Diabetes Metab J.
2024 May;48(3):354-372. 10.4093/dmj.2023.0277.
Glucagon-Like Peptide-1: New Regulator in Lipid Metabolism
- Affiliations
-
- 1Department of Endocrinology, Affiliated Hospital of Jiangsu University, Zhenjiang, China
- KMID: 2555767
- DOI: http://doi.org/10.4093/dmj.2023.0277
Abstract
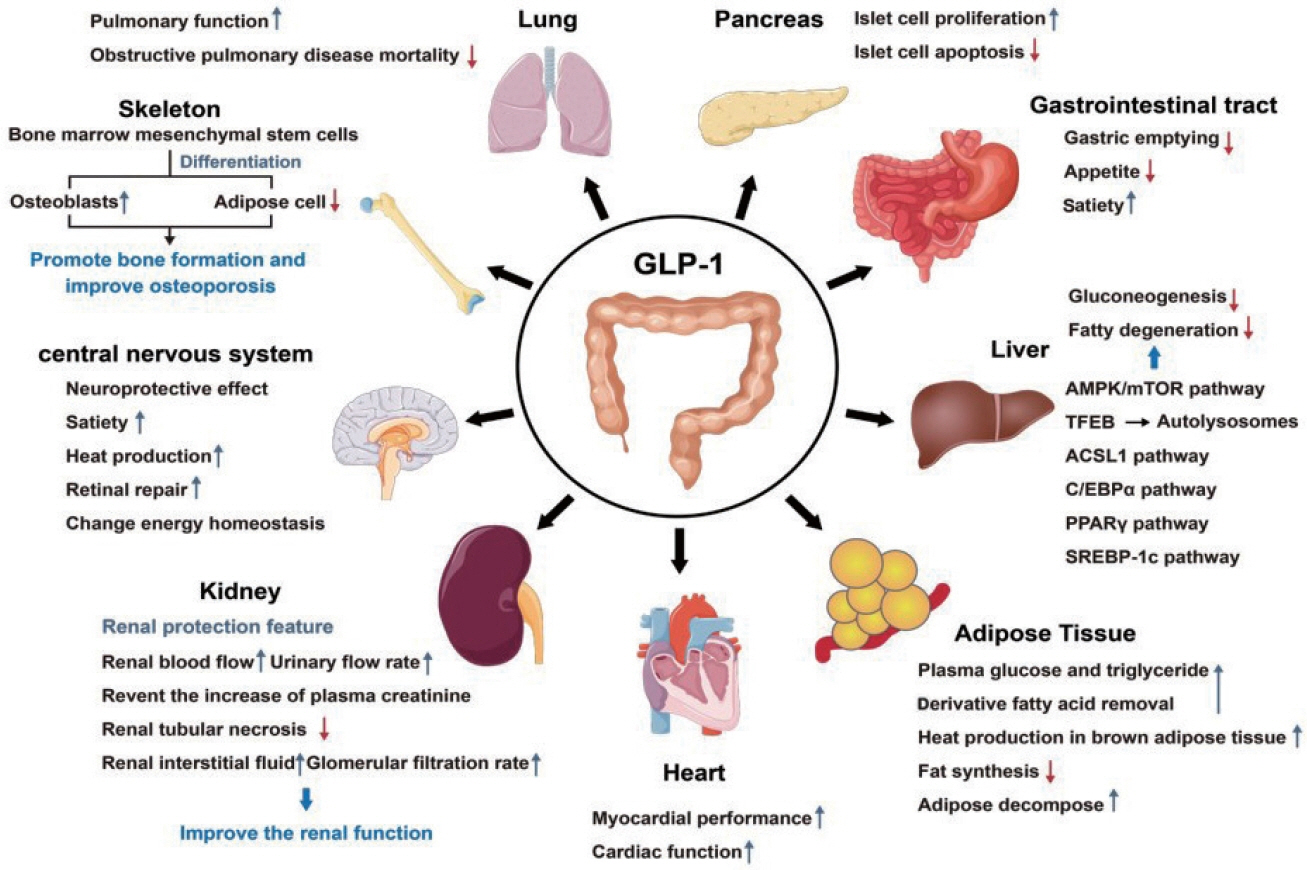
- Glucagon-like peptide-1 (GLP-1) is a 30-amino acid peptide hormone that is mainly expressed in the intestine and hypothalamus. In recent years, basic and clinical studies have shown that GLP-1 is closely related to lipid metabolism, and it can participate in lipid metabolism by inhibiting fat synthesis, promoting fat differentiation, enhancing cholesterol metabolism, and promoting adipose browning. GLP-1 plays a key role in the occurrence and development of metabolic diseases such as obesity, nonalcoholic fatty liver disease, and atherosclerosis by regulating lipid metabolism. It is expected to become a new target for the treatment of metabolic disorders. The effects of GLP-1 and dual agonists on lipid metabolism also provide a more complete treatment plan for metabolic diseases. This article reviews the recent research progress of GLP-1 in lipid metabolism.
Keyword
Figure
Reference
-
1. Holst JJ. The physiology of glucagon-like peptide 1. Physiol Rev. 2007; 87:1409–39.
Article2. Kimball CP, Murlin JR. Aqueous extracts of pancreas. III. Some precipitation reactions of insulin. J Biol Chem. 1923; 58:337–46.3. Bell GI, Sanchez-Pescador R, Laybourn PJ, Najarian RC. Exon duplication and divergence in the human preproglucagon gene. Nature. 1983; 304:368–71.
Article4. Kreymann B, Williams G, Ghatei MA, Bloom SR. Glucagon-like peptide-1 7-36: a physiological incretin in man. Lancet. 1987; 2:1300–4.
Article5. Mehdi SF, Pusapati S, Anwar MS, Lohana D, Kumar P, Nandula SA, et al. Glucagon-like peptide-1: a multi-faceted antiinflammatory agent. Front Immunol. 2023; 14:1148209.
Article6. Gentilella R, Pechtner V, Corcos A, Consoli A. Glucagon-like peptide-1 receptor agonists in type 2 diabetes treatment: are they all the same? Diabetes Metab Res Rev. 2019; 35:e3070.
Article7. Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet. 2006; 368:1696–705.
Article8. Rowlands J, Heng J, Newsholme P, Carlessi R. Pleiotropic effects of GLP-1 and analogs on cell signaling, metabolism, and function. Front Endocrinol (Lausanne). 2018; 9:672.
Article9. Campos RV, Lee YC, Drucker DJ. Divergent tissue-specific and developmental expression of receptors for glucagon and glucagon-like peptide-1 in the mouse. Endocrinology. 1994; 134:2156–64.
