Korean J Pain.
2023 Oct;36(4):408-424. 10.3344/kjp.23220.
Repetitive transcranial magnetic stimulation in central post-stroke pain: current status and future perspective
- Affiliations
-
- 1Faculty of Medicine and Health, Institut Teknologi Sepuluh Nopember, Surabaya, Indonesia
- 2Department of Neurology, Faculty of Medicine, Universitas Airlangga – Dr. Soetomo General Academic Hospital, Surabaya, Indonesia
- KMID: 2546704
- DOI: http://doi.org/10.3344/kjp.23220
Abstract
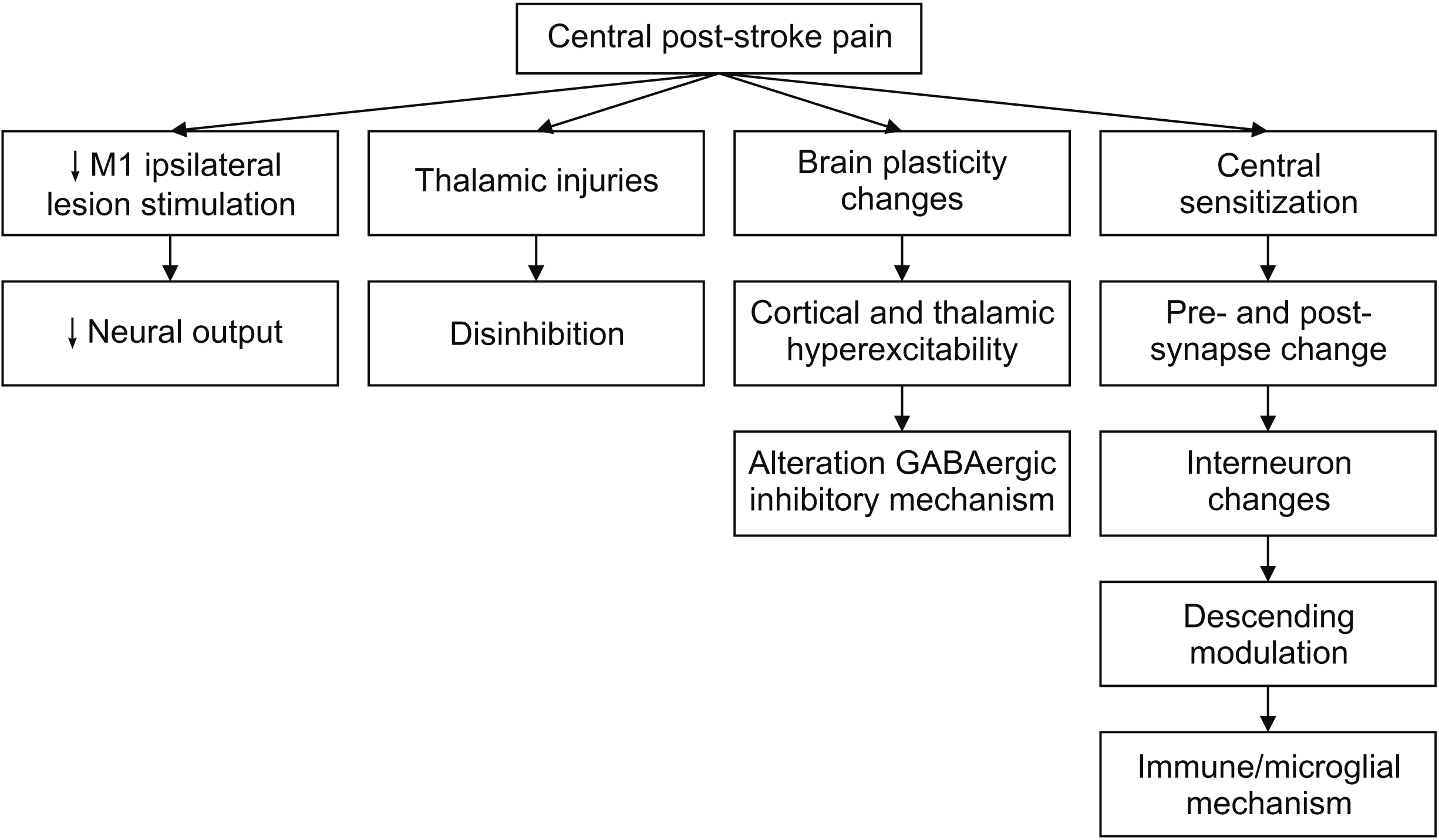
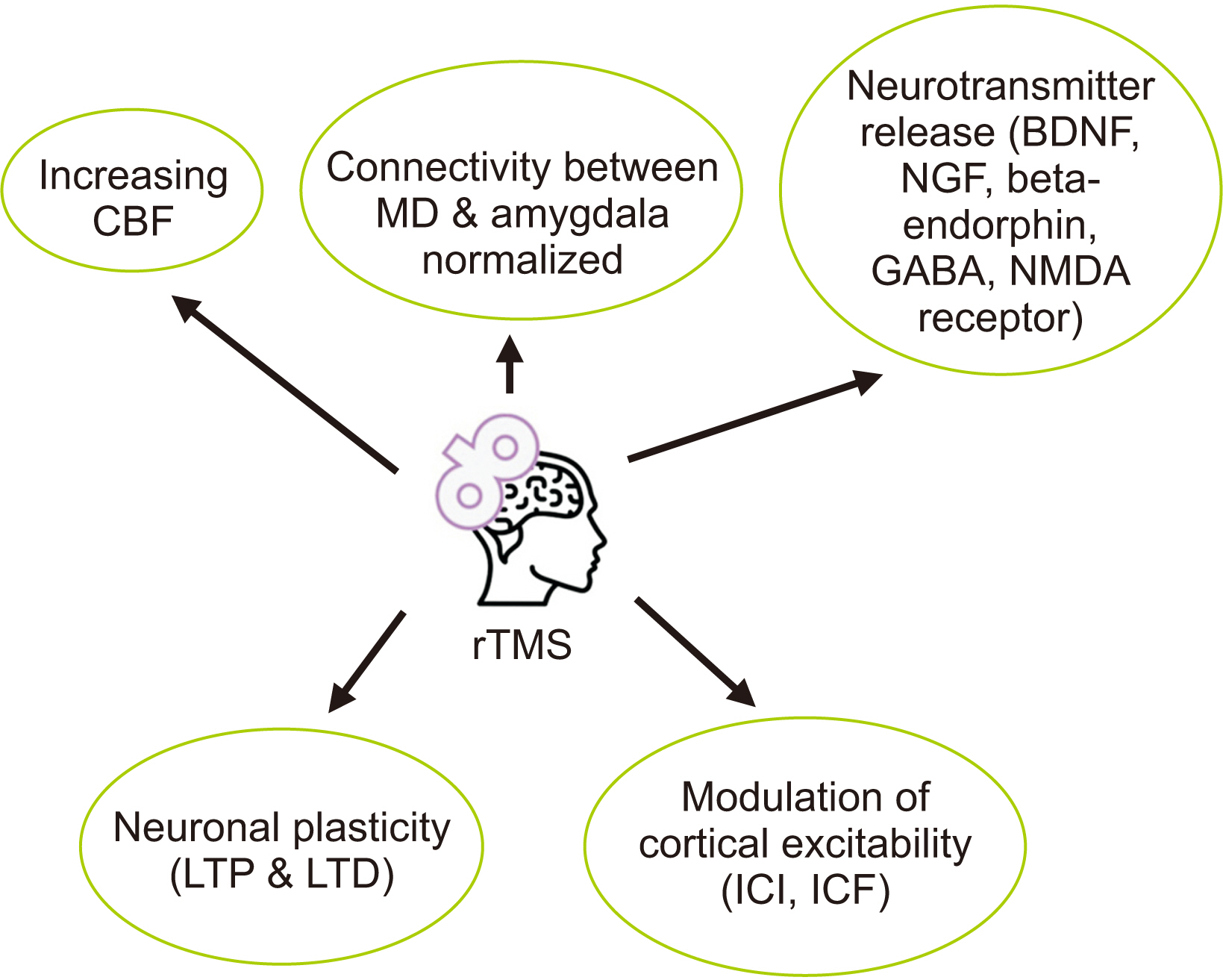
- Central post-stroke pain (CPSP) is an incapacitating disorder that impacts a substantial proportion of stroke survivors and can diminish their quality of life. Conventional therapies for CPSP, including tricyclic antidepressants, anticonvulsants, and opioids, are frequently ineffective, necessitating the investigation of alternative therapeutic strategies. Repetitive transcranial magnetic stimulation (rTMS) is now recognized as a promising noninvasive pain management method for CPSP. rTMS modulates neural activity through the administration of magnetic pulses to specific cortical regions. Trials analyzing the effects of rTMS on CPSP have generated various outcomes, but the evidence suggests possible analgesic benefits. In CPSP and other neuropathic pain conditions, high-frequency rTMS targeting the primary motor cortex (M1) with figure-eight coils has demonstrated significant pain alleviation. Due to its associaton with analgesic benefits, M1 is the most frequently targeted area. The duration and frequency of rTMS sessions, as well as the stimulation intensity, have been studied in an effort to optimize treatment outcomes. The short-term pain relief effects of rTMS have