Korean J Physiol Pharmacol.
2024 May;28(3):285-294. 10.4196/kjpp.2024.28.3.285.
Fibroblast-derived interleukin-6 exacerbates adverse cardiac remodeling after myocardial infarction
- Affiliations
-
- 1Key Laboratory of Cardiovascular Medicine and Clinical Pharmacology of Shanxi Province, Taiyuan 030001, Shanxi, China
- 2Department of Cardiology, The Second Affiliated Hospital of Shanxi Medical University, Taiyuan 030001, Shanxi, China
- KMID: 2555670
- DOI: http://doi.org/10.4196/kjpp.2024.28.3.285
Abstract
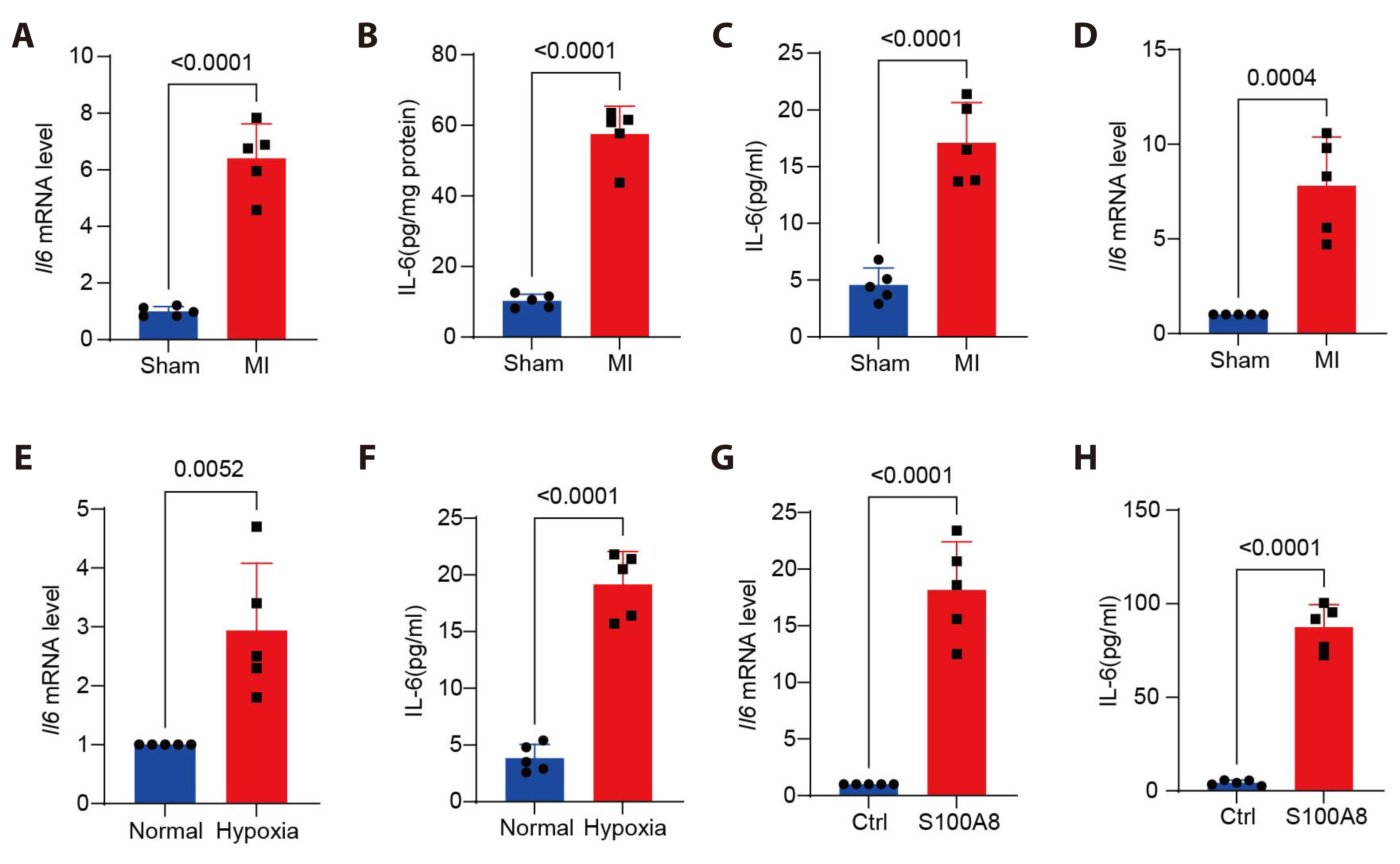
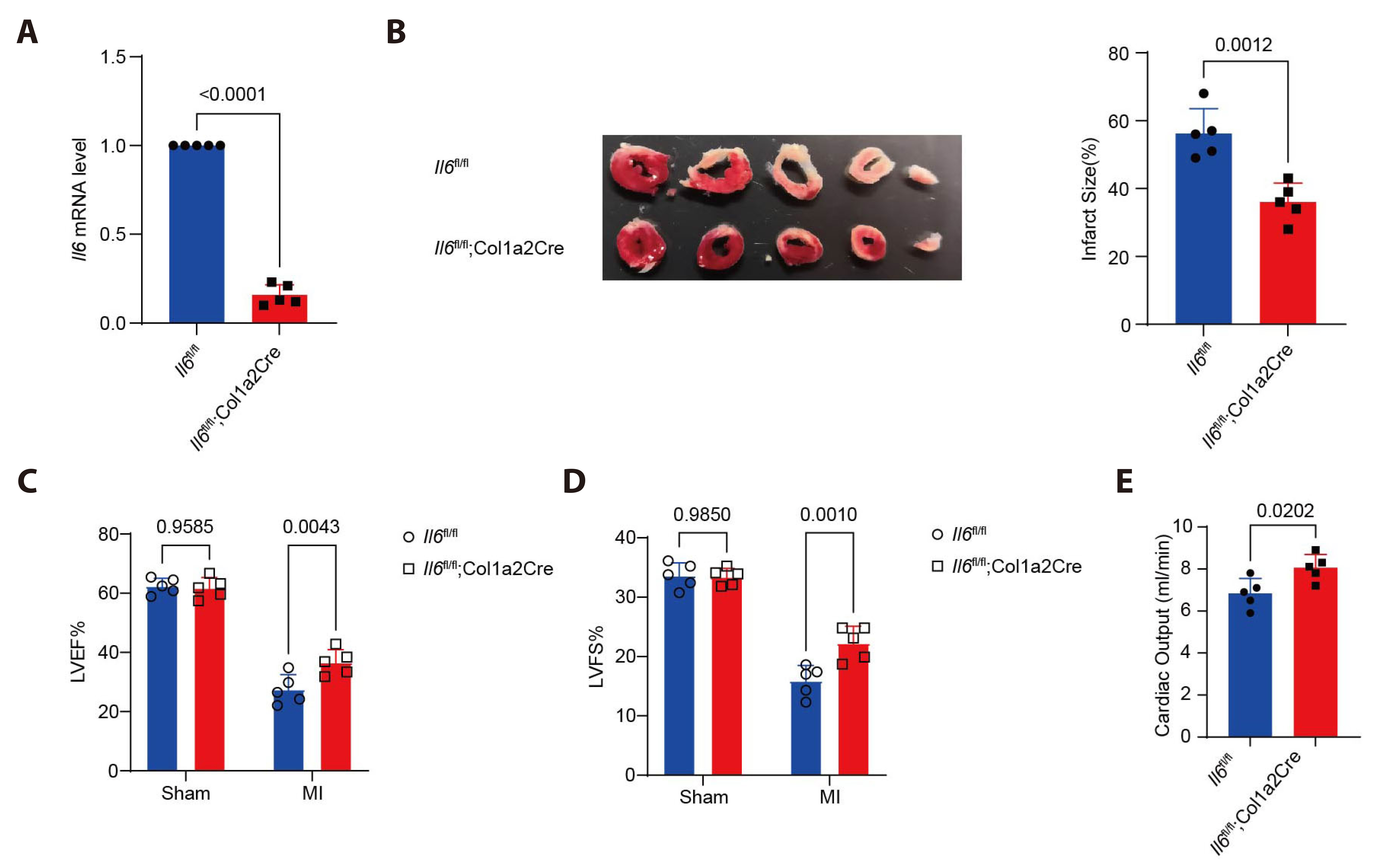
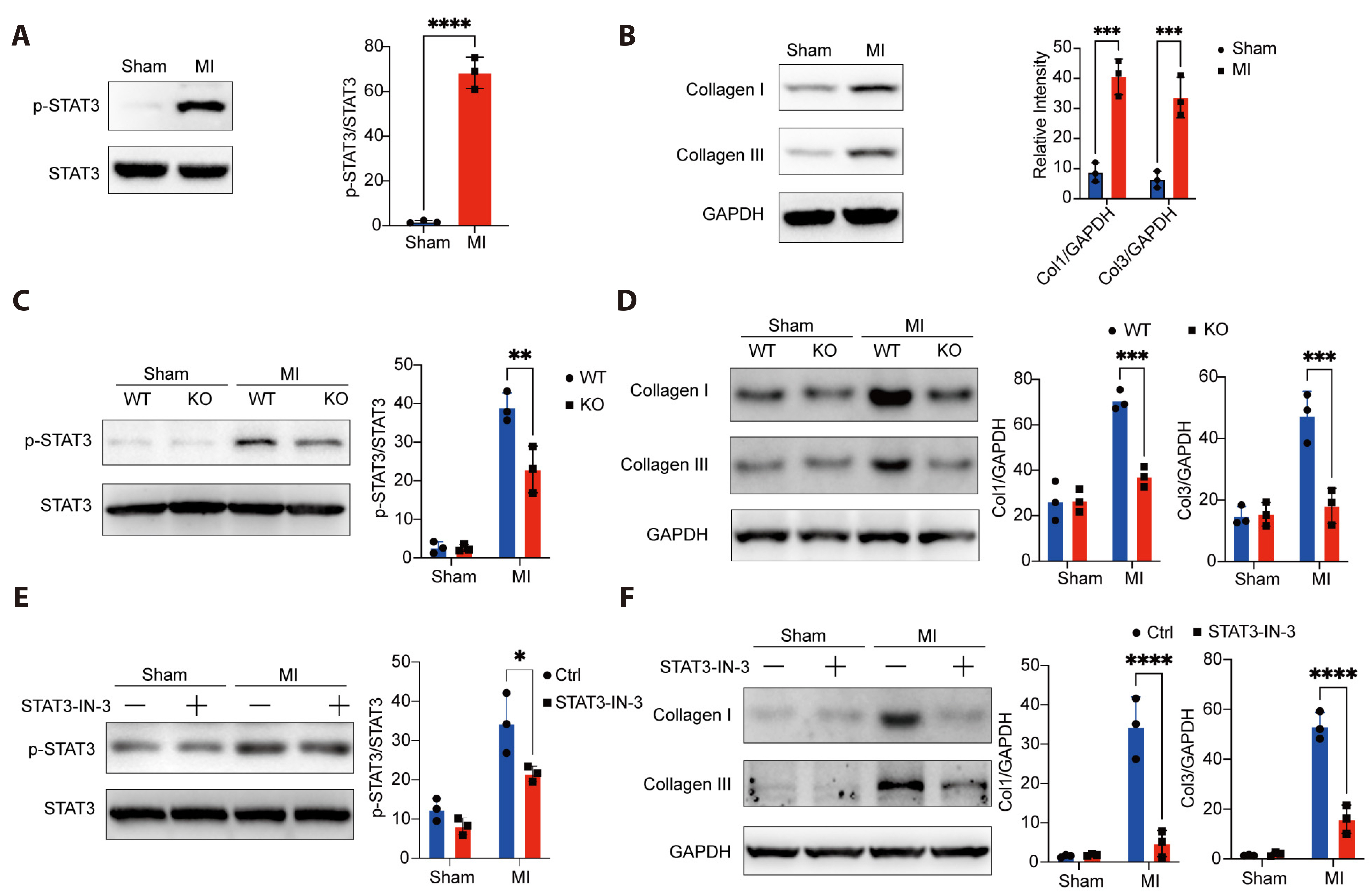
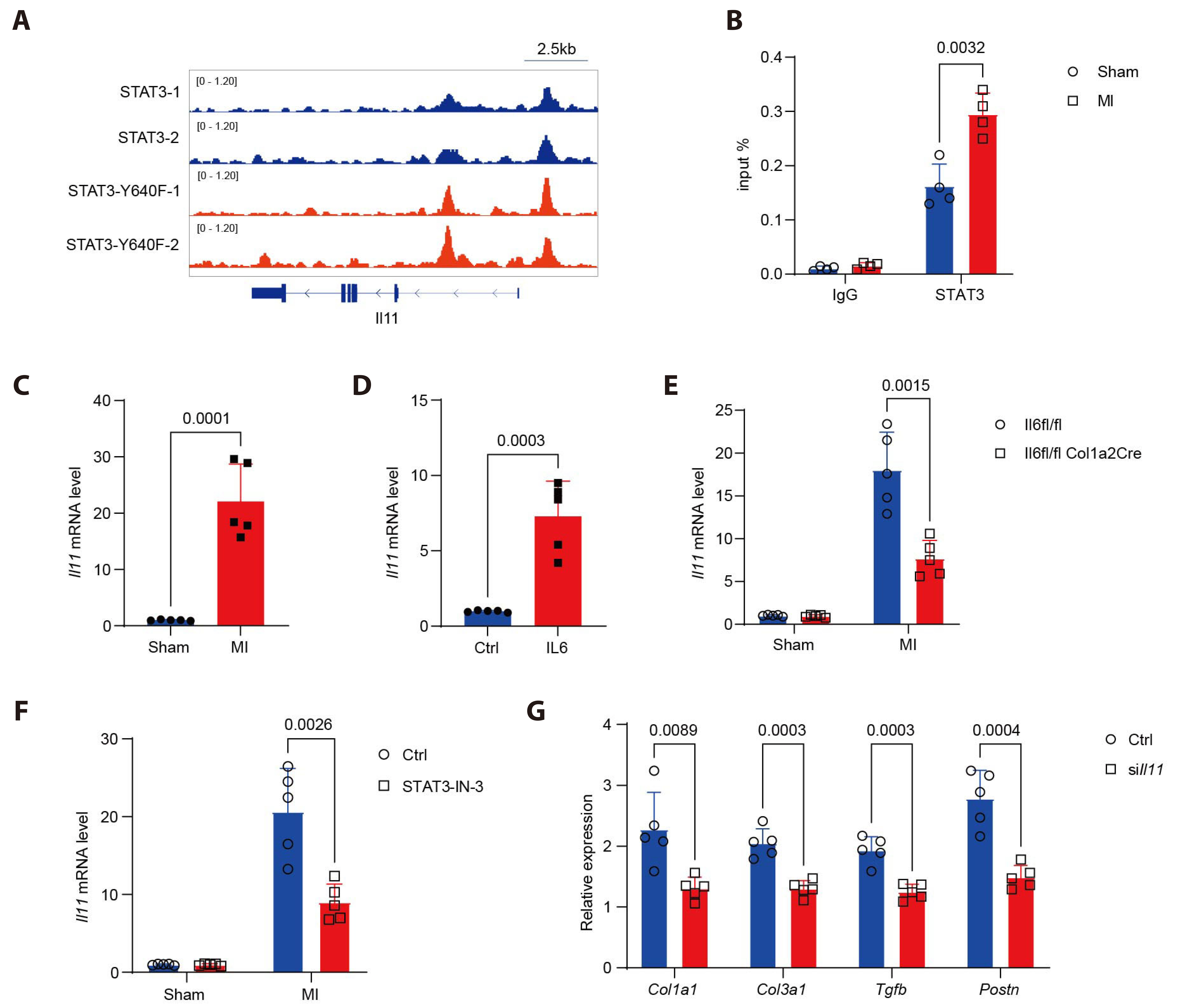