Article10. Shin S, Le Lay J, Everett LJ, Gupta R, Rafiq K, Kaestner KH. CREB mediates the insulinotropic and anti-apoptotic effects of GLP-1 signaling in adult mouse β-cells. Mol Metab. 2014; 3:803–12.
Article11. Madsbad S. Review of head-to-head comparisons of glucagon-like peptide-1 receptor agonists. Diabetes Obes Metab. 2016; 18:317–32.
Article12. Yang H, Li H, Wang Z, Shi Y, Jiang G, Zeng F. Exendin-4 ameliorates renal ischemia-reperfusion injury in the rat. J Surg Res. 2013; 185:825–32.
Article13. Jensen EP, Poulsen SS, Kissow H, Holstein-Rathlou NH, Deacon CF, Jensen BL, et al. Activation of GLP-1 receptors on vascular smooth muscle cells reduces the autoregulatory response in afferent arterioles and increases renal blood flow. Am J Physiol Renal Physiol. 2015; 308:F867–77.
Article14. Muskiet MH, Tonneijck L, Smits MM, van Baar MJ, Kramer MH, Hoorn EJ, et al. GLP-1 and the kidney: from physiology to pharmacology and outcomes in diabetes. Nat Rev Nephrol. 2017; 13:605–28.
Article15. Meng J, Ma X, Wang N, Jia M, Bi L, Wang Y, et al. Activation of GLP-1 receptor promotes bone marrow stromal cell osteogenic differentiation through β-catenin. Stem Cell Reports. 2016; 6:579–91.
Article16. Imbernon M, Saponaro C, Helms HC, Duquenne M, Fernandois D, Deligia E, et al. Tanycytes control hypothalamic liraglutide uptake and its anti-obesity actions. Cell Metab. 2022; 34:1054–63.
Article17. Yoon G, Kim YK, Song J. Glucagon-like peptide-1 suppresses neuroinflammation and improves neural structure. Pharmacol Res. 2020; 152:104615.
Article18. Lee SJ, Sanchez-Watts G, Krieger JP, Pignalosa A, Norell PN, Cortella A, et al. Loss of dorsomedial hypothalamic GLP-1 signaling reduces BAT thermogenesis and increases adiposity. Mol Metab. 2018; 11:33–46.
Article19. Zhang T, Ruan HZ, Wang YC, Shao YQ, Zhou W, Weng SJ, et al. Signaling mechanism for modulation by GLP-1 and exendin-4 of GABA receptors on rat retinal ganglion cells. Neurosci Bull. 2022; 38:622–36.
Article20. Kabahizi A, Wallace B, Lieu L, Chau D, Dong Y, Hwang ES, et al. Glucagon-like peptide-1 (GLP-1) signalling in the brain: from neural circuits and metabolism to therapeutics. Br J Pharmacol. 2022; 179:600–24.
Article21. Ussher JR, Baggio LL, Campbell JE, Mulvihill EE, Kim M, Kabir MG, et al. Inactivation of the cardiomyocyte glucagon-like peptide-1 receptor (GLP-1R) unmasks cardiomyocyte-independent GLP-1R-mediated cardioprotection. Mol Metab. 2014; 3:507–17.
Article22. Ban K, Kim KH, Cho CK, Sauve M, Diamandis EP, Backx PH, et al. Glucagon-like peptide (GLP)-1(9-36)amide-mediated cytoprotection is blocked by exendin(9-39) yet does not require the known GLP-1 receptor. Endocrinology. 2010; 151:1520–31.
Article23. Ban K, Noyan-Ashraf MH, Hoefer J, Bolz SS, Drucker DJ, Husain M. Cardioprotective and vasodilatory actions of glucagon-like peptide 1 receptor are mediated through both glucagon-like peptide 1 receptor-dependent and -independent pathways. Circulation. 2008; 117:2340–50.
Article24. Giblett JP, Clarke SJ, Dutka DP, Hoole SP. Glucagon-like peptide-1: a promising agent for cardioprotection during myocardial ischemia. JACC Basic Transl Sci. 2016; 1:267–76.25. Meloni AR, DeYoung MB, Lowe C, Parkes DG. GLP-1 receptor activated insulin secretion from pancreatic β-cells: mechanism and glucose dependence. Diabetes Obes Metab. 2013; 15:15–27.
Article26. Ding WG, Gromada J. Protein kinase A-dependent stimulation of exocytosis in mouse pancreatic beta-cells by glucosedependent insulinotropic polypeptide. Diabetes. 1997; 46:615–21.
Article27. Skelin M, Rupnik M. cAMP increases the sensitivity of exocytosis to Ca2+ primarily through protein kinase A in mouse pancreatic beta cells. Cell Calcium. 2011; 49:89–99.
Article28. Wagner S, Brierley DI, Leeson-Payne A, Jiang W, Chianese R, Lam BY, et al. Obesity medication lorcaserin activates brainstem GLP-1 neurons to reduce food intake and augments GLP-1 receptor agonist induced appetite suppression. Mol Metab. 2023; 68:101665.
Article29. Vargas EJ, Rizk M, Gomez-Villa J, Edwards PK, Jaruvongvanich V, Storm AC, et al. Effect of endoscopic sleeve gastroplasty on gastric emptying, motility and hormones: a comparative prospective study. Gut. 2023; 72:1073–80.
Article30. Sannaa W, Dilmaghani S, BouSaba J, Maselli D, Atieh J, Eckert D, et al. Factors associated with successful weight loss after liraglutide treatment for obesity. Diabetes Obes Metab. 2023; 25:377–86.
Article31. Cantini G, Mannucci E, Luconi M. Perspectives in GLP-1 research: new targets, new receptors. Trends Endocrinol Metab. 2016; 27:427–38.
Article32. Perez-Garcia A, Hurtado-Carneiro V, Herrero-De-Dios C, Dongil P, Garcia-Maurino JE, Sanchez MD, et al. Storage and utilization of glycogen by mouse liver during adaptation to nutritional changes are GLP-1 and PASK dependent. Nutrients. 2021; 13:2552.
Article33. He Q, Sha S, Sun L, Zhang J, Dong M. GLP-1 analogue improves hepatic lipid accumulation by inducing autophagy via AMPK/mTOR pathway. Biochem Biophys Res Commun. 2016; 476:196–203.
Article34. Wang C, Li Q, Wang W, Guo L, Guo C, Sun Y, et al. GLP-1 contributes to increases in PGC-1α expression by downregulating miR-23a to reduce apoptosis. Biochem Biophys Res Commun. 2015; 466:33–9.
Article35. Mahalingam S, Bellamkonda R, Arumugam MK, Perumal SK, Yoon J, Casey C, et al. Glucagon-like peptide 1 receptor agonist, exendin-4, reduces alcohol-associated fatty liver disease. Biochem Pharmacol. 2023; 213:115613.
