Brain Tumor Res Treat.
2023 Apr;11(2):145-152. 10.14791/btrt.2023.0002.
Awake Craniotomy and Intraoperative Musical Performance for Brain Tumor Surgery: Case Report and Literature Review
- Affiliations
-
- 1Division of Neurosurgery, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, MA, USA
- KMID: 2542087
- DOI: http://doi.org/10.14791/btrt.2023.0002
Abstract
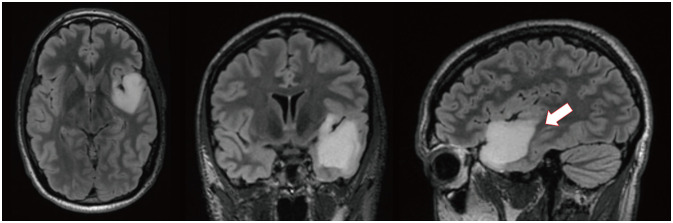
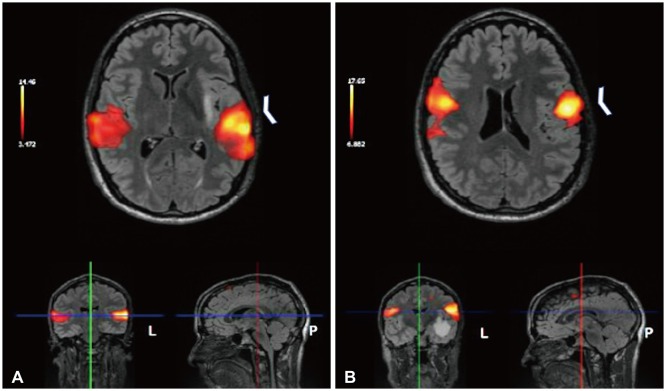
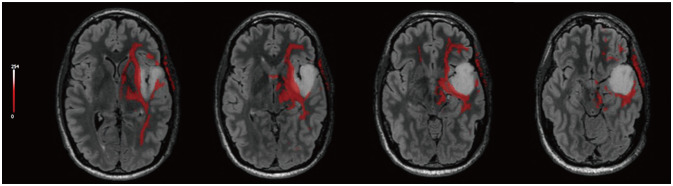
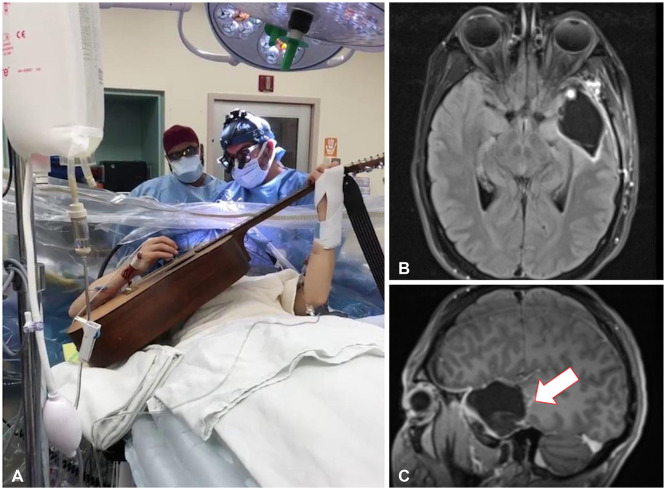
- Music experience and creation is a complex phenomenon that involves multiple brain structures. Music mapping during awake brain surgery, in addition to standard speech and motor mapping, remains a controversial topic. Music function can be impaired selectively, despite overlap with other neural networks commonly tested during direct cortical stimulation. We describe the case of a 34-year-old male patient presenting with a glioma located within eloquent cortex, who is also a professional musician and actor. We performed an awake craniotomy (AC) that mapped the standard motor and speech areas, while the patient played guitar intraoperatively and sang. Outcomes were remarkable with preservation of function and noted improvements in his musical abilities in outpatient follow-up. In addition, we performed a review of the literature in which awake craniotomies were performed for the removal of brain tumors in patients with some background in music (e.g., score reading, humming/singing). To date, only 4 patients have played a musical instrument intraoperatively during an AC for brain tumor resection. Using awake cortical mapping techniques and paradigms for preserving speech function during an intraoperative musical performance with singing is feasible and can yield a great result for patients. The use of standard brain mapping over music processing mapping did not yield a negative outcome. More experience is needed to understand and standardize this procedure as the field of brain mapping continues to grow for tumor resections.