been observed, but the long-term effects (> 3 months) require further investigation. Aspects such as stimulation frequency, location, and treatment period can influence the efficacy of rTMS and ought to be considered while planning the procedure. Standardized guidelines for using rTMS in CPSP would optimize therapy protocols and improve patient outcomes. This review article provides an up-to-date overview of the incidence, clinical characteristics, outcome of rTMS in CPSP patients, and future perspective in the field.
Keyword
Figure
Reference
-
1. Jönsson AC, Lindgren I, Hallström B, Norrving B, Lindgren A. 2006; Prevalence and intensity of pain after stroke: a population based study focusing on patients' perspectives. J Neurol Neurosurg Psychiatry. 77:590–5. DOI: 10.1136/jnnp.2005.079145. PMID: 16354737. PMCID: PMC2117435.
Article2. Kong KH, Woon VC, Yang SY. 2004; Prevalence of chronic pain and its impact on health-related quality of life in stroke survivors. Arch Phys Med Rehabil. 85:35–40. DOI: 10.1016/S0003-9993(03)00369-1. PMID: 14970965.
Article3. Naess H, Lunde L, Brogger J. 2012; The effects of fatigue, pain, and depression on quality of life in ischemic stroke patients: the Bergen Stroke Study. Vasc Health Risk Manag. 8:407–13. DOI: 10.2147/VHRM.S32780. PMID: 22910531. PMCID: PMC3402053.
Article4. Oh H, Seo W. 2015; A comprehensive review of central post-stroke pain. Pain Manag Nurs. 16:804–18. DOI: 10.1016/j.pmn.2015.03.002. PMID: 25962545.
Article5. MacGowan DJ, Janal MN, Clark WC, Wharton RN, Lazar RM, Sacco RL, et al. 1997; Central poststroke pain and Wallenberg's lateral medullary infarction: frequency, character, and determinants in 63 patients. Neurology. 49:120–5. DOI: 10.1212/WNL.49.1.120. PMID: 9222179.