Article36. Ding X, Saxena NK, Lin S, Gupta NA, Anania FA. Exendin-4, a glucagon-like protein-1 (GLP-1) receptor agonist, reverses hepatic steatosis in ob/ob mice. Hepatology. 2006; 43:173–81.
Article37. Fang Y, Ji L, Zhu C, Xiao Y, Zhang J, Lu J, et al. Liraglutide alleviates hepatic steatosis by activating the TFEB-regulated autophagy-lysosomal pathway. Front Cell Dev Biol. 2020; 8:602574.
Article38. Omanovic Kolaric T, Kizivat T, Mihaljevic V, Zjalic M, BilicCurcic I, Kuna L, et al. Liraglutide exerts protective effects by downregulation of PPARγ, ACSL1 and SREBP-1c in Huh7 cell culture models of non-alcoholic steatosis and drug-induced steatosis. Curr Issues Mol Biol. 2022; 44:3465–80.
Article39. Czech TY, Wang Q, Seki E. A new mechanism of action of glucagon-like peptide-1 agonist in hepatic steatosis: promotion of hepatic insulin clearance through induction of carcinoembryonic antigen-related cell adhesion molecule 1. Hepatol Commun. 2018; 2:9–12.
Article40. Geloneze B, de Lima-Junior JC, Velloso LA. Glucagon-like peptide-1 receptor agonists (GLP-1RAs) in the brain-adipocyte axis. Drugs. 2017; 77:493–503.
Article41. Challa TD, Beaton N, Arnold M, Rudofsky G, Langhans W, Wolfrum C. Regulation of adipocyte formation by GLP-1/GLP-1R signaling. J Biol Chem. 2012; 287:6421–30.
Article42. Beiroa D, Imbernon M, Gallego R, Senra A, Herranz D, Villarroya F, et al. GLP-1 agonism stimulates brown adipose tissue thermogenesis and browning through hypothalamic AMPK. Diabetes. 2014; 63:3346–58.
Article43. Kooijman S, Wang Y, Parlevliet ET, Boon MR, Edelschaap D, Snaterse G, et al. Central GLP-1 receptor signalling accelerates plasma clearance of triacylglycerol and glucose by activating brown adipose tissue in mice. Diabetologia. 2015; 58:2637–46.
Article44. Lockie SH, Heppner KM, Chaudhary N, Chabenne JR, Morgan DA, Veyrat-Durebex C, et al. Direct control of brown adipose tissue thermogenesis by central nervous system glucagon-like peptide-1 receptor signaling. Diabetes. 2012; 61:2753–62.
Article45. Li X, Jiang L, Yang M, Wu Y, Sun S, Sun J. GLP-1 receptor agonist increases the expression of CTRP3, a novel adipokine, in 3T3-L1 adipocytes through PKA signal pathway. J Endocrinol Invest. 2015; 38:73–9.
Article46. Chen J, Zhao H, Ma X, Zhang Y, Lu S, Wang Y, et al. GLP-1/GLP-1R signaling in regulation of adipocyte differentiation and lipogenesis. Cell Physiol Biochem. 2017; 42:1165–76.
Article47. El Bekay R, Coin-Araguez L, Fernandez-Garcia D, Oliva-Olivera W, Bernal-Lopez R, Clemente-Postigo M, et al. Effects of glucagon-like peptide-1 on the differentiation and metabolism of human adipocytes. Br J Pharmacol. 2016; 173:1820–34.
Article48. Jin ES, Malloy CR, Sharma G, Finn E, Fuller KN, Reyes YG, et al. Glycerol as a precursor for hepatic de novo glutathione synthesis in human liver. Redox Biol. 2023; 63:102749.
Article49. Huby T, Gautier EL. Immune cell-mediated features of nonalcoholic steatohepatitis. Nat Rev Immunol. 2022; 22:429–43.
Article50. Bu H, Li L, Li X, Guo J. Long-term intake of a high protein diet modulates liver steatosis and fat accumulation in OVX mice. Chin J Comp Med. 2021; 31:69–75.51. Hou H, Qiu Y, Zhang J, Hu Y, Su S, Wang Y. GLP-1 down-regulates mRNA expression of SOCS-3 and SREBP-1c in rats with nonalcoholic fatty liver disease. Chin J Pathophysiol. 2016; 32:1312–6.52. Boland ML, Laker RC, Mather K, Nawrocki A, Oldham S, Boland BB, et al. Resolution of NASH and hepatic fibrosis by the GLP-1R/GcgR dual-agonist cotadutide via modulating mitochondrial function and lipogenesis. Nat Metab. 2020; 2:413–31.
Article53. Burmeister MA, Ferre T, Ayala JE, King EM, Holt RM, Ayala JE. Acute activation of central GLP-1 receptors enhances hepatic insulin action and insulin secretion in high-fat-fed, insulin resistant mice. Am J Physiol Endocrinol Metab. 2012; 302:E334–43.
Article54. Lu J, Chen L, Liu W, Guo W. Effects of liraglutide on lipid metabolism and expression of PI3K protein in KKAy mice. Chin J Drug Appl Monit. 2021; 18:363–6.55. Zhu H, Deng X, Gao J, Li X, Wang W, Wang M. Effects of liraglutide on expressions of lipid metabolism-related genes in rats with T2DM and fatty liver. Chin J Clin Res. 2017; 30:7–10.56. Lee J, Hong SW, Kim MJ, Moon SJ, Kwon H, Park SE, et al. Dulaglutide ameliorates palmitic acid-induced hepatic steatosis by activating FAM3A signaling pathway. Endocrinol Metab (Seoul). 2022; 37:74–83.
Article57. Wang H, Wang L, Li Y, Luo S, Ye J, Lu Z, et al. The HIF-2α/PPARα pathway is essential for liraglutide-alleviated, lipid-induced hepatic steatosis. Biomed Pharmacother. 2021; 140:111778.
Article58. Zhu CG, Luo Y, Wang H, Li JY, Yang J, Liu YX, et al. Liraglutide ameliorates lipotoxicity-induced oxidative stress by activating the NRF2 pathway in HepG2 cells. Horm Metab Res. 2020; 52:532–9.
Article59. Zhou D, Chen YW, Zhao ZH, Yang RX, Xin FZ, Liu XL, et al. Sodium butyrate reduces high-fat diet-induced non-alcoholic steatohepatitis through upregulation of hepatic GLP-1R expression. Exp Mol Med. 2018; 50:1–12.
