Brain Tumor Res Treat.
2023 Jan;11(1):59-65. 10.14791/btrt.2022.0036.
MRI-Based Classification of Rathke’s Cleft Cyst and Its Clinical Implication
- Affiliations
-
- 1Department of Neurosurgery, Yonsei University College of Medicine, Seoul, Korea
- 2Pituitary Tumor Center, Severance Hospital, Seoul, Korea
- 3Yonsei Endocrine Research Institute, Yonsei University College of Medicine, Seoul, Korea
- 4Department of Neurosurgery, Ewha Woman’s University College of Medicine, Seoul, Korea
- KMID: 2539330
- DOI: http://doi.org/10.14791/btrt.2022.0036
Abstract
- Background
Rathke’s cleft cysts (RCCs) are benign tumors of the pituitary gland. Small, asymptomatic RCCs do not require surgical treatment, whereas surgical treatment is required for symptomatic RCCs.
Methods
We retrospectively reviewed medical records of patients with an RCC who were diagnosed and managed in our institution between April 2004 and April 2020 and generated two different cohorts: the observation (n=114) and the surgical group (n=99). Their initial MRI signal characteristics were analyzed. The natural course focusing on cyst size was observed in the observation group and postoperative visual and endocrine outcomes were evaluated in the surgical group.
Results
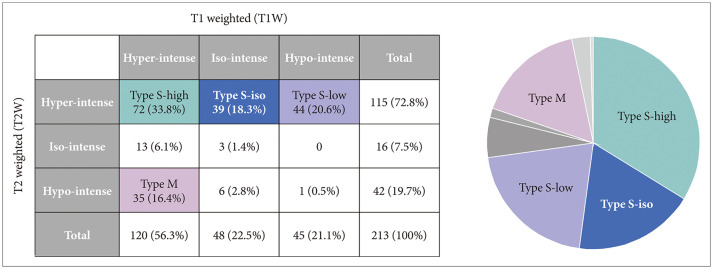
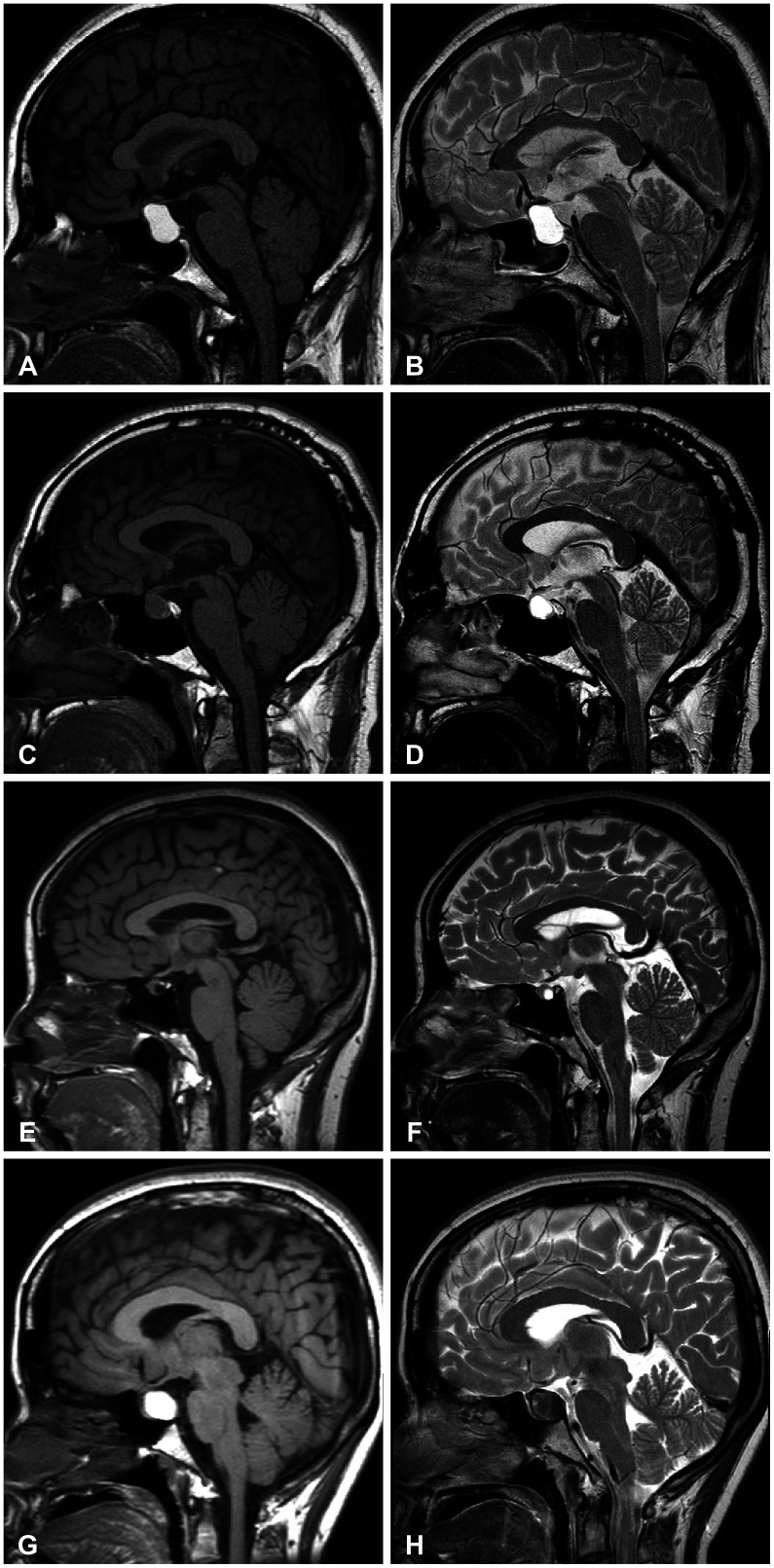
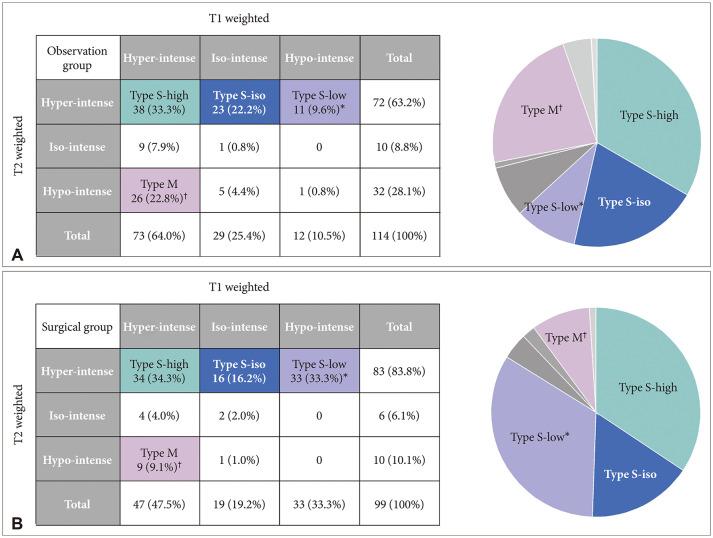
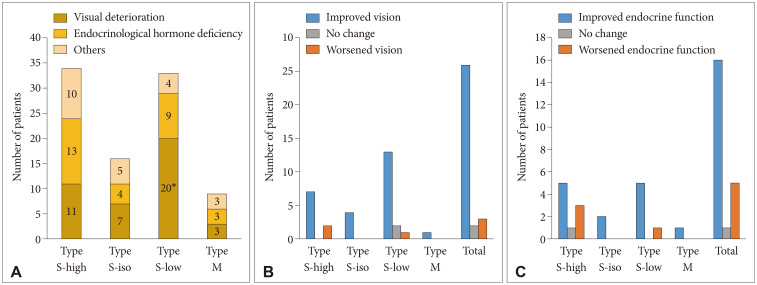
The characterization of MRI signals of cyst contents in both T1-weighted (T1W) and T2-weighted (T2W) images revealed nine combinations for our 213 patients. Among 115 patients with a high T2W signal, the cysts showed hypo-, iso-, and hyper-intensity on T1W images in 72, 39, and 44 patients, respectively; Type