Article6. Klit H, Finnerup NB, Jensen TS. 2009; Central post-stroke pain: clinical characteristics, pathophysiology, and management. Lancet Neurol. 8:857–68. DOI: 10.1016/S1474-4422(09)70176-0. PMID: 19679277.
Article7. Lampl C, Yazdi K, Röper C. 2002; Amitriptyline in the prophylaxis of central poststroke pain. Preliminary results of 39 patients in a placebo-controlled, long-term study. Stroke. 33:3030–2. DOI: 10.1161/01.STR.0000037674.95228.86. PMID: 12468808.
Article8. Head H, Holmes G. 1911; Sensory disturbances from cerebral lesions. Brain. 34:102–254. DOI: 10.1093/brain/34.2-3.102.
Article9. Riddoch G. 1938; The clinical features of central pain. Lancet. 231:1205–9. DOI: 10.1016/S0140-6736(00)89785-8.
Article10. Loeser JD, Treede RD. 2008; The Kyoto protocol of IASP Basic Pain Terminology. Pain. 137:473–77. DOI: 10.1016/j.pain.2008.04.025. PMID: 18583048.11. Seifert CL, Mallar Chakravarty M, Sprenger T. 2013; The complexities of pain after stroke--a review with a focus on central post-stroke pain. Panminerva Med. 55:1–10. DOI: 10.1017/cbo9781139152211.015. PMID: 23474660.12. Andersen G, Vestergaard K, Ingeman-Nielsen M, Jensen TS. 1995; Incidence of central post-stroke pain. Pain. 61:187–93. DOI: 10.1016/0304-3959(94)00144-4. PMID: 7659428.
Article13. Finnerup NB, Otto M, McQuay HJ, Jensen TS, Sindrup SH. 2005; Algorithm for neuropathic pain treatment: an evidence based proposal. Pain. 118:289–305. DOI: 10.1016/j.pain.2005.08.013. PMID: 16213659.
Article14. Westlund KN. Benzon HT, Rathmell JP, Wu CL, Turk DC, Argoff CE, Hurley RW, editors. 2014. Pain pathways: peripheral, spinal, ascending, and descending pathways. Practical management of pain. 5th ed. Elsevier;p. 87–98.e5. DOI: 10.1016/B978-0-323-08340-9.00008-6.15. Sewards TV, Sewards MA. 2002; The medial pain system: neural representations of the motivational aspect of pain. Brain Res Bull. 59:163–80. DOI: 10.1016/S0361-9230(02)00864-X. PMID: 12431746.
Article16. Betancur DFA, Tarragó MDGL, Torres ILDS, Fregni F, Caumo W. 2021; Central post-stroke pain: an integrative review of somatotopic damage, clinical symptoms, and neurophysiological measures. Front Neurol. 12:678198. DOI: 10.3389/fneur.2021.678198. PMID: 34484097. PMCID: PMC8416310. PMID: 78dd962ce5cf4c3f95b1ede7099e80bd.
Article17. Gerges ANH, Hordacre B, Pietro FD, Moseley GL, Berryman C. 2022; Do adults with stroke have altered interhemispheric inhibition? A systematic review with meta-analysis. J Stroke Cerebrovasc Dis. 31:106494. DOI: 10.1016/j.jstrokecerebrovasdis.2022.106494. PMID: 35472653.
Article18. Craig AD. 1998; A new version of the thalamic disinhibition hypothesis of central pain. Pain Forum. 7:1–14. DOI: 10.1016/S1082-3174(98)70004-2.
Article19. Boivie J, Leijon G, Johansson I. 1989; Central post-stroke pain--a study of the mechanisms through analyses of the sensory abnormalities. Pain. 37:173–85. DOI: 10.1016/0304-3959(89)90128-0. PMID: 2748190.20. Vartiainen N, Perchet C, Magnin M, Creac'h C, Convers P, Nighoghossian N, et al. 2016; Thalamic pain: anatomical and physiological indices of prediction. Brain. 139:708–22. DOI: 10.1093/brain/awv389. PMID: 26912644.
Article21. Baliki MN, Schnitzer TJ, Bauer WR, Apkarian AV. 2011; Brain morphological signatures for chronic pain. PLoS One. 6:e26010. DOI: 10.1371/journal.pone.0026010. PMID: 22022493. PMCID: PMC3192794. PMID: b230784446e543b2b222d61a51094565.
Article22. Krause T, Asseyer S, Taskin B, Flöel A, Witte AV, Mueller K, et al. 2016; The cortical signature of central poststroke pain: gray matter decreases in somatosensory, insular, and prefrontal cortices. Cereb Cortex. 26:80–8. DOI: 10.1093/cercor/bhu177. PMID: 25129889.
Article23. Li X, Feng Y, Gao F. 2019; Maladaptive reorganization in pain-related brain network contributing to the central post-stroke pain. Neuropsychiatry (London). 9:2186–97.24. Gritsch S, Bali KK, Kuner R, Vardeh D. 2016; Functional characterization of a mouse model for central post-stroke pain. Mol Pain. 12:1744806916629049. DOI: 10.1177/1744806916629049. PMID: 27030713. PMCID: PMC4956143.
Article25. Kim JS. 1992; Pure sensory stroke. Clinical-radiological correlates of 21 cases. Stroke. 23:983–7. DOI: 10.1161/01.STR.23.7.983. PMID: 1615549.
Article26. Kim JH, Greenspan JD, Coghill RC, Ohara S, Lenz FA. 2007; Lesions limited to the human thalamic principal somatosensory nucleus (ventral caudal) are associated with loss of cold sensations and central pain. J Neurosci. 27:4995–5004. DOI: 10.1523/JNEUROSCI.0716-07.2007. PMID: 17475808. PMCID: PMC6672095.