Article60. Yu P, Xu X, Zhang J, Xia X, Xu F, Weng J, et al. Liraglutide attenuates nonalcoholic fatty liver disease through adjusting lipid metabolism via SHP1/AMPK signaling pathway. Int J Endocrinol. 2019; 2019:1567095.
Article61. Zheng X, Xu F, Liang H, Cao H, Cai M, Xu W, et al. SIRT1/ HSF1/HSP pathway is essential for exenatide-alleviated, lipidinduced hepatic endoplasmic reticulum stress. Hepatology. 2017; 66:809–24.
Article62. Zhu W, Feng PP, He K, Li SW, Gong JP. Liraglutide protects non-alcoholic fatty liver disease via inhibiting NLRP3 inflammasome activation in a mouse model induced by high-fat diet. Biochem Biophys Res Commun. 2018; 505:523–9.
Article63. Gupta NA, Mells J, Dunham RM, Grakoui A, Handy J, Saxena NK, et al. Glucagon-like peptide-1 receptor is present on human hepatocytes and has a direct role in decreasing hepatic steatosis in vitro by modulating elements of the insulin signaling pathway. Hepatology. 2010; 51:1584–92.
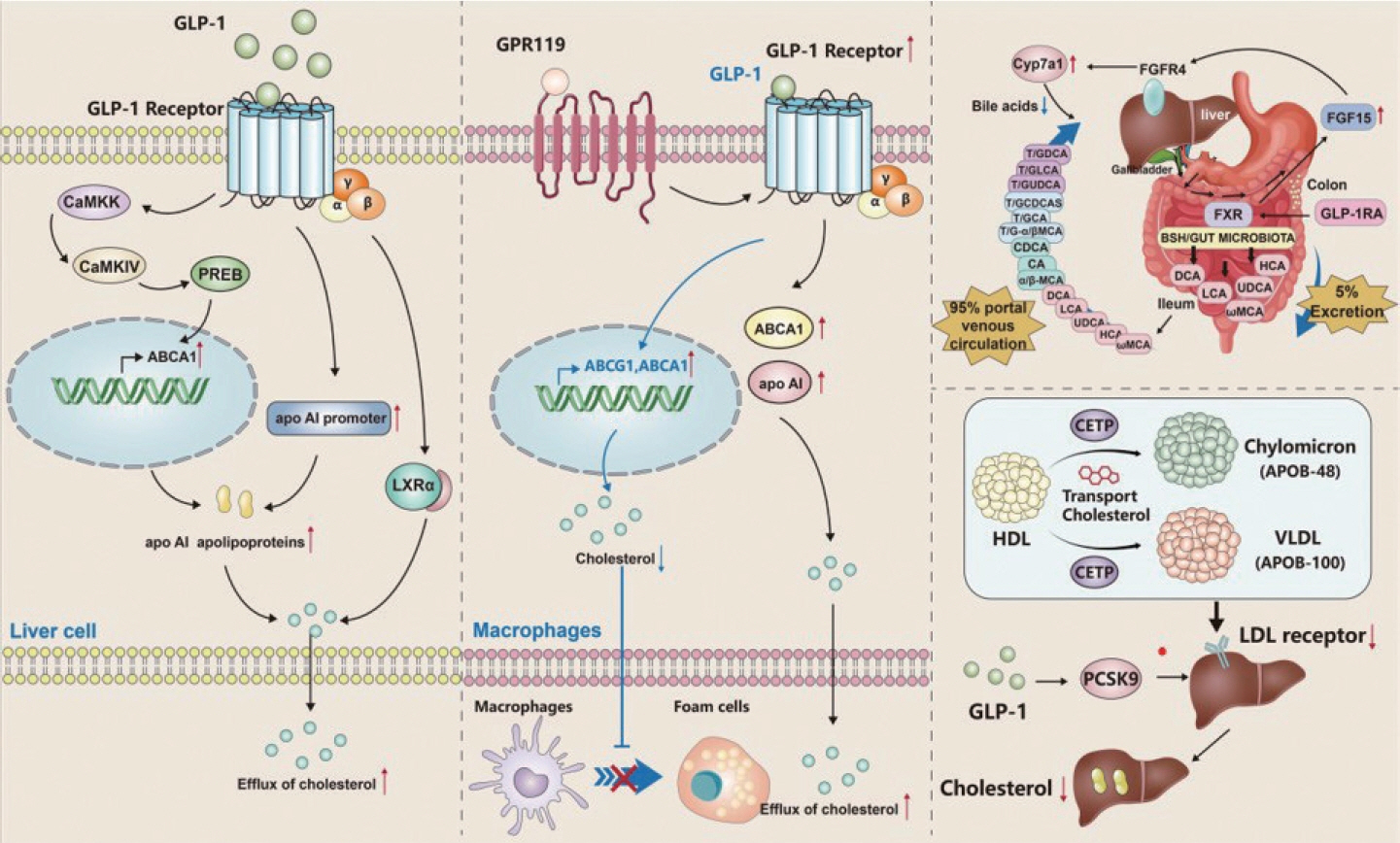
Article64. Sharma S, Mells JE, Fu PP, Saxena NK, Anania FA. GLP-1 analogs reduce hepatocyte steatosis and improve survival by enhancing the unfolded protein response and promoting macroautophagy. PLoS One. 2011; 6:e25269.
Article65. Liu D, Pang J, Shao W, Gu J, Zeng Y, He HH, et al. Hepatic fibroblast growth factor 21 is involved in mediating functions of liraglutide in mice with dietary challenge. Hepatology. 2021; 74:2154–69.
Article66. Zhang G, Han J, Wang L, Yang X, Yan Z, Qu M, et al. The vesicular transporter STX11 governs ATGL-mediated hepatic lipolysis and lipophagy. iScience. 2022; 25:104085.
Article67. Huang Y. Study on the mechanism of liraglutide to improve hepatic lipid metabolism in mice [dissertation]. Yangzhou: Yangzhou University;2022.68. Matikainen N, Soderlund S, Bjornson E, Pietilainen K, Hakkarainen A, Lundbom N, et al. Liraglutide treatment improves postprandial lipid metabolism and cardiometabolic risk factors in humans with adequately controlled type 2 diabetes: a single-centre randomized controlled study. Diabetes Obes Metab. 2019; 21:84–94.
Article69. Luo J, Yang H, Song BL. Mechanisms and regulation of cholesterol homeostasis. Nat Rev Mol Cell Biol. 2020; 21:225–45.
Article70. Ben-Shlomo S, Zvibel I, Rabinowich L, Goldiner I, Shlomai A, Santo EM, et al. Dipeptidyl peptidase 4-deficient rats have improved bile secretory function in high fat diet-induced steatosis. Dig Dis Sci. 2013; 58:172–8.
