Ann Pediatr Endocrinol Metab.
2024 Aug;29(4):242-249. 10.6065/apem.2346180.090.
Long-term tracking of glycosylated hemoglobin levels across the lifespan in type 1 diabetes: from infants to young adults
- Affiliations
-
- 1Department of Pediatrics, Severance Children's Hospital, Endocrine Research Institute, Yonsei University College of Medicine, Seoul, Korea
- 2Dr Kwon's Growth Clinic, Seoul, Korea
- KMID: 2559452
- DOI: http://doi.org/10.6065/apem.2346180.090
Abstract
- Purpose
Glycosylated hemoglobin (HbA1c) is commonly used as a monitoring tool in diabetes. Due to the potential influence of insulin resistance (IR), HbA1c level may fluctuate over a person's lifetime. This study explores the long-term tracking of HbA1c level in individuals diagnosed with type 1 diabetes mellitus (T1DM) from infancy to early adulthood.
Methods
The HbA1c levels in 275 individuals (121 males, 43.8%) diagnosed with T1DM were tracked for an average of 9.4 years. The distribution of HbA1c levels was evaluated according to age with subgroups divided by gender, use of continuous glucose monitoring (CGM), and the presence of complications.
Results
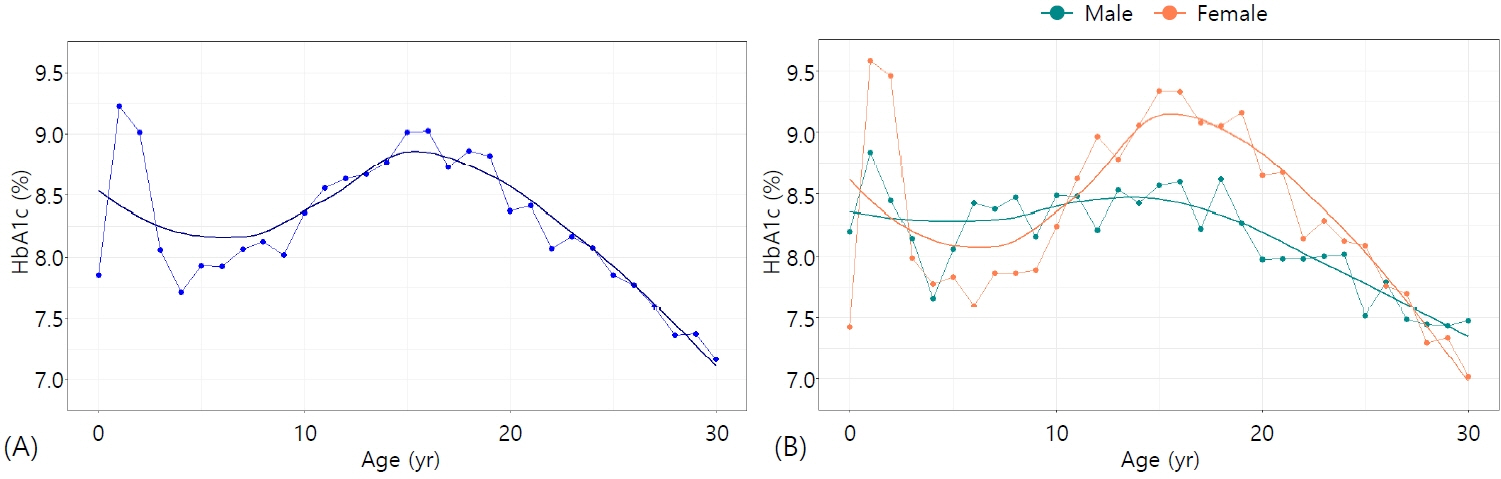
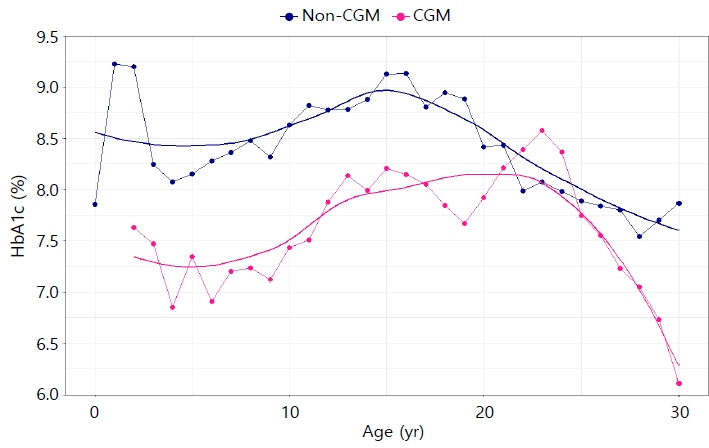
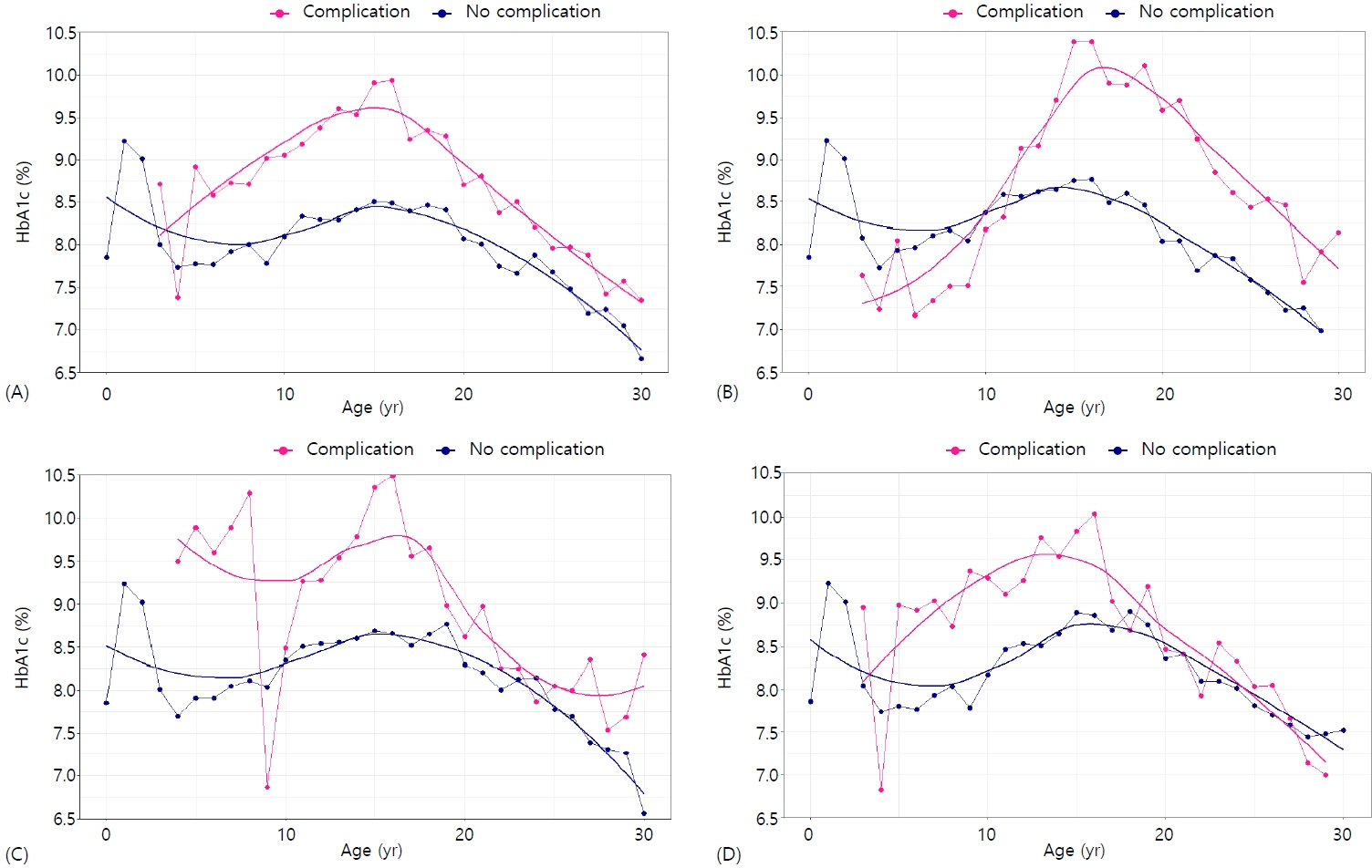
HbA1c levels were highest at the age of 1 year and then declined until age 4, followed by a significant increase, reaching a maximum at ages 15–16 years. The levels subsequently gradually decreased until early adulthood. This pattern was observed in both sexes, but it was more pronounced in females. Additionally, HbA1c levels were higher in CGM nonusers compared with CGM users; however, regardless of CGM usage, an age-dependent pattern was observed. Furthermore, diabetic complications occurred in 26.8% of individuals, and the age-dependent pattern was observed irrespective of diabetic complications, although HbA1c levels were higher in individuals with diabetic complications.
