J Vet Sci.
2006 Dec;7(4):321-326. 10.4142/jvs.2006.7.4.321.
Cellular uptake of magnetic nanoparticle is mediated through energydependent endocytosis in A549 cells
- Affiliations
-
- 1Laboratory of Toxicology, College of Veterinary Medicine, Seoul National University, Seoul 151-742, Korea. mchotox@snu.ac.kr
- 2Materials Chemistry Lab, Seoul National University, Seoul 151-742, Korea.
- 3Laboratory of Molecular Oncology, Korea Institute of Radiological and Medical Sciences, Seoul 139-706, Korea.
- 4Polymeric and Soft Nanomaterials, Seoul National University, Seoul 151-742, Korea.
- 5Diversity Oriented Synthesis and Chemical Biology Lab, Seoul National University, Seoul 151-742, Korea.
- KMID: 1089477
- DOI: http://doi.org/10.4142/jvs.2006.7.4.321
Abstract
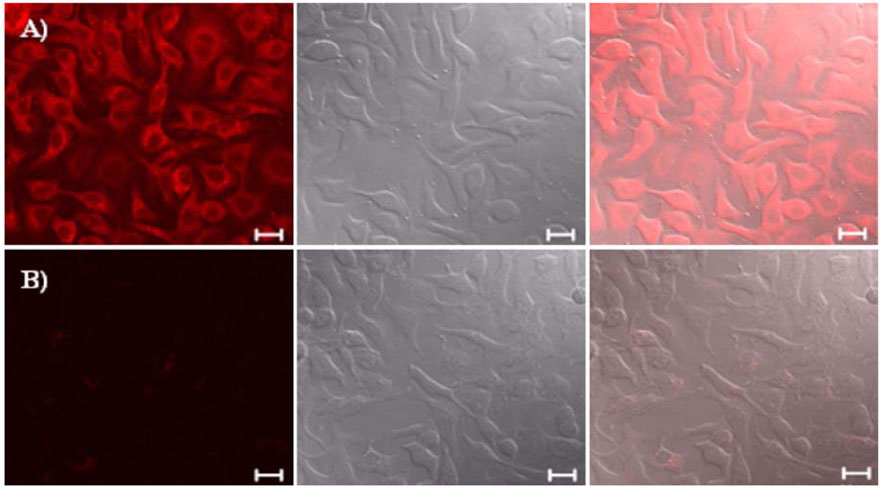
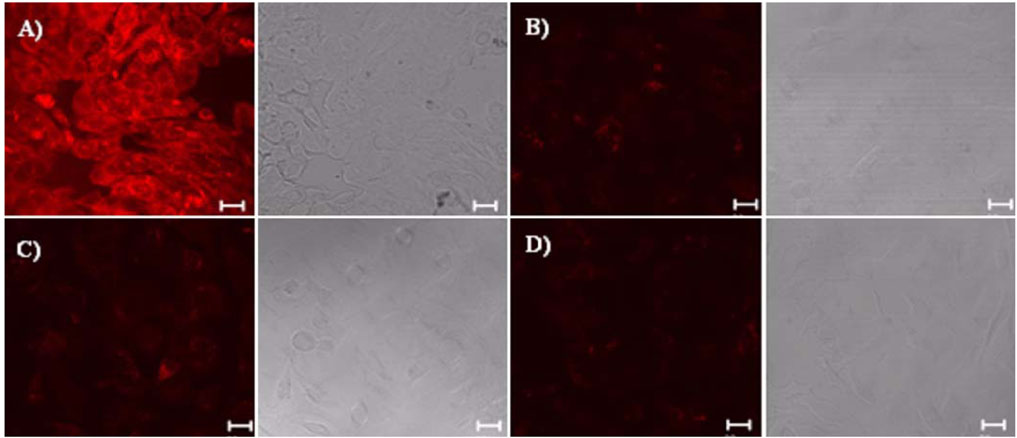
- Biocompatible silica-overcoated magnetic nanoparticles containing an organic fluorescence dye, rhodamine B isothiocyanate (RITC), within a silica shell [50 nm size, MNP@SiO2(RITC)s] were synthesized. For future application of the MNP@SiO2(RITC)s into diverse areas of research such as drug or gene delivery, bioimaging, and biosensors, detailed information of the cellular uptake process of the nanoparticles is essential. Thus, this study was performed to elucidate the precise mechanism by which the lung cancer cells uptake the magnetic nanoparticles. Lung cells were chosen for this study because inhalation is the most likely route of exposure and lung cancer cells were also found to uptake magnetic nanoparticles rapidly in preliminary experiments. The lung cells were pretreated with different metabolic inhibitors. Our results revealed that low temperature disturbed the uptake of magnetic nanoparticles into the cells. Metabolic inhibitors also prevented the delivery of the materials into cells. Use of TEM clearly demonstrated that uptake of the nanoparticles was mediated through endosomes. Taken together, our results demonstrate that magnetic nanoparticles can be internalized into the cells through an energy-dependent endosomal-lysosomal mechanism.