- Myocardial infarction is one of the leading causes of mortality globally. Currently, the pleiotropic inflammatory cytokine interleukin-6 (IL-6) is considered to be intimately related to the severity of myocardial injury during myocardial infarction. Interventions targeting IL-6 are a promising therapeutic option for myocardial infarction, but the underlying molecular mechanisms are not well understood. Here, we report the novel role of IL-6 in regulating adverse cardiac remodeling mediated by fibroblasts in a mouse model of myocardial infarction. It was found that the elevated expression of IL-6 in myocardium and cardiac fibroblasts was observed after myocardial infarction. Further, fibroblast-specific knockdown of Il6 significantly attenuated cardiac fibrosis and adverse cardiac remodeling and preserved cardiac function induced by myocardial infarction. Mechanistically, the role of Il6 contributing to cardiac fibrosis depends on signal transduction and activation of transcription (STAT)3 signaling activation. Additionally, Stat3 binds to the Il11 promoter region and contributes to the increased expression of Il11, which exacerbates cardiac fibrosis. In conclusion, these results suggest a novel role for IL-6 derived from fibroblasts in mediating Stat3 activation and substantially augmented Il11 expression in promoting cardiac fibrosis, highlighting its potential as a therapeutic target for cardiac fibrosis.
Figure
Reference
-