Keyword
Figure
Reference
-
1. Saito Y, Ishii K, Yagi K, Tatsumi IF, Mizusawa H. Cerebral networks for spontaneous and synchronized singing and speaking. Neuroreport. 2006; 17:1893–1897. PMID: 17179865.2. Dalla Bella S, Berkowska M. Singing and its neuronal substrates: evidence from the general population. Contemp Music Rev. 2009; 28:279–291.3. Zarate JM. The neural control of singing. Front Hum Neurosci. 2013; 7:237. PMID: 23761746.4. Zhang J, Lu JF, Wu JS, Yao CJ, Zhuang DX, Qiu TM, et al. A unique case of Chinese language and music dissociation with tumor located in Broca’s area: multimodal mapping for tumor resection and functional preservation. Clin Neurol Neurosurg. 2013; 115:2230–2233. PMID: 23910999.5. Fisher CA, Larner AJ. Jean Langlais (1907-91): an historical case of a blind organist with stroke-induced aphasia and Braille alexia but without amusia. J Med Biogr. 2008; 16:232–234. PMID: 18952998.6. Russell SM, Golfinos JG. Amusia following resection of a Heschl gyrus glioma. Case report. J Neurosurg. 2003; 98:1109–1112. PMID: 12744373.7. Rogalsky C, Rong F, Saberi K, Hickok G. Functional anatomy of language and music perception: temporal and structural factors investigated using functional magnetic resonance imaging. J Neurosci. 2011; 31:3843–3852. PMID: 21389239.8. Kappen PR, Beshay T, Vincent AJPE, Satoer D, Dirven CMF, Jeekel J, et al. The feasibility and added value of mapping music during awake craniotomy: a systematic review. Eur J Neurosci. 2022; 55:388–404. PMID: 34894015.9. Scerrati A, Labanti S, Lofrese G, Mongardi L, Cavallo MA, Ricciardi L, et al. Artists playing music while undergoing brain surgery: a look into the scientific evidence and the social media perspective. Clin Neurol Neurosurg. 2020; 196:105911. PMID: 32505870.10. Scerrati A, Mongardi L, Cavallo MA, Labanti S, Simioni V, Ricciardi L, et al. Awake surgery for skills preservation during a sensory area tumor resection in a clarinet player. Acta Neurol Belg. 2021; 121:1235–1239. PMID: 32372400.11. Bass DI, Shurtleff H, Warner M, Knott D, Poliakov A, Friedman S, et al. Awake mapping of the auditory cortex during tumor resection in an aspiring musical performer: a case report. Pediatr Neurosurg. 2020; 55:351–358. PMID: 33260181.12. Garcea FE, Chernoff BL, Diamond B, Lewis W, Sims MH, Tomlinson SB, et al. Direct electrical stimulation in the human brain disrupts melody processing. Curr Biol. 2017; 27:2684–2691.e7. PMID: 28844645.13. Herbet G, Lafargue G, Almairac F, Moritz-Gasser S, Bonnetblanc F, Duffau H. Disrupting the right pars opercularis with electrical stimulation frees the song: case report. J Neurosurg. 2015; 123:1401–1404. PMID: 26140496.14. Nathoo N, Nair D, Phillips M, Vogelbaum MA. Mapping prosody: correlation of functional magnetic resonance imaging with intraoperative electrocorticography recordings in a patient with a right-sided temporooccipital glioma. Case illustration. J Neurosurg. 2005; 103:930. PMID: 16305000.15. Piai V, Vos SH, Idelberger R, Gans P, Doorduin J, Ter Laan M. Awake surgery for a violin player: monitoring motor and music performance, a case report. Arch Clin Neuropsychol. 2019; 34:132–137. PMID: 29490011.16. Riva M, Casarotti A, Comi A, Pessina F, Bello L. Brain and music: an intraoperative stimulation mapping study of a professional opera singer. World Neurosurg. 2016; 93:486.e13–486.e18.17. Roux FE, Borsa S, Demonet JF. “The mute who can sing”: a cortical stimulation study on singing. J Neurosurg. 2009; 110:282–288. PMID: 18821833.18. Roux FE, Lubrano V, Lotterie JA, Giussani C, Pierroux C, Demonet JF. When “abegg” is read and (“A, B, E, G, G”) is not: a cortical stimulation study of musical score reading. J Neurosurg. 2007; 106:1017–1027. PMID: 17564174.19. Leonard MK, Desai M, Hungate D, Cai R, Singhal NS, Knowlton RC, et al. Direct cortical stimulation of inferior frontal cortex disrupts both speech and music production in highly trained musicians. Cogn Neuropsychol. 2019; 36:158–166. PMID: 29786470.20. Cargnelutti E, Ius T, Skrap M, Tomasino B. What do we know about pre- and postoperative plasticity in patients with glioma? A review of neuroimaging and intraoperative mapping studies. Neuroimage Clin. 2020; 28:102435. PMID: 32980599.21. Sanai N, Berger MS. Operative techniques for gliomas and the value of extent of resection. Neurotherapeutics. 2009; 6:478–486. PMID: 19560738.22. Papatzalas C, Fountas K, Kapsalaki E, Papathanasiou I. The use of standardized intraoperative language tests in awake craniotomies: a scoping review. Neuropsychol Rev. 2022; 32:20–50. PMID: 33786797.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Anesthetic considerations for awake craniotomy
- Anesthetic management with propofol/remifentanil target controlled infusion for awake craniotomy: A case report
- Awake Surgery for Lesional Epilepsy in Resource-Limited Settings: Case Report and Review of Literature
- Development of a contralateral acute subdural hematoma during awake craniotomy for glial tumor in a 12-year-old boy: A case report
- Anesthetic management with scalp nerve block and propofol/remifentanil infusion during awake craniotomy in an adolescent patient: A case report