MeSH Terms
-
Biocompatible Materials/*pharmacokinetics
Cell Line, Tumor
Drug Delivery Systems/methods
Endocytosis/*physiology
Endosomes/physiology
Humans
Lung Neoplasms/drug therapy/*metabolism
Macrolides/pharmacology
Microscopy, Confocal
Microscopy, Electron, Transmission
Nanoparticles/*administration & dosage
Sodium Azide/pharmacology
Sucrose/pharmacology
Temperature
Figure
Cited by 1 articles
-
Trastuzumab-Conjugated Liposome-Coated Fluorescent Magnetic Nanoparticles to Target Breast Cancer
Mijung Jang, Young Il Yoon, Yong Soo Kwon, Tae-Jong Yoon, Hak Jong Lee, Sung Il Hwang, Bo La Yun, Sun Mi Kim
Korean J Radiol. 2014;15(4):411-422. doi: 10.3348/kjr.2014.15.4.411.
Reference
-
1. Barbé C, Bartlett J, Kong L, Finnie K, Lin HQ, Larkin M, Calleja S, Bush A, Calleja G. Silica particles: a novel drug-delivery system. Adv Mater. 2004. 16:1959–1966.
Article2. Chauhan SS, Liang XJ, Su AW, Pai-Panandiker A, Shen DW, Hanover JA, Gottesman MM. Reduced endocytosis and altered lysosome function in cisplatin-resistant cell lines. Br J Cancer. 2003. 88:1327–1334.
Article3. Gemeinhart RA, Luo D, Saltzman WM. Cellular fate of a modular DNA delivery system mediated by silica nanoparticles. Biotechnol Prog. 2005. 21:532–537.
Article4. Gupta AK, Berry C, Gupta M, Curtis A. Receptor-mediated targeting of magnetic nanoparticles using insulin as a surface ligand to prevent endocytosis. IEEE Trans Nanobioscience. 2003. 2:255–261.
Article5. Heuser JE, Anderson RGW. Hypertonic media inhibit receptor-mediated endocytosis by blocking clathrin-coated pit formation. J Cell Biol. 1989. 108:389–400.
Article6. Lu Y, McLellan J, Xia Y. Synthesis and crystallization of hybrid spherical colloids composed of polystyrene cores and silica shells. Langmuir. 2004. 20:3464–3470.
Article7. Nann T, Mulvaney P. Single quantum dots in spherical silica particles. Angew Chem Int Ed Engl. 2004. 43:5393–5396.
Article8. Nieland TJ, Ehrlich M, Krieger M, Kirchhausen T. Endocytosis is not required for the selective lipid uptake mediated by murine SR-BI. Biochim Biophys Acta. 2005. 1734:44–51.
Article9. Nishi T, Forgac M. The vacuolar (H+)-ATPases-nature's most versatile proton pumps. Nat Rev Mol Cell Biol. 2002. 3:94–103.
Article10. Ow H, Larson DR, Srivastava M, Baird BA, Webb WW, Wiesner U. Bright and stable core-shell fluorescent silica nanoparticles. Nano Lett. 2005. 5:113–117.
Article11. Pearse BMF, Crowther RA. Structure and assembly of coated vesicles. Annu Rev Biophys Biophys Chem. 1987. 16:49–68.
Article12. Rao GC, Kumar MS, Mathivanan N, Rao ME. Nanosuspensions as the most promising approach in nanoparticulate drug delivery systems. Pharmazie. 2004. 59:5–9.13. Robinson DG, Albrecht S, Moriysu Y. The V-ATPase inhibitors concanamycin A and bafilomycin A lead to Golgi swelling in tobacco BY-2 cells. Protoplasma. 2004. 224:255–260.
Article14. Shchipunov YA. Sol-gel-derived biomaterials of silica and carrageenans. J Colloid Interface Sci. 2003. 268:68–76.
Article15. Torchilin VP, Rammohan R, Weissig V, Levchenko TS. TAT peptide on the surface of liposomes affords their efficient intracellular delivery even at low temperature and in the presence of metabolic inhibitors. Proc Natl Acad Sci USA. 2001. 98:8786–8791.
Article16. Wieczorek H, Brown D, Grinstein S, Ehrenfeld J, Harvey WR. Animal plasma membrane energization by protonmotive V-ATPases. Bioassays. 1999. 21:637–648.
Article17. Yoon TJ, Kim JS, Kim BG, Yu KN, Cho MH, Lee JK. Multifunctional nanoparticles possessing a "magnetic motor effect" for drug or gene delivery. Angew Chem Int Ed Engl. 2005. 44:1068–1071.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Phagocytosis and Endocytosis of Silver Nanoparticles Induce Interleukin-8 Production in Human Macrophages
- Multifactorial Regulation of G Protein-Coupled Receptor Endocytosis
- Dynamin 2-mediated endocytosis of BLT1 is required for IL-8 production in HMC-1 cells induced by Trichomonas vaginalis-derived secretory products
- Versatile Functions of Caveolin-1 in Aging-related Diseases
- Comparative Uptake of Tc-99m Sestamibi and Tc-99m Tetrofosmin in Cancer Cells and Tissue Expressing P-Glycoprotein or Multidrug Resistance Associated Protein