1. Bing R, Dweck MR. 2019; Myocardial fibrosis: why image, how to image and clinical implications. Heart. 105:1832–1840. DOI: 10.1136/heartjnl-2019-315560. PMID: 31649047. PMCID: PMC6900237.
Article2. Li Y, Li Z, Zhang C, Li P, Wu Y, Wang C, Bond Lau W, Ma XL, Du J. 2017; Cardiac fibroblast-specific activating transcription factor 3 protects against heart failure by suppressing MAP2K3-p38 signaling. Circulation. 135:2041–2057. DOI: 10.1161/CIRCULATIONAHA.116.024599. PMID: 28249877. PMCID: PMC5542579.
Article3. Su JH, Luo MY, Liang N, Gong SX, Chen W, Huang WQ, Tian Y, Wang AP. 2021; Interleukin-6: a novel target for cardio-cerebrovascular diseases. Front Pharmacol. 12:745061. DOI: 10.3389/fphar.2021.745061. PMID: 34504432. PMCID: PMC8421530.
Article4. Ridker PM, Rane M. 2021; Interleukin-6 signaling and anti-interleukin-6 therapeutics in cardiovascular disease. Circ Res. 128:1728–1746. DOI: 10.1161/CIRCRESAHA.121.319077. PMID: 33998272.
Article5. Fu W, Fang X, Wu L, Hu W, Yang T. 2022; Neogambogic acid relieves myocardial injury induced by sepsis via p38 MAPK/NF-κB pathway. Korean J Physiol Pharmacol. 26:511–518. DOI: 10.4196/kjpp.2022.26.6.511. PMID: 36302625. PMCID: PMC9614397.
Article6. Shen J, Ma H, Wang C. 2021; Triptolide improves myocardial fibrosis in rats through inhibition of nuclear factor kappa B and NLR family pyrin domain containing 3 inflammasome pathway. Korean J Physiol Pharmacol. 25:533–543. DOI: 10.4196/kjpp.2021.25.6.533. PMID: 34697264. PMCID: PMC8552823.