Conclusion
HbA1c levels vary throughout the lifespan, with higher levels during adolescence. This trend is observed regardless of sex and CGM usage, potentially due to physiological IR observed during adolescence. Hence, physiological IR should be considered when interpretating HbA1c levels during adolescence.
Keyword
Figure
Reference
-
References
1. Mayer-Davis EJ, Lawrence JM, Dabelea D, Divers J, Isom S, Dolan L, et al. Incidence trends of type 1 and type 2 diabetes among youths, 2002-2012. N Engl J Med. 2017; 376:1419–29.
Article2. Silverstein J, Klingensmith G, Copeland K, Plotnick L, Kaufman F, Laffel L, et al. Care of children and adolescents with type 1 diabetes: a statement of the American Diabetes Association. Diabetes Care. 2005; 28:186–212.3. Secrest AM, Becker DJ, Kelsey SF, Laporte RE, Orchard TJ. Characterizing sudden death and dead-in-bed syndrome in Type 1 diabetes: analysis from two childhood-onset type 1 diabetes registries. Diabet Med. 2011; 28:293–300.
Article4. Stettler C, Allemann S, Jüni P, Cull CA, Holman RR, Egger M, et al. Glycemic control and macrovascular disease in types 1 and 2 diabetes mellitus: meta-analysis of randomized trials. Am Heart J. 2006; 152:27–38.
Article5. American Diabetes Association. (2) Classification and diagnosis of diabetes. Diabetes Care. 2015; 38 Suppl:S8–16.6. Brown A, Reynolds LR, Bruemmer D. Intensive glycemic control and cardiovascular disease: an update. Nat Rev Cardiol. 2010; 7:369–75.
Article7. American Diabetes Association Professional Practice Committee. 6. Glycemic targets: standards of medical care in diabetes-2022. Diabetes Care. 2022; 45(Suppl 1):S83–96.8. Gerstl EM, Rabl W, Rosenbauer J, Gröbe H, Hofer SE, Krause U, et al. Metabolic control as reflected by HbA1c in children, adolescents and young adults with type-1 diabetes mellitus: combined longitudinal analysis including 27,035 patients from 207 centers in Germany and Austria during the last decade. Eur J Pediatr. 2008; 167:447–53.
Article9. Guercio G, Rivarola MA, Chaler E, Maceiras M, Belgorosky A. Relationship between the growth hormone/insulin-like growth factor-I axis, insulin sensitivity, and adrenal androgens in normal prepubertal and pubertal girls. J Clin Endocrinol Metab. 2003; 88:1389–93.10. Cook JS, Hoffman RP, Stene MA, Hansen JR. Effects of maturational stage on insulin sensitivity during puberty. J Clin Endocrinol Metab. 1993; 77:725–30.
Article11. Roemmich JN, Clark PA, Lusk M, Friel A, Weltman A, Epstein LH, et al. Pubertal alterations in growth and body composition. VI. Pubertal insulin resistance: relation to adiposity, body fat distribution and hormone release. Int J Obes Relat Metab Disord. 2002; 26:701–9.