Article71. Lyu J, Imachi H, Fukunaga K, Sato S, Kobayashi T, Dong T, et al. Role of ATP-binding cassette transporter A1 in suppressing lipid accumulation by glucagon-like peptide-1 agonist in hepatocytes. Mol Metab. 2020; 34:16–26.
Article72. Han Y, Yue Y, Zhang K, Shen X. GLP-1(7-36) inhibits atherosclerosis progression in ApoE-/- mouse aorta by lowering expressions of CD36 and ACAT1 in foam cells. J Amry Med Univ. 2019; 41:1947–53.73. Hu YW, Yang JY, Ma X, Chen ZP, Hu YR, Zhao JY, et al. A lincRNA-DYNLRB2-2/GPR119/GLP-1R/ABCA1-dependent signal transduction pathway is essential for the regulation of cholesterol homeostasis. J Lipid Res. 2014; 55:681–97.74. Wang YG, Yang TL. Liraglutide reduces oxidized LDL-induced oxidative stress and fatty degeneration in Raw 264.7 cells involving the AMPK/SREBP1 pathway. J Geriatr Cardiol. 2015; 12:410–6.75. Hori M, Hasegawa Y, Hayashi Y, Nakagami T, Harada-Shiba M. Acute cholesterol-lowering effect of exendin-4 in Ldlr-/- and C57BL/6J mice. J Atheroscler Thromb. 2023; 30:74–86.76. Zhang Z, Du Z, Liu Q, Wu T, Tang Q, Zhang J, et al. Glucagonlike peptide 1 analogue prevents cholesterol gallstone formation by modulating intestinal farnesoid X receptor activity. Metabolism. 2021; 118:154728.
Article77. Fielding CJ, Fielding PE. Molecular physiology of reverse cholesterol transport. J Lipid Res. 1995; 36:211–28.
Article78. Yang SH, Xu RX, Cui CJ, Wang Y, Du Y, Chen ZG, et al. Liraglutide downregulates hepatic LDL receptor and PCSK9 expression in HepG2 cells and db/db mice through a HNF-1a dependent mechanism. Cardiovasc Diabetol. 2018; 17:48.
Article79. Hoffman S, Alvares D, Adeli K. GLP-1 attenuates intestinal fat absorption and chylomicron production via vagal afferent nerves originating in the portal vein. Mol Metab. 2022; 65:101590.
Article80. Yang J, Ren J, Song J, Liu F, Wu C, Wang X, et al. Glucagon-like peptide 1 regulates adipogenesis in 3T3-L1 preadipocytes. Int J Mol Med. 2013; 31:1429–35.
Article81. Cui J. Study on mechanisms of 3T3-L1 adipocyte differentiation promoted by GLP-1 mediated upregulation of LncRNA PFAR and its effect on obesity in mice [dissertation]. Taiyuan: Shanxi Medical University;2020.82. Rodriguez-Barrueco R, Latorre J, Devis-Jauregui L, Lluch A, Bonifaci N, Llobet FJ, et al. A microRNA cluster controls fat cell differentiation and adipose tissue expansion by regulating SNCG. Adv Sci (Weinh). 2022; 9:e2104759.
Article83. Tabuchi C, Sul HS. Corrigendum: signaling pathways regulating thermogenesis. Front Endocrinol (Lausanne). 2021; 12:698619.