S-low, Type S-iso, and Type S-high. One more major group of 35 patients showed RCCs with hyperintensity on the T1W images and hypointensity on the T2W images named as Type M. In the comparison between observation and surgical groups, we identified only two major groups in which the number of patients in the surgical and observation groups was statistically different: more Type S-low in a surgical group (p<0.001) and more Type M in an observation group (p=0.007). In subgroup analysis, the range of change in the cyst size was the highest in Type S-high in the observation group (p=0.028), and intergroup differences in visual and endocrine outcomes were not evident in the surgical group.
Conclusion
MRI characteristics help to predict the natural course of RCCs. We identified subgroups of RCCs which are more or less likely to require surgical intervention.
Figure
Reference
-
1. Karavitaki N, Wass JA. Non-adenomatous pituitary tumours. Best Pract Res Clin Endocrinol Metab. 2009; 23:651–665. PMID: 19945029.
Article2. Wen L, Hu LB, Feng XY, Desai G, Zou LG, Wang WX, et al. Rathke’s cleft cyst: clinicopathological and MRI findings in 22 patients. Clin Radiol. 2010; 65:47–55. PMID: 20103421.
Article3. Trifanescu R, Ansorge O, Wass JA, Grossman AB, Karavitaki N. Rathke’s cleft cysts. Clin Endocrinol (Oxf). 2012; 76:151–160. PMID: 21951110.
Article4. Osborn AG, Preece MT. Intracranial cysts: radiologic-pathologic correlation and imaging approach. Radiology. 2006; 239:650–664. PMID: 16714456.
Article5. Eguchi K, Uozumi T, Arita K, Kurisu K, Yano T, Sumida M, et al. Pituitary function in patients with Rathke’s cleft cyst: significance of surgical management. Endocr J. 1994; 41:535–540. PMID: 7889113.