Article12. Amiel SA, Sherwin RS, Simonson DC, Lauritano AA, Tamborlane WV. Impaired insulin action in puberty. A contributing factor to poor glycemic control in adolescents with diabetes. N Engl J Med. 1986; 315:215–9.13. Ball GD, Huang TT, Gower BA, Cruz ML, Shaibi GQ, Weigensberg MJ, et al. Longitudinal changes in insulin sensitivity, insulin secretion, and beta-cell function during puberty. J Pediatr. 2006; 148:16–22.
Article14. Jeffery AN, Metcalf BS, Hosking J, Streeter AJ, Voss LD, Wilkin TJ. Age before stage: insulin resistance rises before the onset of puberty: a 9-year longitudinal study (EarlyBird 26). Diabetes Care. 2012; 35:536–41.15. Lee JM, Okumura MJ, Davis MM, Herman WH, Gurney JG. Prevalence and determinants of insulin resistance among U.S. adolescents: a population-based study. Diabetes Care. 2006; 29:2427–32.16. Peplies J, Jiménez-Pavón D, Savva SC, Buck C, Günther K, Fraterman A, et al. Percentiles of fasting serum insulin, glucose, HbA1c and HOMA-IR in pre-pubertal normal weight European children from the IDEFICS cohort. Int J Obes (Lond). 2014; 38 Suppl 2:S39–47.17. Greenbaum CJ. Insulin resistance in type 1 diabetes. Diabetes Metab Res Rev. 2002; 18:192–200.
Article18. Rahmati M, Keshvari M, Mirnasuri S, Yon DK, Lee SW, Il Shin J, et al. The global impact of COVID-19 pandemic on the incidence of pediatric new-onset type 1 diabetes and ketoacidosis: a systematic review and meta-analysis. J Med Virol. 2022; 94:5112–27.
Article19. Shashaj B, Luciano R, Contoli B, Morino GS, Spreghini MR, Rustico C, et al. Reference ranges of HOMA-IR in normal-weight and obese young Caucasians. Acta Diabetol. 2016; 53:251–60.20. Almeida CA, Pinho AP, Ricco RG, Pepato MT, Brunetti IL. Determination of glycemia and insulinemia and the homeostasis model assessment (HOMA) in schoolchildren and adolescents with normal body mass index. J Pediatr (Rio J). 2008; 84:136–40.
Article21. Hovestadt I, Kiess W, Lewien C, Willenberg A, Poulain T, Meigen C, et al. HbA1c percentiles and the association between BMI, age, gender, puberty, and HbA1c levels in healthy German children and adolescents. Pediatr Diabetes. 2022; 23:194–202.
Article22. Gordon CM, Mansfield MJ. Changing needs of the patient with diabetes mellitus during the teenage years. Curr Opin Pediatr. 1996; 8:319–27.
Article23. Pinhas-Hamiel O, Hamiel U, Boyko V, Graph-Barel C, Reichman B, Lerner-Geva L. Trajectories of HbA1c levels in children and youth with type 1 diabetes. PLoS One. 2014; 9:e109109.
Article24. Hoffman RP, Vicini P, Sivitz WI, Cobelli C. Pubertal adolescent male-female differences in insulin sensitivity and glucose effectiveness determined by the one compartment minimal model. Pediatr Res. 2000; 48:384–8.
Article25. Moran A, Jacobs DR Jr, Steinberger J, Hong CP, Prineas R, Luepker R, et al. Insulin resistance during puberty: results from clamp studies in 357 children. Diabetes. 1999; 48:2039–44.
Article26. Kim S, Song K, Lee M, Suh J, Chae HW, Kim HS, et al. Trends in HOMA-IR values among South Korean adolescents from 2007-2010 to 2019-2020: a sex-, age-, and weight status-specific analysis. Int J Obes (Lond). 2023; 47:865–72.
Article27. Travers SH, Jeffers BW, Bloch CA, Hill JO, Eckel RH. Gender and Tanner stage differences in body composition and insulin sensitivity in early pubertal children. J Clin Endocrinol Metab. 1995; 80:172–8.