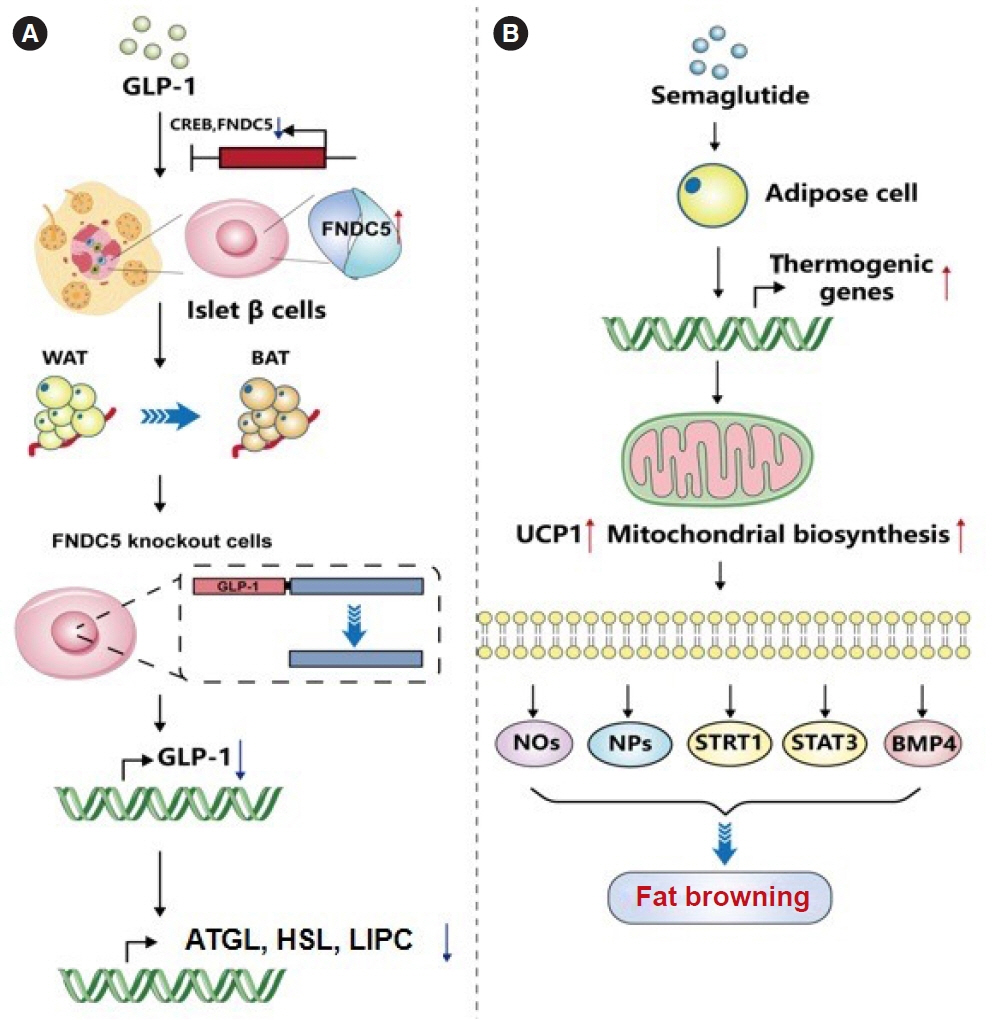
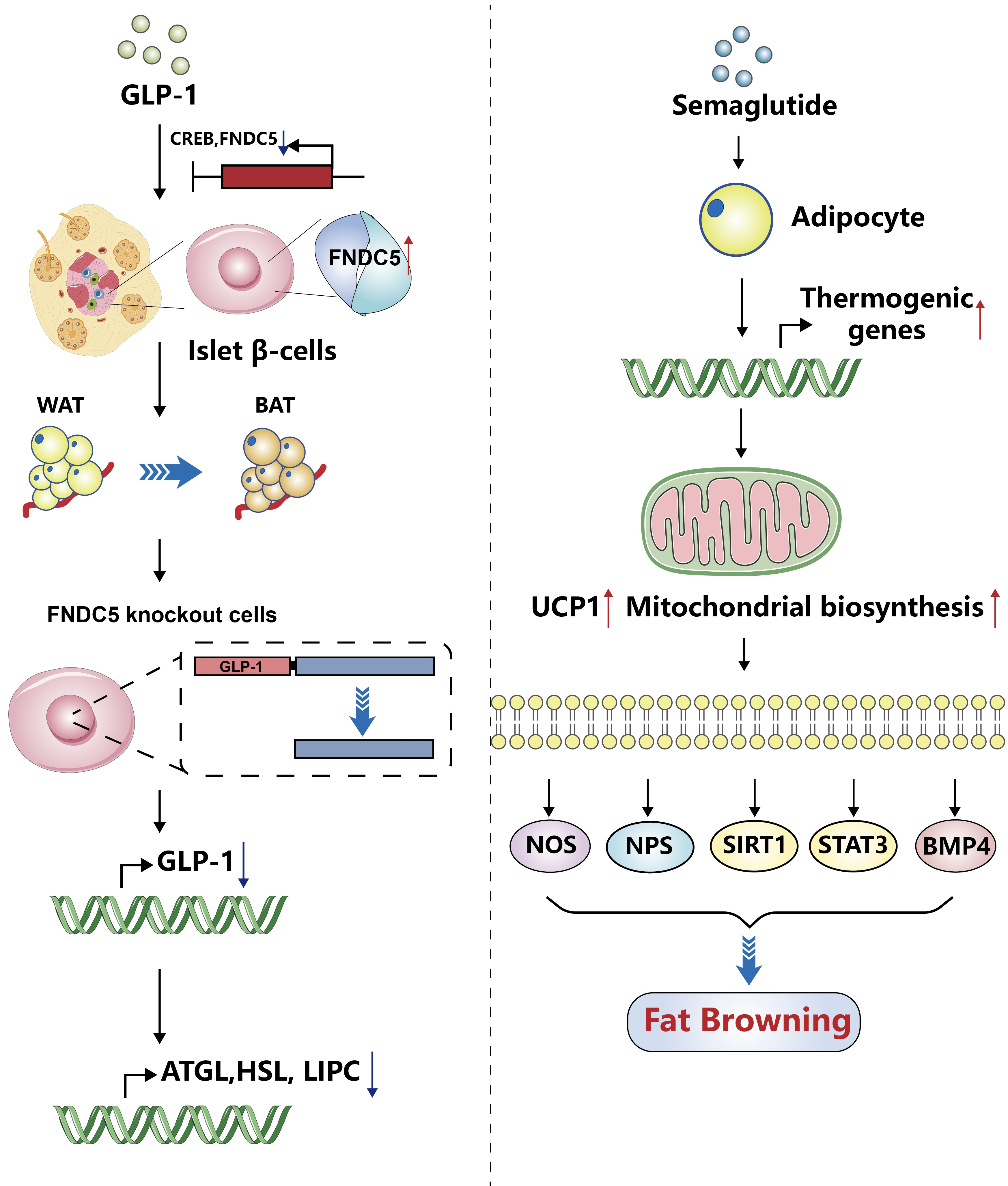
Article84. Kaisanlahti A, Glumoff T. Browning of white fat: agents and implications for beige adipose tissue to type 2 diabetes. J Physiol Biochem. 2019; 75:1–10.
Article85. Li H. GLP-1 induces the expression of FNDC5 derivatives that execute lipolytic actions [dissertation]. Jinan: Shandong University;2022.86. Martins FF, Marinho TS, Cardoso LE, Barbosa-da-Silva S, Souza-Mello V, Aguila MB, et al. Semaglutide (GLP-1 receptor agonist) stimulates browning on subcutaneous fat adipocytes and mitigates inflammation and endoplasmic reticulum stress in visceral fat adipocytes of obese mice. Cell Biochem Funct. 2022; 40:903–13.
Article87. Maiseyeu A, Di L, Ravodina A, Barajas-Espinosa A, Sakamoto A, Chaplin A, et al. Plaque-targeted, proteolysis-resistant, activatable and MRI-visible nano-GLP-1 receptor agonist targets smooth muscle cell differentiation in atherosclerosis. Theranostics. 2022; 12:2741–57.
Article88. Boutant M, Joffraud M, Kulkarni SS, Garcia-Casarrubios E, Garcia-Roves PM, Ratajczak J, et al. SIRT1 enhances glucose tolerance by potentiating brown adipose tissue function. Mol Metab. 2014; 4:118–31.
Article89. Gutierrez AD, Gao Z, Hamidi V, Zhu L, Saint Andre KB, Riggs K, et al. Anti-diabetic effects of GLP1 analogs are mediated by thermogenic interleukin-6 signaling in adipocytes. Cell Rep Med. 2022; 3:100813.
Article90. Wang X, Ma B, Chen J, You H, Sheng C, Yang P, et al. Glucagon-like peptide-1 improves fatty liver and enhances thermogenesis in brown adipose tissue via inhibiting BMP4-related signaling pathway in high-fat-diet-induced obese mice. Int J Endocrinol. 2021; 2021:6620289.
Article91. Yoon HS, Cho CH, Yun MS, Jang SJ, You HJ, Kim JH, et al. Akkermansia muciniphila secretes a glucagon-like peptide1-inducing protein that improves glucose homeostasis and ameliorates metabolic disease in mice. Nat Microbiol. 2021; 6:563–73.
Article92. Muscelli E, Mari A, Casolaro A, Camastra S, Seghieri G, Gastaldelli A, et al. Separate impact of obesity and glucose tolerance on the incretin effect in normal subjects and type 2 diabetic patients. Diabetes. 2008; 57:1340–8.
Article93. Vilsboll T, Krarup T, Sonne J, Madsbad S, Volund A, Juul AG, et al. Incretin secretion in relation to meal size and body weight in healthy subjects and people with type 1 and type 2 diabetes mellitus. J Clin Endocrinol Metab. 2003; 88:2706–13.
