J Vet Sci.
2011 Sep;12(3):243-249. 10.4142/jvs.2011.12.3.243.
Evaluation of glycoproteins purified from adult and larval camel ticks (Hyalomma dromedarii) as a candidate vaccine
- Affiliations
-
- 1Molecular Biology Department, Genetic Engineering and Biotechnology Division, National Research Centre, Dokki, Cairo, Egypt. yassershahein_nrc@yahoo.com
- 2Parasitology and Animal Diseases Department, Veterinary Research Division, National Research Centre, Dokki, Cairo, Egypt.
- 3Radiation Biology Department, National Centre for Radiation Research and Technology, Cairo, Egypt.
- KMID: 1067396
- DOI: http://doi.org/10.4142/jvs.2011.12.3.243
Abstract
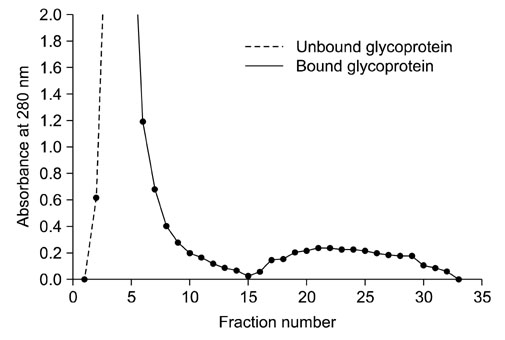
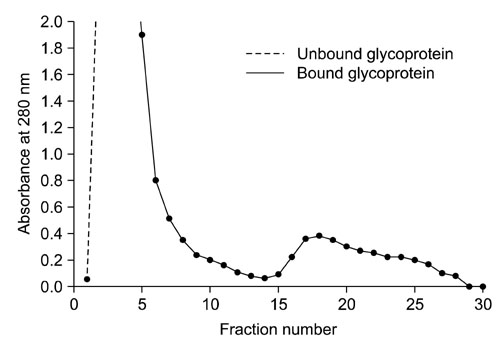
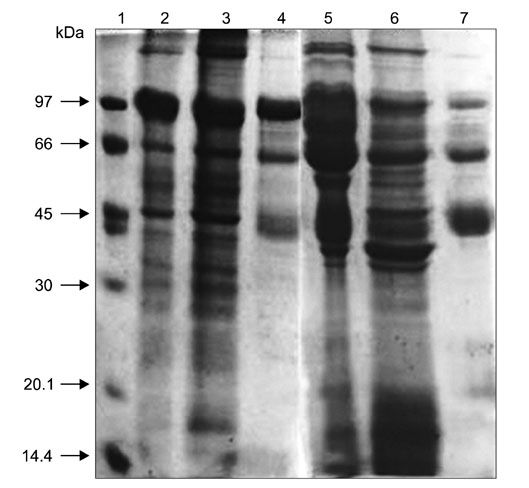
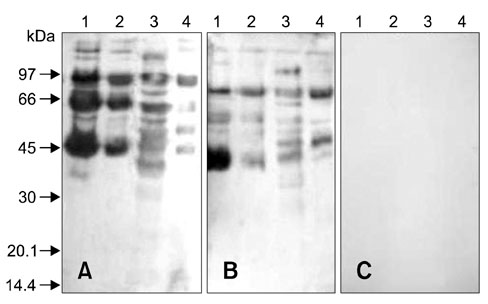
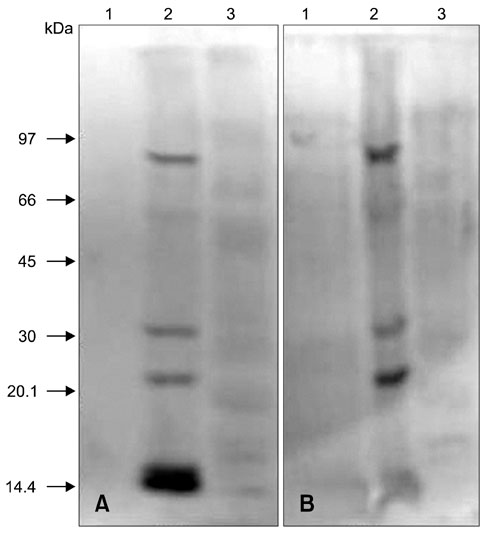
- In order to identify antigens that can help prevent camel tick infestations, three major glycoproteins (GLPs) about 97, 66 and 40 kDa in size were purified from adult and larval Egyptian ticks, Hyalomma (H.) dromedarii, using a single-step purification method with Con-A sepharose. The purified GLPs were evaluated as vaccines against camel tick infestation in rabbits. The rabbits received three intramuscular inoculations of GLPs (20 microg/animal) on days 0, 14, and 28. In the immunoblot analysis, Sera from the immunized rabbits recognized the native GLPs and other proteins from larval and adult H. dromedarii ticks along with those from other tick species such as Rhipicephalus sanguineus but not Ornithodoros moubata. The effects of immunity induced by these GLPs were determined by exposing rabbits to adult H. dromedarii ticks. These results demonstrated that GLP immunization led to a slightly decreased reproductive index and significantly reduced rates of egg hatchability. These results demonstrated that immunization with the purified GLPs can provide protection against infestation by H. dromedarii and some other tick species. Further studies are needed to confirm the effectiveness of immunization with GLPs against other tick species.