Article28. Foste r NC, Beck RW, Miller KM, Clements MA, Rickels MR, DiMeglio LA, et al. State of type 1 diabetes management and outcomes from the T1D exchange in 2016-2018. Diabetes Technol Ther. 2019; 21:66–72.
Article29. Teo E, Hassan N, Tam W, Koh S. Effectiveness of continuous glucose monitoring in maintaining glycaemic control among people with type 1 diabetes mellitus: a systematic review of randomised controlled trials and meta-analysis. Diabetologia. 2022; 65:604–19.
Article30. Lin R, Brown F, James S, Jones J, Ekinci E. Continuous glucose monitoring: a review of the evidence in type 1 and 2 diabetes mellitus. Diabet Med. 2021; 38:e14528.
Article31. Hermanns N, Heinemann L, Freckmann G, Waldenmaier D, Ehrmann D. Impact of CGM on the management of hypoglycemia problems: overview and secondary analysis of the HypoDE study. J Diabetes Sci Technol. 2019; 13:636–44.
Article32. Lockhart MJ, Smith D. Should continuous glucose monitoring systems be offered to all patients with type 1 diabetes mellitus? Ir J Med Sci. 2022; 191:957–60.
Article33. Hovind P, Tarnow L, Rossing P, Jensen BR, Graae M, Torp I, et al. Predictors for the development of microalbuminuria and macroalbuminuria in patients with type 1 diabetes: inception cohort study. BMJ. 2004; 328:1105.
Article34. Diabetes Control and Complications Trial Research Group. Effect of intensive diabetes treatment on the development and progression of long-term complications in adolescents with insulin-dependent diabetes mellitus: Diabetes Control and Complications Trial. J Pediatr. 1994; 125:177–88.35. Morales A. A better future for children with type 1 diabetes: review of the conclusions from the Diabetes Control and Complications Trial and the Epidemiology of Diabetes Interventions and Complications study. J Ark Med Soc. 2009; 106:90–3.36. Burger W, Hövener G, Düsterhus R, Hartmann R, Weber B. Prevalence and development of retinopathy in children and adolescents with type 1 (insulin-dependent) diabetes mellitus. A longitudinal study. Diabetologia. 1986; 29:17–22.
Article37. Cho YH, Craig ME, Donaghue KC. Puberty as an accelerator for diabetes complications. Pediatr Diabetes. 2014; 15:18–26.
Article38. Danne T, Kordonouri O, Enders I, Hövener G, Weber B. Factors modifying the effect of hyperglycemia on the development of retinopathy in adolescents with diabetes. Results of the Berlin Retinopathy Study. Horm Res. 1998; 50 Suppl 1:28–32.
Article39. Nordwall M, Fredriksson M, Ludvigsson J, Arnqvist HJ. Impact of age of onset, puberty, and glycemic control followed from diagnosis on incidence of retinopathy in type 1 diabetes: the VISS study. Diabetes Care. 2019; 42:609–16.
Article40. Sosenko JM, Miettinen OS, Williamson JR, Gabbay KH. Muscle capillary basement-membrane thickness and longterm glycemia in type I diabetes mellitus. N Engl J Med. 1984; 311:694–8.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Combination of Fasting Plasma Glucose and Glycosylated Hemoglobin as a Predictor for Type 2 Diabetes in Korean Adults (Korean Diabetes J 33(4):306-314, 2009)
- The Combination of Fasting Plasma Glucose and Glycosylated Hemoglobin as a Predictor for Type 2 Diabetes in Korean Adults (Korean Diabetes J 33(4):306-314, 2009)
- Clinical usefulness of the measurement of serum fructosamine in childhood diabetes mellitus
- Letter: The Association of Serum Cystatin C with Glycosylated Hemoglobin in Korean Adults (Diabetes Metab J 2016;40:62-9)
- Cystatin C as a Predictor for Diabetes according to Glycosylated Hemoglobin Levels in Korean Patients