Article94. Vilsboll T, Krarup T, Deacon CF, Madsbad S, Holst JJ. Reduced postprandial concentrations of intact biologically active glucagon-like peptide 1 in type 2 diabetic patients. Diabetes. 2001; 50:609–13.
Article95. Vendrell J, El Bekay R, Peral B, Garcia-Fuentes E, Megia A, Macias-Gonzalez M, et al. Study of the potential association of adipose tissue GLP-1 receptor with obesity and insulin resistance. Endocrinology. 2011; 152:4072–9.
Article96. Wang L, Chen T, Wang H, Wu X, Cao Q, Wen K, et al. Engineered bacteria of MG1363-pMG36e-GLP-1 attenuated obesity-induced by high fat diet in mice. Front Cell Infect Microbiol. 2021; 11:595575.
Article97. Cantini G, Trabucco M, Di Franco A, Mannucci E, Luconi M. Glucagon modulates proliferation and differentiation of human adipose precursors. J Mol Endocrinol. 2019; 63:249–60.
Article98. Cantini G, Di Franco A, Samavat J, Forti G, Mannucci E, Luconi M. Effect of liraglutide on proliferation and differentiation of human adipose stem cells. Mol Cell Endocrinol. 2015; 402:43–50.
Article99. Baggio LL, Drucker DJ. Glucagon-like peptide-1 receptors in the brain: controlling food intake and body weight. J Clin Invest. 2014; 124:4223–6.
Article100. Allegretti JR, Kassam Z, Mullish BH, Chiang A, Carrellas M, Hurtado J, et al. Effects of fecal microbiota transplantation with oral capsules in obese patients. Clin Gastroenterol Hepatol. 2020; 18:855–63.
Article101. Sandsdal RM, Juhl CR, Jensen SB, Lundgren JR, Janus C, Blond MB, et al. Combination of exercise and GLP-1 receptor agonist treatment reduces severity of metabolic syndrome, abdominal obesity, and inflammation: a randomized controlled trial. Cardiovasc Diabetol. 2023; 22:41.
Article102. Zhang Y, Zhao X, Dong X, Zhang Y, Zou H, Jin Y, et al. Activity-balanced GLP-1/GDF15 dual agonist reduces body weight and metabolic disorder in mice and non-human primates. Cell Metab. 2023; 35:287–98.
Article103. El-Agroudy NN, Kurzbach A, Rodionov RN, O’Sullivan J, Roden M, Birkenfeld AL, et al. Are lifestyle therapies effective for NAFLD treatment? Trends Endocrinol Metab. 2019; 30:701–9.
Article104. Li S, Wang X, Zhang J, Li J, Liu X, Ma Y, et al. Exenatide ameliorates hepatic steatosis and attenuates fat mass and FTO gene expression through PI3K signaling pathway in nonalcoholic fatty liver disease. Braz J Med Biol Res. 2018; 51:e7299.
Article105. Czaja MJ. JNK regulation of hepatic manifestations of the metabolic syndrome. Trends Endocrinol Metab. 2010; 21:707–13.
Article106. Yu X, Hao M, Liu Y, Ma X, Lin W, Xu Q, et al. Liraglutide ameliorates non-alcoholic steatohepatitis by inhibiting NLRP3 inflammasome and pyroptosis activation via mitophagy. Eur J Pharmacol. 2019; 864:172715.
Article107. Carvalho T. Efruxifermin combined with a GLP-1 receptor agonist reduces liver fat in NASH. Nat Med. 2023; 29:1881.
Article108. Xie J, Yang M, Xing Y. Effects of liraglutide on glucose and lipid metabolism and insulin resistance in type 2 diabetes mellitus patients with non-alcoholic fatty liver disease. Chin J Liver Dis. 2021; 13:46–53.109. Patel Chavez C, Cusi K, Kadiyala S. The emerging role of glucagon-like peptide-1 receptor agonists for the management of NAFLD. J Clin Endocrinol Metab. 2022; 107:29–38.
Article110. Jianping W, Xuelian Z, Anjiang W, Haiying X. Efficacy and safety of glucagon-like peptide-1 receptor agonists in the treatment of metabolic associated fatty liver disease: a systematic review and meta-analysis. J Clin Gastroenterol. 2021; 55:586–93.111. Hopkins ND, Cuthbertson DJ, Kemp GJ, Pugh C, Green DJ, Cable NT, et al. Effects of 6 months glucagon-like peptide-1 receptor agonist treatment on endothelial function in type 2 diabetes mellitus patients. Diabetes Obes Metab. 2013; 15:770–3.112. Gastaldelli A, Repetto E, Guja C, Hardy E, Han J, Jabbour SA, et al. Exenatide and dapagliflozin combination improves markers of liver steatosis and fibrosis in patients with type 2 diabetes. Diabetes Obes Metab. 2020; 22:393–403.
Article113. Alkhouri N, Herring R, Kabler H, Kayali Z, Hassanein T, Kohli A, et al. Safety and efficacy of combination therapy with semaglutide, cilofexor and firsocostat in patients with non-alcoholic steatohepatitis: a randomised, open-label phase II trial. J Hepatol. 2022; 77:607–18.
Article114. Flint A, Andersen G, Hockings P, Johansson L, Morsing A, Sundby Palle M, et al. Randomised clinical trial: semaglutide versus placebo reduced liver steatosis but not liver stiffness in subjects with non-alcoholic fatty liver disease assessed by magnetic resonance imaging. Aliment Pharmacol Ther. 2021; 54:1150–61.115. Song N, Xu H, Liu J, Zhao Q, Chen H, Yan Z, et al. Design of a highly potent GLP-1R and GCGR dual-agonist for recovering hepatic fibrosis. Acta Pharm Sin B. 2022; 12:2443–61.116. Romero-Gomez M, Lawitz E, Shankar RR, Chaudhri E, Liu J, Lam RL, et al. A phase IIa active-comparator-controlled study to evaluate the efficacy and safety of efinopegdutide in patients with non-alcoholic fatty liver disease. J Hepatol. 2023; 79:888–97.117. Yabut JM, Drucker DJ. Glucagon-like peptide-1 receptor-based therapeutics for metabolic liver disease. Endocr Rev. 2023; 44:14–32.118. Kalogirou MS, Patoulias D, Haidich AB, Akriviadis E, Sinakos E. Liraglutide in patients with non-alcoholic fatty liver disease: a systematic review and meta-analysis of randomized controlled trials. Clin Res Hepatol Gastroenterol. 2021; 45:101568.
Article119. Newsome PN, Buchholtz K, Cusi K, Linder M, Okanoue T, Ratziu V, et al. A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. N Engl J Med. 2021; 384:1113–24.
