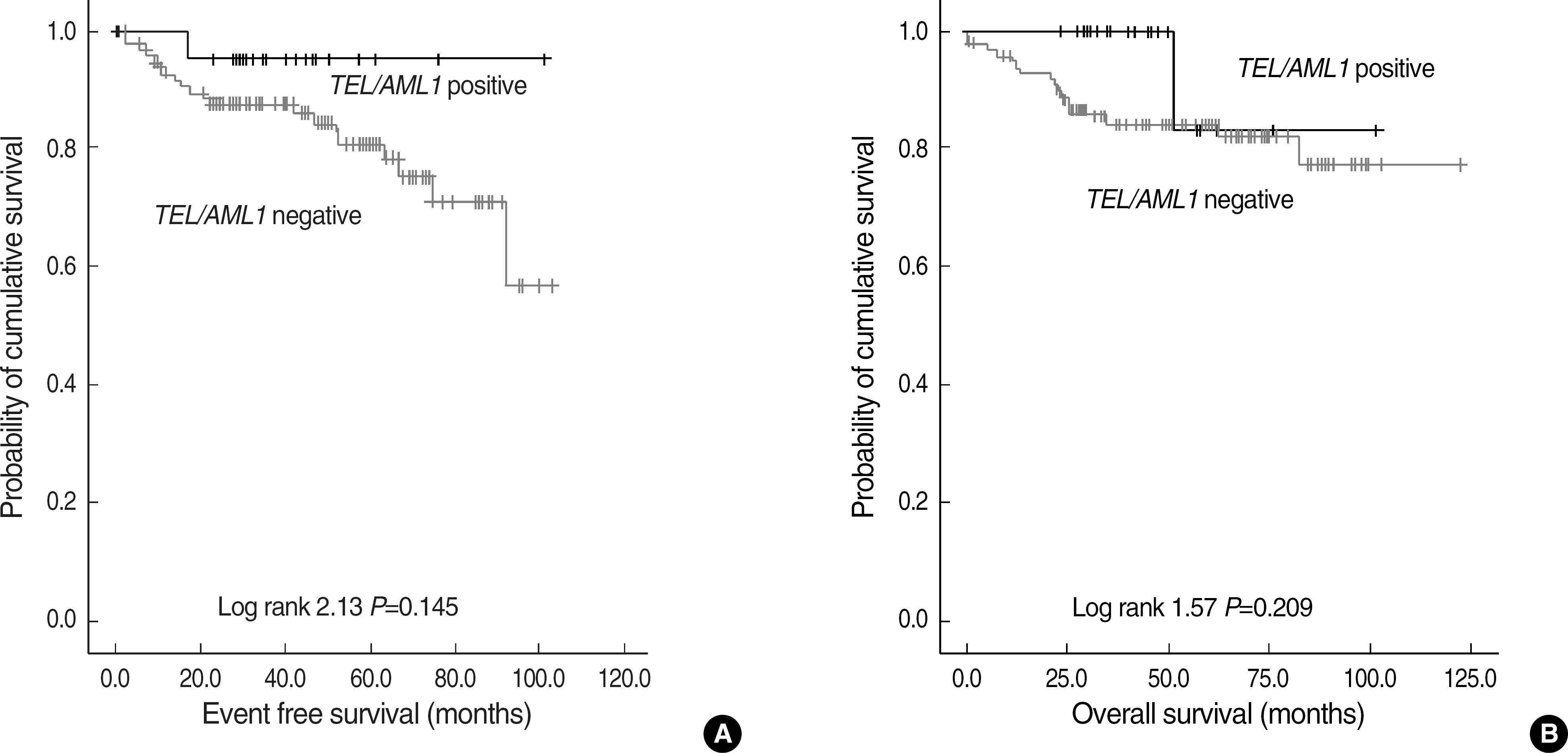
The frequency of TEL/AML1 was 17.1% (21/123) in patients with precursor B-ALL. TEL/ AML1-positive group showed male predominance (P=0.012) and younger age of onset than TEL/ AML1-negative group by 1.6 yr (P=0.013). The outcome of TEL/AML1-positive group tended to show lower incidences of relapse (1/21 vs 20/102), death (1/21 vs 17/102) and longer event free survival. Among TEL/AML1-positive patients, unrearranged TEL deletion, AML1 gain, and unrearranged TEL deletion combined with AML1 gain were detected in 61.9%, 23.8%, and 9.5%, respectively. There were no significant differences in the clinical features and outcome according to the presence or absence of additional genetic changes.
CONCLUSIONS
The frequency of TEL/AML1 and additional genetic changes in TEL and AML1 is higher than previous studies in Korean children, and in close agreement with usually reported one, 20-25%. TEL/AML1-positive group showed a tendency toward better prognosis. Further study is needed to clarify the prognostic significance of additional changes in TEL and AML1 based on a large sample size.
Keyword
MeSH Terms
-
Age Factors
Asian Continental Ancestry Group/*genetics
Child
Child, Preschool
Core Binding Factor Alpha 2 Subunit/*genetics
Disease-Free Survival
Female
Gene Deletion
Humans
In Situ Hybridization, Fluorescence
Karyotyping
Leukocyte Count
Male
Precursor B-Cell Lymphoblastic Leukemia-Lymphoma/*genetics/mortality
Prognosis
Proto-Oncogene Proteins c-ets/*genetics
Repressor Proteins/*genetics
Republic of Korea
Survival Rate
*Translocation, Genetic
Figure
Reference
-
1.Rubnitz JE., Look AT. Molecular genetics of childhood leukemias. J Pediatr Hematol Oncol. 1998. 20:1–11.