MeSH Terms
-
Animals
Antigens/immunology/isolation & purification
Argasidae/immunology
Chromatography, Affinity/veterinary
Electrophoresis, Polyacrylamide Gel/veterinary
Female
Glycoproteins/*immunology/isolation & purification
Immunoblotting/veterinary
Injections, Intramuscular/veterinary
Ixodidae/growth & development/*immunology
Life Cycle Stages
Male
Rabbits/*immunology/parasitology
Reproduction
Species Specificity
Tick Infestations/immunology/prevention & control/*veterinary
Figure
Reference
-
1. Abdel-Shafy S, Mahmoud MS, Abdel-Aziz MM. Evaluation of crude and fractionated gut extract antigens for protection against camel tick Hyalomma dromedarii (Acari: Ixodidae). J Entomol. 2008. 5:91–102.2. Allen JR, Humphreys SJ. Immunisation of guinea pigs and cattle against ticks. Nature. 1979. 280:491–493.
Article3. Anderson JF. The natural history of ticks. Med Clin North Am. 2002. 86:205–218.
Article4. Apanaskevich DA, Schuster AL, Horak IG. The Genus Hyalomma: VII. Redescription of all Parasitic Stages of H. (Euhyalomma) dromedarii and H. E. schulzei (Acari: Ixodidae). J Med Entomol. 2008. 45:817–831.5. Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976. 72:248–254.
Article6. Brown SJ, Askenase PW. Amblyomma americanum: physiochemical isolation of a protein derived from the tick salivary gland that is capable of inducing immune resistance in guinea pigs. Exp Parasitol. 1986. 62:40–50.
Article7. Canales M, Enríquez A, Ramos E, Cabrera D, Dandie H, Soto A, Falcón V, Rodríguez M, de la Fuente J. Large-scale production in Pichia pastoris of the recombinant vaccine Gavac against cattle tick. Vaccine. 1997. 15:414–422.
Article8. Das G, Ghosh S, Khan MH, Sharma JK. Immunization of cross-bred cattle against Hyalomma anatolicum anatolicum by purified antigens. Exp Appl Acarol. 2000. 24:645–659.9. de la Fuente J, Kocan KM. Strategies for development of vaccines for control of ixodid tick species. Parasite Immunol. 2006. 28:275–283.
Article10. Estrada-Peña A, Bouattour A, Camicas JL, Walker AR. Ticks of Domestic Animals in the Mediterranean Region. A Guide to Identification of Species. 2004. 1st ed. Zaragoza: University of Zaragoza;131.11. García-García JC, Montero C, Redondo M, Vargas M, Canales M, Boue O, Rodríguez M, Joglar M, Machado H, González IL, Valdés M, Méndez L, de la Fuente J. Control of ticks resistant to immunization with Bm86 in cattle vaccinated with the recombinant antigen Bm95 isolated from the cattle tick, Boophilus microplus. Vaccine. 2000. 18:2275–2287.