Article120. Romero-Gomez M, Armstrong MJ, Funuyet-Salas J, Mangla KK, Ladelund S, Sejling AS, et al. Improved health-related quality of life with semaglutide in people with non-alcoholic steatohepatitis: a randomised trial. Aliment Pharmacol Ther. 2023; 58:395–403.
Article121. Dichtel LE. The glucagon-like peptide-1 receptor agonist, semaglutide, for the treatment of nonalcoholic steatohepatitis. Hepatology. 2021; 74:2290–2.
Article122. Ishikawa S, Shimano M, Watarai M, Koyasu M, Uchikawa T, Ishii H, et al. Impact of sitagliptin on carotid intima-media thickness in patients with coronary artery disease and impaired glucose tolerance or mild diabetes mellitus. Am J Cardiol. 2014; 114:384–8.
Article123. Burgmaier M, Liberman A, Mollmann J, Kahles F, Reith S, Lebherz C, et al. Glucagon-like peptide-1 (GLP-1) and its split products GLP-1(9-37) and GLP-1(28-37) stabilize atherosclerotic lesions in apoe-/- mice. Atherosclerosis. 2013; 231:427–35.
Article124. Koska J, Migrino RQ, Chan KC, Cooper-Cox K, Reaven PD. The effect of exenatide once weekly on carotid atherosclerosis in individuals with type 2 diabetes: an 18-month randomized placebo-controlled study. Diabetes Care. 2021; 44:1385–92.
Article125. Min T, Bain SC. The role of tirzepatide, dual GIP and GLP-1 receptor agonist, in the management of type 2 diabetes: the SURPASS clinical trials. Diabetes Ther. 2021; 12:143–57.
Article126. Frias JP, Nauck MA, Van J, Kutner ME, Cui X, Benson C, et al. Efficacy and safety of LY3298176, a novel dual GIP and GLP-1 receptor agonist, in patients with type 2 diabetes: a randomised, placebo-controlled and active comparator-controlled phase 2 trial. Lancet. 2018; 392:2180–93.
Article127. Liu C, Li C, Cai X, Zou Y, Mo J, Chen B, et al. Discovery of a novel GLP-1/GIP dual receptor agonist CY-5 as long-acting hypoglycemic, anti-obesity agent. Bioorg Chem. 2021; 106:104492.
Article128. Dahl D, Onishi Y, Norwood P, Huh R, Bray R, Patel H, et al. Effect of subcutaneous tirzepatide vs placebo added to titrated insulin glargine on glycemic control in patients with type 2 diabetes: the SURPASS-5 randomized clinical trial. JAMA. 2022; 327:534–45.
Article129. Yu D, Shen S, Zhang J, Wang Q. Effect of the dual glucose‐dependent insulinotropic peptide/gulcagon‐like peptide 1 receptor agonist tirzepatide on lipid profile and waist circumference: a systematic review and meta‐analysis. Clin Ther. 2023; 45:787–96.
Article130. Zhao F, Zhou Q, Cong Z, Hang K, Zou X, Zhang C, et al. Structural insights into multiplexed pharmacological actions of tirzepatide and peptide 20 at the GIP, GLP-1 or glucagon receptors. Nat Commun. 2022; 13:1057.
Article131. Gastaldelli A, Cusi K, Fernandez Lando L, Bray R, Brouwers B, Rodriguez A. Effect of tirzepatide versus insulin degludec on liver fat content and abdominal adipose tissue in people with type 2 diabetes (SURPASS-3 MRI): a substudy of the randomised, open-label, parallel-group, phase 3 SURPASS-3 trial. Lancet Diabetes Endocrinol. 2022; 10:393–406.
Article132. Syed YY. Tirzepatide: first approval. Drugs. 2022; 82:1213–20.
Article133. Pedrosa MR, Franco DR, Gieremek HW, Vidal CM, Bronzeri F, de Cassia Rocha A, et al. GLP-1 agonist to treat obesity and prevent cardiovascular disease: what have we achieved so far? Curr Atheroscler Rep. 2022; 24:867–84.
Article134. Samms RJ, Coghlan MP, Sloop KW. How may GIP enhance the therapeutic efficacy of GLP-1? Trends Endocrinol Metab. 2020; 31:410–21.
Article135. Wang L. Designing a dual GLP-1R/GIPR agonist from tirzepatide: comparing residues between tirzepatide, GLP-1, and GIP. Drug Des Devel Ther. 2022; 16:1547–59.
Article136. Jastreboff AM, Aronne LJ, Ahmad NN, Wharton S, Connery L, Alves B, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022; 387:205–16.
Article137. Garvey WT, Frias JP, Jastreboff AM, le Roux CW, Sattar N, Aizenberg D, et al. Tirzepatide once weekly for the treatment of obesity in people with type 2 diabetes (SURMOUNT-2): a double-blind, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2023; 402:613–26.138. Rizvi AA, Rizzo M. The emerging role of dual GLP-1 and GIP receptor agonists in glycemic management and cardiovascular risk reduction. Diabetes Metab Syndr Obes. 2022; 15:1023–30.
Article139. Fisman EZ, Tenenbaum A. The dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist tirzepatide: a novel cardiometabolic therapeutic prospect. Cardiovasc Diabetol. 2021; 20:225.
Article140. Buse JB, Wexler DJ, Tsapas A, Rossing P, Mingrone G, Mathieu C, et al. 2019 Update to: management of hyperglycemia in type 2 diabetes, 2018: a consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2020; 43:487–93.
Article141. Ma T, Lu W, Wang Y, Qian P, Tian H, Gao X, et al. An oral GLP1 and GIP dual receptor agonist improves metabolic disorders in high fat-fed mice. Eur J Pharmacol. 2022; 914:174635.
Article142. Nogueiras R, Nauck MA, Tschop MH. Gut hormone co-agonists for the treatment of obesity: from bench to bedside. Nat Metab. 2023; 5:933–44.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Glucagon-like Peptide-1 Analogue and Dipeptidyl Peptidase-IV Inhibitors
- Letter: Expression of Glucagon-Like Peptide-1 Receptor in Papillary Thyroid Carcinoma and Its Clinicopathologic Significance (Endocrinol Metab 2014;29:536-44, Min Jung Jung et al.)
- Response: Expression of Glucagon-Like Peptide-1 Receptor in Papillary Thyroid Carcinoma and Its Clinicopathologic Significance (Endocrinol Metab 2014;29:536-44, Min Jung Jung et al.)
- GLP-1 Receptor Agonist and Non-Alcoholic Fatty Liver Disease
- New Potential Targets of Glucagon-Like Peptide 1 Receptor Agonists in Pancreatic β-Cells and Hepatocytes