Article7. Xing C, Xiang D, Caiying L. 2020; Effects of troxerutin on vascular inflammatory mediators and expression of microRNA-146a/NF-κB signaling pathway in aorta of healthy and diabetic rats. Korean J Physiol Pharmacol. 24:395–402. DOI: 10.4196/kjpp.2020.24.5.395. PMID: 32830146. PMCID: PMC7445477.
Article8. Jin Y, Wang H, Li J, Dang M, Zhang W, Lei Y, Zhao H. 2020; Exploring the beneficial role of telmisartan in sepsis-induced myocardial injury through inhibition of high-mobility group box 1 and glycogen synthase kinase-3β/nuclear factor-κB pathway. Korean J Physiol Pharmacol. 24:311–317. DOI: 10.4196/kjpp.2020.24.4.311. PMID: 32587125. PMCID: PMC7317178.
Article9. Gibb AA, Lazaropoulos MP, Elrod JW. 2020; Myofibroblasts and fibrosis: mitochondrial and metabolic control of cellular differentiation. Circ Res. 127:427–447. DOI: 10.1161/CIRCRESAHA.120.316958. PMID: 32673537. PMCID: PMC7982967.10. Wang B, Tan Y, Zhou W, Yang J, Jiang Y, Liu X, Zhan Z. 2022; Loss of BTK ameliorates the pathological cardiac fibrosis and dysfunction. Matrix Biol. 112:171–189. DOI: 10.1016/j.matbio.2022.08.010. PMID: 36031013.
Article11. Wong HH, Seet SH, Bascom CC, Isfort RJ, Bard FA. 2023; Tonic repression of collagen I by the bradykinin receptor 2 in skin fibroblasts. Matrix Biol. 118:110–128. DOI: 10.1016/j.matbio.2023.03.004. PMID: 36924903.
Article12. Zhong S, Guo H, Wang H, Xing D, Lu T, Yang J, Wang C. 2020; Apelin-13 alleviated cardiac fibrosis via inhibiting the PI3K/Akt pathway to attenuate oxidative stress in rats with myocardial infarction-induced heart failure. Biosci Rep. 40:BSR20200040. DOI: 10.1042/BSR20200040. PMID: 32207519. PMCID: PMC7133518.
Article13. Erasmus M, Samodien E, Lecour S, Cour M, Lorenzo O, Dludla P, Pheiffer C, Johnson R. 2020; Linking LOXL2 to cardiac interstitial fibrosis. Int J Mol Sci. 21:5913. DOI: 10.3390/ijms21165913. PMID: 32824630. PMCID: PMC7460598.
Article14. Tian X, Sun C, Wang X, Ma K, Chang Y, Guo Z, Si J. 2020; ANO1 regulates cardiac fibrosis via ATI-mediated MAPK pathway. Cell Calcium. 92:102306. DOI: 10.1016/j.ceca.2020.102306. PMID: 33075549.
Article15. Chen X, Su J, Feng J, Cheng L, Li Q, Qiu C, Zheng Q. 2019; TRIM72 contributes to cardiac fibrosis via regulating STAT3/Notch-1 signaling. J Cell Physiol. 234:17749–17756. DOI: 10.1002/jcp.28400. PMID: 30820965.
Article16. Tao H, Yang JJ, Shi KH, Li J. 2016; Wnt signaling pathway in cardiac fibrosis: new insights and directions. Metabolism. 65:30–40. DOI: 10.1016/j.metabol.2015.10.013. PMID: 26773927.
Article17. Alter C, Henseler AS, Owenier C, Hesse J, Ding Z, Lautwein T, Bahr J, Hayat S, Kramann R, Kostenis E, Scheller J, Schrader J. 2023; IL-6 in the infarcted heart is preferentially formed by fibroblasts and modulated by purinergic signaling. J Clin Invest. 133:e163799. DOI: 10.1172/JCI163799. PMID: 36943408. PMCID: PMC10232006.