Article27. Wang G, Thompson SM. 2008; Maladaptive homeostatic plasticity in a rodent model of central pain syndrome: thalamic hyperexcitability after spinothalamic tract lesions. J Neurosci. 28:11959–69. DOI: 10.1523/JNEUROSCI.3296-08.2008. PMID: 19005061. PMCID: PMC2627563.
Article28. Lenz FA, Weiss N, Ohara S, Lawson C, Greenspan JD. Hallett M, Phillips LH, Schomer DL, Massey JM, editors. 2004. The role of the thalamus in pain. Supplements to clinical neurophysiology. Elsevier;p. 50–61. DOI: 10.1016/S1567-424X(09)70342-3. PMID: 16106605.29. Kadono Y, Koguchi K, Okada KI, Hosomi K, Hiraishi M, Ueguchi T, et al. 2021; Repetitive transcranial magnetic stimulation restores altered functional connectivity of central poststroke pain model monkeys. Sci Rep. 11:6126. DOI: 10.1038/s41598-021-85409-w. PMID: 33731766. PMCID: PMC7969937. PMID: 180bae92094a4b86881262a4c95591e2.
Article30. Kuan YH, Shih HC, Shyu BC. 2018; Involvement of P2X7 receptors and BDNF in the pathogenesis of central poststroke pain. Adv Exp Med Biol. 1099:211–27. DOI: 10.1007/978-981-13-1756-9_18. PMID: 30306527.
Article31. Schwartzman RJ, Grothusen J, Kiefer TR, Rohr P. 2001; Neuropathic central pain: epidemiology, etiology, and treatment options. Arch Neurol. 58:1547–50. DOI: 10.1001/archneur.58.10.1547. PMID: 11594911.32. Latremoliere A, Woolf CJ. 2009; Central sensitization: a generator of pain hypersensitivity by central neural plasticity. J Pain. 10:895–926. DOI: 10.1016/j.jpain.2009.06.012. PMID: 19712899. PMCID: PMC2750819.
Article33. Campbell JN, Meyer RA. 2006; Mechanisms of neuropathic pain. Neuron. 52:77–92. DOI: 10.1016/j.neuron.2006.09.021. PMID: 17015228. PMCID: PMC1810425.
Article34. Kohno T, Ji RR, Ito N, Allchorne AJ, Befort K, Karchewski LA, et al. 2005; Peripheral axonal injury results in reduced mu opioid receptor pre- and post-synaptic action in the spinal cord. Pain. 117:77–87. DOI: 10.1016/j.pain.2005.05.035. PMID: 16098668.
Article35. Li CY, Song YH, Higuera ES, Luo ZD. 2004; Spinal dorsal horn calcium channel alpha2delta-1 subunit upregulation contributes to peripheral nerve injury-induced tactile allodynia. J Neurosci. 24:8494–9. DOI: 10.1523/JNEUROSCI.2982-04.2004. PMID: 15456823. PMCID: PMC1635787.36. Yoshimura M, Yonehara N. 2006; Alteration in sensitivity of ionotropic glutamate receptors and tachykinin receptors in spinal cord contribute to development and maintenance of nerve injury-evoked neuropathic pain. Neurosci Res. 56:21–8. DOI: 10.1016/j.neures.2006.04.015. PMID: 16901566.
Article37. Marchand F, Perretti M, McMahon SB. 2005; Role of the immune system in chronic pain. Nat Rev Neurosci. 6:521–32. DOI: 10.1038/nrn1700. PMID: 15995723.
Article38. Attal N, Fermanian C, Fermanian J, Lanteri-Minet M, Alchaar H, Bouhassira D. 2008; Neuropathic pain: are there distinct subtypes depending on the aetiology or anatomical lesion? Pain. 138:343–53. DOI: 10.1016/j.pain.2008.01.006. PMID: 18289791.
Article39. Hansen AP, Marcussen NS, Klit H, Andersen G, Finnerup NB, Jensen TS. 2012; Pain following stroke: a prospective study. Eur J Pain. 16:1128–36. DOI: 10.1002/j.1532-2149.2012.00123.x. PMID: 22407963.
Article40. Scholz J, Finnerup NB, Attal N, Aziz Q, Baron R, Bennett MI, et al. Classification Committee of the Neuropathic Pain Special Interest Group (NeuPSIG). 2019; The IASP classification of chronic pain for ICD-11: chronic neuropathic pain. Pain. 160:53–9. DOI: 10.1097/j.pain.0000000000001365. PMID: 30586071. PMCID: PMC6310153.
Article41. Kim MS, Kim BY, Saghetlians A, Zhang X, Okida T, Kim SY. 2022; Anti-nociceptive effects of dual neuropeptide antagonist therapy in mouse model of neuropathic and inflammatory pain. Korean J Pain. 35:173–82. DOI: 10.3344/kjp.2022.35.2.173. PMID: 35354680. PMCID: PMC8977203.
Article42. Obata H. 2017; Analgesic mechanisms of antidepressants for neuropathic pain. Int J Mol Sci. 18:2483. DOI: 10.3390/ijms18112483. PMID: 29160850. PMCID: PMC5713449. PMID: 3e077adbdbd5447ba5b53544596fceaf.
Article43. Dworkin RH, O'Connor AB, Backonja M, Farrar JT, Finnerup NB, Jensen TS, et al. 2007; Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain. 132:237–51. DOI: 10.1016/j.pain.2007.08.033. PMID: 17920770.