Article2.Kobayashi H., Satake N., Kaneko Y. Detection of the der (21)t(12;21) chromosome forming the TEL-AML1 fusion gene in childhood acute lymphoblastic leukemia. Leuk Lymphoma. 1997. 28:43–50.3.Jamil A., Theil KS., Kahwash S., Ruymann FB., Klopfenstein KJ. TEL/AML-1 fusion gene. its frequency and prognostic significance in childhood acute lymphoblastic leukemia. Cancer Genet Cytogenet. 2000. 122:73–8.4.Takahashi Y., Horibe K., Kiyoi H., Miyashita Y., Fukuda M., Mori H, et al. Prognostic significance of TEL/AML1 fusion transcript in childhood B-precursor acute lymphoblastic leukemia. J Pediatr Hematol Oncol. 1998. 20:190–5.5.Borowitz MJ., Rubnitz J., Nash M., Pullen DJ., Camitta B. Surface antigen phenotype can predict TEL-AML1 rearrangement in childhood B-precursor ALL: a Pediatric Oncology Group study. Leukemia. 1998. 12:1764–70.6.Zelent A., Greaves M., Enver T. Role of TEL-AML1 fusion gene in the molecular pathogenesis of childhood acute lymphoblastic leukaemia. Oncogene. 2004. 23:4275–83.7.Wiemels JL., Ford AM., Van Wering ER., Postma A., Greaves M. Protracted and variable latency of acute lymphoblastic leukemia after TEL-AML1 gene fusion in utero. Blood. 1999. 94:1057–62.8.Raynaud SD., Dastugue N., Zoccola D., Shurtleff SA., Mathew S., Raimondi SC. Cytogenetic abnormalities associated with the t(12;21): a collaborative study of 169 children with t(12;21)-positive acute lymphoblastic leukemia. Leukemia. 1999. 13:1325–30.
Article9.Loncarevic IF., Roitzheim B., Ritterbach J., Viehmann S., Borkhardt A., Lampert F, et al. Trisomy 21 is a recurrent secondary aberration in childhood acute lymphoblastic leukemia with TEL/AML1 gene fusion. Genes Chromosomes Cancer. 1999. 24:272–7.10.Stams WA., Beverloo HB., den Boer ML., de Menezes RX., Stigter RL., van Drunen E, et al. Incidence of additional genetic changes in the TEL and AML1 genes in DCOG and COALL-treated t(12;21)-positive pediatric ALL, and their relation with drug sensitivity and clinical outcome. Leukemia. 2006. 20:410–6.11.Al-Sweedan SA., Neglia JP., Steiner ME., Bostrom BC., Casey T., Hirsch BA. Characteristics of patients with TEL-AML1 -positive acute lymphoblastic leukemia with single or multiple fusions. Pediatr Blood Cancer. 2007. 48:510–4.12.Choi SJ., Chi HS., Park CJ., Seo EZ., Seo JJ., Ghim T, et al. TEL/AML1 fusion transcripts in childhood B-lineage acute lymphoblastic leukemia. Korean J Hematol. 2002. 37:169–76. (최수진, 지현숙, 서을주, 박찬정, 서종진, 김태형등. 소아B세포계급성림프구성백혈병에서의 TEL/AML1 재배열분석. 대한혈액학회지 2002;37:169-76.).13.Park KU., She CJ., Shin HY., Ahn HS., Kim CJ., Cho BK, et al. Low incidence of TEL/AML1 fusion and TEL deletion in Korean childhood acute leukemia by extra-signal fluorescence in situ hybridization. Cancer Genet Cytogenet. 2001. 126:73–7.14.McLean TW., Ringold S., Neuberg D., Stegmaier K., Tantravahi R., Ritz J, et al. TEL/AML-1 dimerizes and is associated with a favorable outcome in childhood acute lymphoblastic leukemia. Blood. 1996. 88:4252–8.15.Rubnitz JE., Downing JR., Pui CH., Shurtleff SA., Raimondi SC., Evans WE, et al. TEL gene rearrangement in acute lymphoblastic leukemia: a new genetic marker with prognostic significance. J Clin Oncol. 1997. 15:1150–7.16.Liang DC., Chou TB., Chen JS., Shurtleff SA., Rubnitz JE., Downing JR, et al. High incidence of TEL/AML1 fusion resulting from a cryptic t(12;21) in childhood B-lineage acute lymphoblastic leukemia in Taiwan. Leukemia. 1996. 10:991–3.17.Attarbaschi A., Mann G., König M., Dworzak MN., Trebo MM., Mühlegger N, et al. Incidence and relevance of secondary chromosome abnormalities in childhood TEL/AML1+ acute lymphoblastic leukemia: an interphase FISH analysis. Leukemia. 2004. 18:1611–6.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- TEL/AML1 Fusion Transcripts in Childhood B-Lineage Acute Lymphoblastic Leukemia
- Clinical Utility of Fluorescence in-situ Hybridization Profile Test in Detecting Genetic Aberrations in Acute Leukemia
- Molecular Cytogenetic Analysis of Gene Rearrangements in Childhood Acute Lymphoblastic Leukemia
- TEL(ETV 6)-AML1 Translocations with TEL and CDKN2 Inactivation in Acute Lymphoblastic Leukemia(ALL)
- Precursor B-Cell Acute Lymphoblastic Leukemia in Two Patients with a History of Cytotoxic Therapy