Article18. Chou CH, Hung CS, Liao CW, Wei LH, Chen CW, Shun CT, Wen WF, Wan CH, Wu XM, Chang YY, Wu VC, Wu KD, Lin YH. TAIPAI Study Group. 2018; IL-6 trans-signalling contributes to aldosterone-induced cardiac fibrosis. Cardiovasc Res. 114:690–702. DOI: 10.1093/cvr/cvy013. PMID: 29360942.
Article19. Sun JY, Du LJ, Shi XR, Zhang YY, Liu Y, Wang YL, Chen BY, Liu T, Zhu H, Liu Y, Ruan CC, Gan Z, Ying H, Yin Z, Gao PJ, Yan X, Li RG, Duan SZ. 2023; An IL-6/STAT3/MR/FGF21 axis mediates heart-liver cross-talk after myocardial infarction. Sci Adv. 9:eade4110. DOI: 10.1126/sciadv.ade4110. PMID: 37018396. PMCID: PMC10075967.
Article20. Kleveland O, Kunszt G, Bratlie M, Ueland T, Broch K, Holte E, Michelsen AE, Bendz B, Amundsen BH, Espevik T, Aakhus S, Damås JK, Aukrust P, Wiseth R, Gullestad L. 2016; Effect of a single dose of the interleukin-6 receptor antagonist tocilizumab on inflammation and troponin T release in patients with non-ST-elevation myocardial infarction: a double-blind, randomized, placebo-controlled phase 2 trial. Eur Heart J. 37:2406–2413. DOI: 10.1093/eurheartj/ehw171. PMID: 27161611.
Article21. Kleveland O, Ueland T, Kunszt G, Bratlie M, Yndestad A, Broch K, Holte E, Ryan L, Amundsen BH, Bendz B, Aakhus S, Espevik T, Halvorsen B, Mollnes TE, Wiseth R, Gullestad L, Aukrust P, Damås JK. 2018; Interleukin-6 receptor inhibition with tocilizumab induces a selective and substantial increase in plasma IP-10 and MIP-1β in non-ST-elevation myocardial infarction. Int J Cardiol. 271:1–7. DOI: 10.1016/j.ijcard.2018.04.136. PMID: 29961572.
Article22. Garbers C, Hermanns HM, Schaper F, Müller-Newen G, Grötzinger J, Rose-John S, Scheller J. 2012; Plasticity and cross-talk of interleukin 6-type cytokines. Cytokine Growth Factor Rev. 23:85–97. DOI: 10.1016/j.cytogfr.2012.04.001. PMID: 22595692.
Article23. Ernst M, Najdovska M, Grail D, Lundgren-May T, Buchert M, Tye H, Matthews VB, Armes J, Bhathal PS, Hughes NR, Marcusson EG, Karras JG, Na S, Sedgwick JD, Hertzog PJ, Jenkins BJ. 2008; STAT3 and STAT1 mediate IL-11-dependent and inflammation-associated gastric tumorigenesis in gp130 receptor mutant mice. J Clin Invest. 118:1727–1738. DOI: 10.1172/JCI34944.
Article24. Tsujioka H, Kunieda T, Katou Y, Shirahige K, Fukazawa T, Kubo T. 2017; Interleukin-11 induces and maintains progenitors of different cell lineages during Xenopus tadpole tail regeneration. Nat Commun. 8:495. DOI: 10.1038/s41467-017-00594-5. PMID: 28887447. PMCID: PMC5591189.
Article25. Li Y, Luo C, Zeng Y, Zheng Z, Tao D, Liu Q, Hong Y, Wang S, Long H, Xu Z. 2023; Renal fibrosis is alleviated through targeted inhibition of IL-11-induced renal tubular epithelial-to-mesenchymal transition. Am J Pathol. 193:1936–1952. DOI: 10.1016/j.ajpath.2023.07.005. PMID: 37673330.