Article44. Offord J, Isom LL. 2015; Drugging the undruggable: gabapentin, pregabalin and the calcium channel α2δ subunit. Crit Rev Biochem Mol Biol. 51:246–56. DOI: 10.3109/10409238.2016.1173010. PMID: 27112431.45. Vranken JH, Dijkgraaf MG, Kruis MR, van der Vegt MH, Hollmann MW, Heesen M. 2008; Pregabalin in patients with central neuropathic pain: a randomized, double-blind, placebo-controlled trial of a flexible-dose regimen. Pain. 136:150–7. DOI: 10.1016/j.pain.2007.06.033. PMID: 17703885.
Article46. Stefani A, Spadoni F, Siniscalchi A, Bernardi G. 1996; Lamotrigine inhibits Ca2+ currents in cortical neurons: functional implications. Eur J Pharmacol. 307:113–6. DOI: 10.1016/0014-2999(96)00265-8. PMID: 8831112.
Article47. Sansone RA, Sansone LA. 2014; Serotonin norepinephrine reuptake inhibitors: a pharmacological comparison. Innov Clin Neurosci. 11:37–42. DOI: 10.4135/9781483365817.n1227. PMID: 24800132. PMCID: PMC4008300.48. Jo S, Bean BP. 2014; Sidedness of carbamazepine accessibility to voltage-gated sodium channels. Mol Pharmacol. 85:381–7. DOI: 10.1124/mol.113.090472. PMID: 24319110. PMCID: PMC3913360.
Article49. Edinoff AN, Kaplan LA, Khan S, Petersen M, Sauce E, Causey CD, et al. 2021; Full opioid agonists and tramadol: pharmacological and clinical considerations. Anesth Pain Med. 11:e119156. DOI: 10.5812/aapm.119156. PMID: 34692448. PMCID: PMC8520671.
Article50. Holbech JV, Jung A, Jonsson T, Wanning M, Bredahl C, Bach FW. 2017; Combination treatment of neuropathic pain: Danish expert recommendations based on a Delphi process. J Pain Res. 10:1467–75. DOI: 10.2147/JPR.S138099. PMID: 28721089. PMCID: PMC5499948.
Article51. Kim JS. 2014; Pharmacological management of central post-stroke pain: a practical guide. CNS Drugs. 28:787–97. DOI: 10.1007/s40263-014-0194-y. PMID: 25112542.
Article52. Banerjee M, Pal S, Bhattacharya B, Ghosh B, Mondal S, Basu J. 2013; A comparative study of efficacy and safety of gabapentin versus amitriptyline as coanalgesics in patients receiving opioid analgesics for neuropathic pain in malignancy. Indian J Pharmacol. 45:334–8. DOI: 10.4103/0253-7613.115000. PMID: 24014906. PMCID: PMC3757599.
Article53. Winstein CJ, Stein J, Arena R, Bates B, Cherney LR, Cramer SC, et al. American Heart Association Stroke Council. Council on Cardiovascular and Stroke Nursing. Council on Clinical Cardiology. Council on Quality of Care and Outcomes Research. 2016; Guidelines for adult stroke rehabilitation and recovery: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 47:e98–169. Erratum in: Stroke 2017; 48: e78. Erratum in: Stroke 2017; 48: e369. DOI: 10.1161/STR.0000000000000098. PMID: 27145936.54. Intercollegiate Stroke Working Party. 2016. National clinical guideline for stroke. 5th ed. Royal College of Physicians;p. 1–178. DOI: 10.7861/clinmedicine.17-5-478.55. Teasell R, Salbach NM, Foley N, Mountain A, Cameron JI, Jong A, et al. 2020; Canadian stroke best practice recommendations: rehabilitation, recovery, and community participation following stroke. Part one: rehabilitation and recovery following stroke;. 6th edition update 2019. Int J Stroke. 15:763–88. DOI: 10.1177/1747493019897843. PMID: 31983296.56. Iglesias AH. 2020; Transcranial magnetic stimulation as treatment in multiple neurologic conditions. Curr Neurol Neurosci Rep. 20:1. DOI: 10.1007/s11910-020-1021-0. PMID: 32020300.
Article57. Vidal-Dourado M, Conforto AB, Caboclo LO, Scaff M, Guilhoto LM, Yacubian EM. 2014; Magnetic fields in noninvasive brain stimulation. Neuroscientist. 20:112–21. DOI: 10.1177/1073858413491145. PMID: 23787954.
Article58. Diana M, Raij T, Melis M, Nummenmaa A, Leggio L, Bonci A. 2017; Rehabilitating the addicted brain with transcranial magnetic stimulation. Nat Rev Neurosci. 18:685–93. DOI: 10.1038/nrn.2017.113. PMID: 28951609.
Article59. Barker AT, Jalinous R, Freeston IL. 1985; Non-invasive magnetic stimulation of human motor cortex. Lancet. 1:1106–7. DOI: 10.1016/S0140-6736(85)92413-4. PMID: 2860322.
Article60. Marcos Z. 2013; Álvaro Pascual-Leone: a pioneer of non-invasive brain stimulation. Lancet Neurol. 12:853. DOI: 10.1016/S1474-4422(13)70129-7. PMID: 23759363.
Article61. Burke MJ, Fried PJ, Pascual-Leone A. 2019; Transcranial magnetic stimulation: Neurophysiological and clinical applications. Handb Clin Neurol. 163:73–92. DOI: 10.1016/B978-0-12-804281-6.00005-7. PMID: 31590749.
Article62. Hallett M. 2000; Transcranial magnetic stimulation and the human brain. Nature. 406:147–50. DOI: 10.1038/35018000. PMID: 10910346.
Article63. Di Lazzaro V, Profice P, Ranieri F, Capone F, Dileone M, Oliviero A, et al. 2012; I-wave origin and modulation. Brain Stimul. 5:512–25. DOI: 10.1016/j.brs.2011.07.008. PMID: 21962980.