Article6. Mukherjee JJ, Islam N, Kaltsas G, Lowe DG, Charlesworth M, Afshar F, et al. Clinical, radiological and pathological features of patients with Rathke’s cleft cysts: tumors that may recur. J Clin Endocrinol Metab. 1997; 82:2357–2362. PMID: 9215319.7. Kim JE, Kim JH, Kim OL, Paek SH, Kim DG, Chi JG, et al. Surgical treatment of symptomatic Rathke cleft cysts: clinical features and results with special attention to recurrence. J Neurosurg. 2004; 100:33–40. PMID: 14743909.8. Nishioka H, Haraoka J, Izawa H, Ikeda Y. Magnetic resonance imaging, clinical manifestations, and management of Rathke’s cleft cyst. Clin Endocrinol (Oxf). 2006; 64:184–188. PMID: 16430718.
Article9. Choi SH, Kwon BJ, Na DG, Kim JH, Han MH, Chang KH. Pituitary adenoma, craniopharyngioma, and Rathke cleft cyst involving both intrasellar and suprasellar regions: differentiation using MRI. Clin Radiol. 2007; 62:453–462. PMID: 17398271.
Article10. Hayashi Y, Tachibana O, Muramatsu N, Tsuchiya H, Tada M, Arakawa Y, et al. Rathke cleft cyst: MR and biomedical analysis of cyst content. J Comput Assist Tomogr. 1999; 23:34–38. PMID: 10050804.
Article11. Byun WM, Kim OL, Kim D. MR imaging findings of Rathke’s cleft cysts: significance of intracystic nodules. AJNR Am J Neuroradiol. 2000; 21:485–488. PMID: 10730639.12. Kim EH, Oh MC, Chang JH, Moon JH, Ku CR, Chang WS, et al. Postoperative gamma knife radiosurgery for cavernous sinus-invading growth hormone-secreting pituitary adenomas. World Neurosurg. 2018; 110:e534–e545. PMID: 29155347.
Article13. Chung YS, Na M, Yoo J, Kim W, Jung IH, Moon JH, et al. Optical coherent tomography predicts long-term visual outcome of pituitary adenoma surgery: new perspectives from a 5-year follow-up study. Neurosurgery. 2021; 88:106–112.14. Cheng L, Guo P, Jin P, Li H, Fan M, Cai E. Spontaneous involution of a Rathke cleft cyst. J Craniofac Surg. 2016; 27:e791–e793. PMID: 28005827.
Article15. Salle L, Teissier-Clément MP, Mas R, Boncoeur-Martel MP, Salle H. Spontaneous involution of a Rathke cleft cyst. Ann Endocrinol (Paris). 2021; 82:626–628. PMID: 34672996.
Article16. Mendelson ZS, Husain Q, Elmoursi S, Svider PF, Eloy JA, Liu JK. Rathke’s cleft cyst recurrence after transsphenoidal surgery: a meta-analysis of 1151 cases. J Clin Neurosci. 2014; 21:378–385. PMID: 24269553.
Article17. Park JK, Lee EJ, Kim SH. Optimal surgical approaches for Rathke cleft cyst with consideration of endocrine function. Oper Neurosurg. 2012; 70:ons250–ons257.18. Hama S, Arita K, Nishisaka T, Fukuhara T, Tominaga A, Sugiyama K, et al. Changes in the epithelium of Rathke cleft cyst associated with inflammation. J Neurosurg. 2002; 96:209–216. PMID: 11838792.19. Potts MB, Jahangiri A, Lamborn KR, Blevins LS, Kunwar S, Aghi MK. Suprasellar Rathke cleft cysts: clinical presentation and treatment outcomes. Neurosurgery. 2011; 69:1058–1069. PMID: 21673610.20. Ozoner B, Aydin S, Akgun MY, Durmaz ES, Sahin S, Gazioglu N, et al. Predictive factors for Rathke’s cleft cyst consistency. World Neurosurg. 2019; 128:e522–e530. PMID: 31048050.
Article21. Schmidt B, Cattin F, Aubry S. Prevalence of Rathke cleft cysts in children on magnetic resonance imaging. Diagn Interv Imaging. 2020; 101:209–215. PMID: 31953055.22. Sade B, Albrecht S, Assimakopoulos P, Vézina JL, Mohr G. Management of Rathke’s cleft cysts. Surg Neurol. 2005; 63:459–466. PMID: 15883073.23. Voelker JL, Campbell RL, Muller J. Clinical, radiographic, and pathological features of symptomatic Rathke’s cleft cysts. J Neurosurg. 1991; 74:535–544. PMID: 2002366.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Symptomatic Rathke's Cleft Cyst in the Interpeduncular Cistern: Case Report
- Suprasellar Rathke Cleft Cyst: A case report
- Rathke's Cleft Cyst: Case Report
- Large Ossified Rathke's Cleft Cyst: A Case Report and Review of the Literature
- A Rathke's Cleft Cyst with Chronic Inflammation Mimicking a Pituitary Macroadenoma