Article26. Jiang LF, Yang M, Meng HW, Jia PC, Du CL, Liu JY, Lv XW, Cheng-Huang , Li J. 2023; The effect of hepatic stellate cell derived-IL-11 on hepatocyte injury in hepatic fibrosis. Life Sci. 330:121974. DOI: 10.1016/j.lfs.2023.121974. PMID: 37495078.
Article27. Ma J, Xie Y, Xu Y, Gu P, Zhang Y, Fan L, Zhou Y, Wang H, Zhou T, He J, Wang D, Chen W. 2023; Neutralization of interleukin-11 attenuates silica particles-induced pulmonary inflammation and fibrosis in vivo. J Environ Sci (China). 126:772–783. DOI: 10.1016/j.jes.2022.03.015. PMID: 36503802.
Article28. Schafer S, Viswanathan S, Widjaja AA, Lim WW, Moreno-Moral A, DeLaughter DM, Ng B, Patone G, Chow K, Khin E, Tan J, Chothani SP, Ye L, Rackham OJL, Ko NSJ, Sahib NE, Pua CJ, Zhen NTG, Xie C, Wang M, et al. 2017; IL-11 is a crucial determinant of cardiovascular fibrosis. Nature. 552:110–115. DOI: 10.1038/nature24676. PMID: 29160304. PMCID: PMC5807082.
Article29. Chen Y, Wang L, Huang S, Ke J, Wang Q, Zhou Z, Chang W. 2021; Lutein attenuates angiotensin II- induced cardiac remodeling by inhibiting AP-1/IL-11 signaling. Redox Biol. 44:102020. DOI: 10.1016/j.redox.2021.102020. PMID: 34077894. PMCID: PMC8181194.
Article30. Widjaja AA, Singh BK, Adami E, Viswanathan S, Dong J, D'Agostino GA, Ng B, Lim WW, Tan J, Paleja BS, Tripathi M, Lim SY, Shekeran SG, Chothani SP, Rabes A, Sombetzki M, Bruinstroop E, Min LP, Sinha RA, Albani S, et al. 2019; Inhibiting interleukin 11 signaling reduces hepatocyte death and liver fibrosis, inflammation, and steatosis in mouse models of nonalcoholic steatohepatitis. Gastroenterology. 157:777–792.e14. DOI: 10.1053/j.gastro.2019.05.002. PMID: 31078624.
Article31. Ng B, Dong J, D'Agostino G, Viswanathan S, Widjaja AA, Lim WW, Ko NSJ, Tan J, Chothani SP, Huang B, Xie C, Pua CJ, Chacko AM, Guimarães-Camboa N, Evans SM, Byrne AJ, Maher TM, Liang J, Jiang D, Noble PW, et al. 2019; Interleukin-11 is a therapeutic target in idiopathic pulmonary fibrosis. Sci Transl Med. 11:eaaw1237. DOI: 10.1126/scitranslmed.aaw1237. PMID: 31554736.
Article32. Obana M, Maeda M, Takeda K, Hayama A, Mohri T, Yamashita T, Nakaoka Y, Komuro I, Takeda K, Matsumiya G, Azuma J, Fujio Y. 2010; Therapeutic activation of signal transducer and activator of transcription 3 by interleukin-11 ameliorates cardiac fibrosis after myocardial infarction. Circulation. 121:684–691. DOI: 10.1161/CIRCULATIONAHA.109.893677. PMID: 20100971.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- KLF9 deficiency protects the heart from inflammatory injury triggered by myocardial infarction
- Stem Cell Therapy for Ischemic Heart Disease: A Status Report
- Peiminine inhibits myocardial injury and fibrosis after myocardial infarction in rats by regulating mitogen-activated protein kinase pathway
- Crosstalk between the heart and peripheral organs in heart failure
- The Impact of Early Regular Cardiac Rehabilitation Program on Myocardial Function after Acute Myocardial Infarction