Article64. Leung A, Donohue M, Xu R, Lee R, Lefaucheur JP, Khedr EM, et al. 2009; rTMS for suppressing neuropathic pain: a meta-analysis. J Pain. 10:1205–16. DOI: 10.1016/j.jpain.2009.03.010. PMID: 19464959.
Article65. Jin Y, Xing G, Li G, Wang A, Feng S, Tang Q, et al. 2015; High frequency repetitive transcranial magnetic stimulation therapy for chronic neuropathic pain: a meta-analysis. Pain Physician. 18:E1029–46. DOI: 10.36076/ppj.2015/18/E1029. PMID: 26606017.66. Pecuch PW, Evers S, Folkerts HW, Michael N, Arolt V. 2000; The cerebral hemodynamics of repetitive transcranial magnetic stimulation. Eur Arch Psychiatry Clin Neurosci. 250:320–4. DOI: 10.1007/s004060070007. PMID: 11153967.
Article67. Vernieri F, Maggio P, Tibuzzi F, Filippi MM, Pasqualetti P, Melgari JM, et al. 2009; High frequency repetitive transcranial magnetic stimulation decreases cerebral vasomotor reactivity. Clin Neurophysiol. 120:1188–94. DOI: 10.1016/j.clinph.2009.03.021. PMID: 19423387.
Article68. Ohn SH, Chang WH, Park CH, Kim ST, Lee JI, Pascual-Leone A, et al. 2012; Neural correlates of the antinociceptive effects of repetitive transcranial magnetic stimulation on central pain after stroke. Neurorehabil Neural Repair. 26:344–52. DOI: 10.1177/1545968311423110. PMID: 21980153. PMCID: PMC3541021.
Article69. Goto T, Saitoh Y, Hashimoto N, Hirata M, Kishima H, Oshino S, et al. 2008; Diffusion tensor fiber tracking in patients with central post-stroke pain; correlation with efficacy of repetitive transcranial magnetic stimulation. Pain. 140:509–18. DOI: 10.1016/j.pain.2008.10.009. PMID: 19004554.
Article70. Ahmed MA, Mohamed SA, Sayed D. 2011; Long-term antalgic effects of repetitive transcranial magnetic stimulation of motor cortex and serum beta-endorphin in patients with phantom pain. Neurol Res. 33:953–8. DOI: 10.1179/1743132811Y.0000000045. PMID: 22080997.
Article71. Pan LJ, Zhu HQ, Zhang XA, Wang XQ. 2023; The mechanism and effect of repetitive transcranial magnetic stimulation for post-stroke pain. Front Mol Neurosci. 15:1091402. DOI: 10.3389/fnmol.2022.1091402. PMID: 36683849. PMCID: PMC9855274. PMID: 6567f373f4d746649942446e61fd2c4e.
Article72. Lin RL, Douaud G, Filippini N, Okell TW, Stagg CJ, Tracey I. 2017; Structural connectivity variances underlie functional and behavioral changes during pain relief induced by neuromodulation. Sci Rep. 7:41603. DOI: 10.1038/srep41603. PMID: 28148969. PMCID: PMC5288647.
Article73. Mitchell AS, Chakraborty S. 2013; What does the mediodorsal thalamus do? Front Syst Neurosci. 7:37. DOI: 10.3389/fnsys.2013.00037. PMID: 23950738. PMCID: PMC3738868. PMID: 08259ac40f784cf394f03c14579af1d3.
Article74. Zhao CG, Sun W, Ju F, Jiang S, Wang H, Sun XL, et al. 2021; Analgesic effects of navigated repetitive transcranial magnetic stimulation in patients with acute central poststroke pain. Pain Ther. 10:1085–100. Erratum in: Pain Ther 2021; 10: 1101-3. DOI: 10.1007/s40122-021-00270-z. PMID: 34021496. PMCID: PMC8586304.
Article75. Garcia-Larrea L, Peyron R. 2007; Motor cortex stimulation for neuropathic pain: from phenomenology to mechanisms. Neuroimage. 37 Suppl 1:S71–9. DOI: 10.1016/j.neuroimage.2007.05.062. PMID: 17644413.
Article76. Attal N, Ayache SS, Ciampi De Andrade D, Mhalla A, Baudic S, Jazat F, et al. 2016; Repetitive transcranial magnetic stimulation and transcranial direct-current stimulation in neuropathic pain due to radiculopathy: a randomized sham-controlled comparative study. Pain. 157:1224–31. DOI: 10.1097/j.pain.0000000000000510. PMID: 26845524.77. Lindholm P, Lamusuo S, Taiminen T, Pesonen U, Lahti A, Virtanen A, et al. 2015; Right secondary somatosensory cortex-a promising novel target for the treatment of drug-resistant neuropathic orofacial pain with repetitive transcranial magnetic stimulation. Pain. 156:1276–83. DOI: 10.1097/j.pain.0000000000000175. PMID: 25830924.
Article78. Lefaucheur JP. 2006; The use of repetitive transcranial magnetic stimulation (rTMS) in chronic neuropathic pain. Neurophysiol Clin. 36:117–24. DOI: 10.1016/j.neucli.2006.08.002. PMID: 17046606.79. Kisler LB, Weissman-Fogel I, Sinai A, Sprecher E, Chistyakov AV, Shamay-Tsoory S, et al. 2017; Bi-phasic activation of the primary motor cortex by pain and its relation to pain-evoked potentials - an exploratory study. Behav Brain Res. 328:209–17. DOI: 10.1016/j.bbr.2017.04.006. PMID: 28389338.