Article12. Ghosh S, Khan MH. Immunization of cattle against Hyalomma anatolicum anatolicum using larval antigens. Indian J Exp Biol. 1999. 37:203–205.13. Ghosh S, Khan MH, Ahmed N. Cross-bred cattle protected against Hyalomma anatolicum anatolicum by larval antigens purified by immunoaffinity chromatography. Trop Anim Health Prod. 1999. 31:263–273.14. Ghosh S, Singh NK, Das G. Assessment of duration of immunity in crossbred cattle immunized with glycoproteins isolated from Hyalomma anatolicum anatolicum and Boophilus microplus. Parasitol Res. 2005. 95:319–326.
Article15. Ghosh S, Azhahianambi P, Yadav MP. Upcoming and future strategies of tick control: a review. J Vector Borne Dis. 2007. 44:79–89.16. Hoogstraal H. African Ixodoidea. I. Ticks of the Sudan. 1956. Cairo: United State Naval Medical Research Unit No. 3;1101.17. Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970. 227:680–685.
Article18. Nolan J. Acaricide resistance in single and multi-host ticks and strategies for control. Parassitologia. 1990. 32:145–153.19. Patrick CD, Hair JA. Laboratory rearing procedures and equipment for multi-host ticks (Acarina: Ixodidae). J Med Entomol. 1975. 12:389–390.
Article20. Pegram RG, James AD, Oosterwijk GPM, Killorn KJ, Lemche J, Ghirotti M, Tekle Z, Chizyuka HGB, Mwase ET, Chizyuka F. Studies on the economics of ticks in Zambia. Exp Appl Acarol. 1991. 12:9–26.
Article21. Pruett JH. Immunological control of arthropod ectoparasites -a review. Int J Parasitol. 1999. 29:25–32.22. Randolph SE. Simpson SJ, Casas J, editors. Epidemiological consequences of the ecological physiology of ticks. Advances in Insect Physiology. Vol. 37. 2009. Amsterdam: Elsevier;297–339.23. Sharma JK, Ghosh S, Khan MH, Das G. Immunoprotective efficacy of a purified 39 kDa nymphal antigen of Hyalomma anatolicum anatolicum. Trop Anim Health Prod. 2001. 33:103–116.24. Singh NK, Ghosh S. Experimental immunisation of crossbred cattle with glycoproteins isolated from the larvae of Hyalomma anatolicum anatolicum and Boophilus microplus. Exp Appl Acarol. 2003. 31:297–314.
Article25. Szabó MPJ, Bechara GH. Immunisation of dogs and guinea pigs against Rhipicephalus sanguineus ticks using gut extract. Vet Parasitol. 1997. 68:283–294.
Article26. Towbin H, Staehelin T, Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA. 1979. 76:4350–4354.
Article27. Willadsen P, Riding GA, McKenna RV, Kemp DH, Tellam RL, Nielsen JN, Lahnstein J, Cobon GS, Gough JM. Immunologic control of a parasitic arthropod. Identification of a protective antigen from Boophilus microplus. J Immunol. 1989. 143:1346–1351.28. Willadsen P. Novel vaccines for ectoparasites. Vet Parasitol. 1997. 71:209–222.
Article29. Willadsen P. The molecular revolution in the development of vaccines against ectoparasites. Vet Parasitol. 2001. 101:353–368.
Article30. Wong JYM, Opdebeeck JP. Larval membrane antigens protect Hereford cattle against infestation with Boophilus microplus. Parasite Immunol. 1990. 12:75–83.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Prevalence of ixodid ticks on cattle in Mazandaran province, Iran
- Immunization effect of recombinant P27/30 protein expressed in Escherichia coli against the hard tick Haemaphysalis longicornis (Acari: Ixodidae) in rabbits
- Immunization of mice with recombinant P27/30 protein confers protection against hard tick Haemaphysalis longicornis (Acari: Ixodidae) infestation
- Purification and characterization of two larval glycoproteins from the cattle tick, Boophilus annulatus
- Morphological Characteristics of Normal and Gynandromorphic Hyalomma asiaticum Schulze and Schlottke, 1930