Article80. Cha M, Ji Y, Masri R. 2013; Motor cortex stimulation activates the incertothalamic pathway in an animal model of spinal cord injury. J Pain. 14:260–9. DOI: 10.1016/j.jpain.2012.11.007. PMID: 23332495. PMCID: PMC3594418.
Article81. Yang L, Liu X, Yao K, Sun Y, Jiang F, Yan H, et al. 2019; HCN channel antagonist ZD7288 ameliorates neuropathic pain and associated depression. Brain Res. 1717:204–13. DOI: 10.1016/j.brainres.2019.03.036. PMID: 30940440.
Article82. Lanza G, Aricò D, Lanuzza B, Cosentino FII, Tripodi M, Giardina F, et al. 2020; Facilitatory/inhibitory intracortical imbalance in REM sleep behavior disorder: early electrophysiological marker of neurodegeneration? Sleep. 43:zsz242. DOI: 10.1093/sleep/zsz242. PMID: 31599326.
Article83. Hosomi K, Kishima H, Oshino S, Hirata M, Tani N, Maruo T, et al. 2013; Cortical excitability changes after high-frequency repetitive transcranial magnetic stimulation for central poststroke pain. Pain. 154:1352–7. DOI: 10.1016/j.pain.2013.04.017. PMID: 23707310.
Article84. Hoogendam JM, Ramakers GM, Di Lazzaro V. 2010; Physiology of repetitive transcranial magnetic stimulation of the human brain. Brain Stimul. 3:95–118. DOI: 10.1016/j.brs.2009.10.005. PMID: 20633438.
Article85. Cooke SF, Bliss TV. 2006; Plasticity in the human central nervous system. Brain. 129:1659–73. DOI: 10.1093/brain/awl082. PMID: 16672292.
Article86. Obata K, Yamanaka H, Dai Y, Tachibana T, Fukuoka T, Tokunaga A, et al. 2003; Differential activation of extracellular signal-regulated protein kinase in primary afferent neurons regulates brain-derived neurotrophic factor expression after peripheral inflammation and nerve injury. J Neurosci. 23:4117–26. DOI: 10.1523/JNEUROSCI.23-10-04117.2003. PMID: 12764099. PMCID: PMC6741084.
Article87. Ikeda K, Hazama K, Itano Y, Ouchida M, Nakatsuka H. 2020; Development of a novel analgesic for neuropathic pain targeting brain-derived neurotrophic factor. Biochem Biophys Res Commun. 531:390–5. DOI: 10.1016/j.bbrc.2020.07.109. PMID: 32800540.
Article88. Siuciak JA, Altar CA, Wiegand SJ, Lindsay RM. 1994; Antinociceptive effect of brain-derived neurotrophic factor and neurotrophin-3. Brain Res. 633:326–30. DOI: 10.1016/0006-8993(94)91556-3. PMID: 7511037.
Article89. Chail A, Saini RK, Bhat PS, Srivastava K, Chauhan V. 2018; Transcranial magnetic stimulation: a review of its evolution and current applications. Ind Psychiatry J. 27:172–80. DOI: 10.4103/ipj.ipj_88_18. PMID: 31359968. PMCID: PMC6592198.
Article90. Treister R, Lang M, Klein MM, Oaklander AL. 2013; Non-invasive transcranial magnetic stimulation (TMS) of the motor cortex for neuropathic pain-at the tipping point? Rambam Maimonides Med J. 4:e0023. DOI: 10.5041/RMMJ.10130. PMID: 24228166. PMCID: PMC3820296. PMID: 5eaa33864b6948479f6044dd70236fca.
Article91. Lin H, Li W, Ni J, Wang Y. 2018; Clinical study of repetitive transcranial magnetic stimulation of the motor cortex for thalamic pain. Medicine (Baltimore). 97:e11235. DOI: 10.1097/MD.0000000000011235. PMID: 29979386. PMCID: PMC6076143.
Article92. Najib U, Bashir S, Edwards D, Rotenberg A, Pascual-Leone A. 2011; Transcranial brain stimulation: clinical applications and future directions. Neurosurg Clin N Am. 22:233–51. DOI: 10.1016/j.nec.2011.01.002. PMID: 21435574. PMCID: PMC3547606.
Article93. Machado S, Arias-Carrión O, Paes F, Vieira RT, Caixeta L, Novaes F, et al. 2013; Repetitive transcranial magnetic stimulation for clinical applications in neurological and psychiatric disorders: an overview. Eurasian J Med. 45:191–206. DOI: 10.5152/eajm.2013.39. PMID: 25610279. PMCID: PMC4261426.
Article94. Kobayashi M, Fujimaki T, Mihara B, Ohira T. 2015; Repetitive transcranial magnetic stimulation once a week induces sustainable long-term relief of central poststroke pain. Neuromodulation. 18:249–54. DOI: 10.1111/ner.12301. PMID: 25906811.
Article95. Khedr EM, Kotb H, Kamel NF, Ahmed MA, Sadek R, Rothwell JC. 2005; Longlasting antalgic effects of daily sessions of repetitive transcranial magnetic stimulation in central and peripheral neuropathic pain. J Neurol Neurosurg Psychiatry. 76:833–8. DOI: 10.1136/jnnp.2004.055806. PMID: 15897507. PMCID: PMC1739662.
Article96. Alhassani G, Liston MB, Schabrun SM. 2019; Interhemispheric inhibition is reduced in response to acute muscle pain: a cross-sectional study using transcranial magnetic stimulation. J Pain. 20:1091–9. DOI: 10.1016/j.jpain.2019.03.007. PMID: 30904520.
Article97. Migita K, Uozumi T, Arita K, Monden S. 1995; Transcranial magnetic coil stimulation of motor cortex in patients with central pain. Neurosurgery. 36:1037–9. DOI: 10.1227/00006123-199505000-00025. PMID: 7540735.
Article98. Saitoh Y, Hirayama A, Kishima H, Shimokawa T, Oshino S, Hirata M, et al. 2007; Reduction of intractable deafferentation pain due to spinal cord or peripheral lesion by high-frequency repetitive transcranial magnetic stimulation of the primary motor cortex. J Neurosurg. 107:555–9. DOI: 10.3171/JNS-07/09/0555. PMID: 17886555.
Article99. Matsumura Y, Hirayama T, Yamamoto T. 2013; Comparison between pharmacologic evaluation and repetitive transcranial magnetic stimulation-induced analgesia in poststroke pain patients. Neuromodulation. 16:349–54. DOI: 10.1111/ner.12019. PMID: 23311356.
Article100. Ojala J, Vanhanen J, Harno H, Lioumis P, Vaalto S, Kaunisto MA, et al. 2022; A randomized, sham-controlled trial of repetitive transcranial magnetic stimulation targeting M1 and S2 in central poststroke pain: a pilot trial. Neuromodulation. 25:538–48. DOI: 10.1111/ner.13496. PMID: 35670063.
Article101. Hasan M, Whiteley J, Bresnahan R, MacIver K, Sacco P, Das K, et al. 2014; Somatosensory change and pain relief induced by repetitive transcranial magnetic stimulation in patients with central poststroke pain. Neuromodulation. 17:731–6. DOI: 10.1111/ner.12198. PMID: 24934719.
Article102. de Oliveira RA, de Andrade DC, Mendonça M, Barros R, Luvisoto T, Myczkowski ML, et al. 2014; Repetitive transcranial magnetic stimulation of the left premotor/dorsolateral prefrontal cortex does not have analgesic effect on central poststroke pain. J Pain. 15:1271–81. DOI: 10.1016/j.jpain.2014.09.009. PMID: 25267523.
Article103. Malfitano C, Rossetti A, Scarano S, Malloggi C, Tesio L. 2021; Efficacy of repetitive transcranial magnetic stimulation for acute central post-stroke pain: a case study. Front Neurol. 12:742567. DOI: 10.3389/fneur.2021.742567. PMID: 34858311. PMCID: PMC8631781. PMID: ac7f4668695044ab8ac9cb43c4b18cab.
Article104. Yang S, Chang MC. 2020; Effect of repetitive transcranial magnetic stimulation on pain management: a systematic narrative review. Front Neurol. 11:114. DOI: 10.3389/fneur.2020.00114. PMID: 32132973. PMCID: PMC7040236. PMID: 3ca78e41b4514a7f9015c4043e99bdbd.
Article105. Lefaucheur JP, Drouot X, Nguyen JP. 2001; Interventional neurophysiology for pain control: duration of pain relief following repetitive transcranial magnetic stimulation of the motor cortex. Neurophysiol Clin. 31:247–52. DOI: 10.1016/S0987-7053(01)00260-X. PMID: 11601430.
Article106. Cruccu G, Aziz TZ, Garcia-Larrea L, Hansson P, Jensen TS, Lefaucheur JP, et al. 2007; EFNS guidelines on neurostimulation therapy for neuropathic pain. Eur J Neurol. 14:952–70. DOI: 10.1111/j.1468-1331.2007.01916.x. PMID: 17718686.
Article107. Machii K, Cohen D, Ramos-Estebanez C, Pascual-Leone A. 2006; Safety of rTMS to non-motor cortical areas in healthy participants and patients. Clin Neurophysiol. 117:455–71. DOI: 10.1016/j.clinph.2005.10.014. PMID: 16387549.
Article108. Klomjai W, Katz R, Lackmy-Vallée A. 2015; Basic principles of transcranial magnetic stimulation (TMS) and repetitive TMS (rTMS). Ann Phys Rehabil Med. 58:208–13. DOI: 10.1016/j.rehab.2015.05.005. PMID: 26319963.
Article109. Rossini PM, Burke D, Chen R, Cohen LG, Daskalakis Z, Di Iorio R, et al. 2015; Non-invasive electrical and magnetic stimulation of the brain, spinal cord, roots and peripheral nerves: basic principles and procedures for routine clinical and research application. An updated report from an I.F.C.N. Committee. Clin Neurophysiol. 126:1071–107. DOI: 10.1016/j.clinph.2015.02.001. PMID: 25797650. PMCID: PMC6350257.
Article110. Wassermann EM. 1998; Risk and safety of repetitive transcranial magnetic stimulation: report and suggested guidelines from the International Workshop on the Safety of Repetitive Transcranial Magnetic Stimulation, June 5-7, 1996. Electroencephalogr Clin Neurophysiol. 108:1–16. DOI: 10.1016/S0168-5597(97)00096-8. PMID: 9474057.
Article111. Bae EH, Schrader LM, Machii K, Alonso-Alonso M, Riviello JJ Jr, Pascual-Leone A, et al. 2007; Safety and tolerability of repetitive transcranial magnetic stimulation in patients with epilepsy: a review of the literature. Epilepsy Behav. 10:521–8. DOI: 10.1016/j.yebeh.2007.03.004. PMID: 17493877.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Application of Non-invasive Brain Stimulation on Dysphagia after Stroke
- Stroke Update 2011: Stroke Rehabilitation
- Non-Invasive Brain Stimulation for Treatment of Focal Hand Dystonia: Update and Future Direction
- Repetitive transcranial magnetic stimulation for neuropathic pain
- Noninvasive brain stimulation: repetitive transcranial magnetic stimulation and transcranial direct current stimulation