Lab Med Online.
2022 Apr;12(2):134-137. 10.47429/lmo.2022.12.2.134.
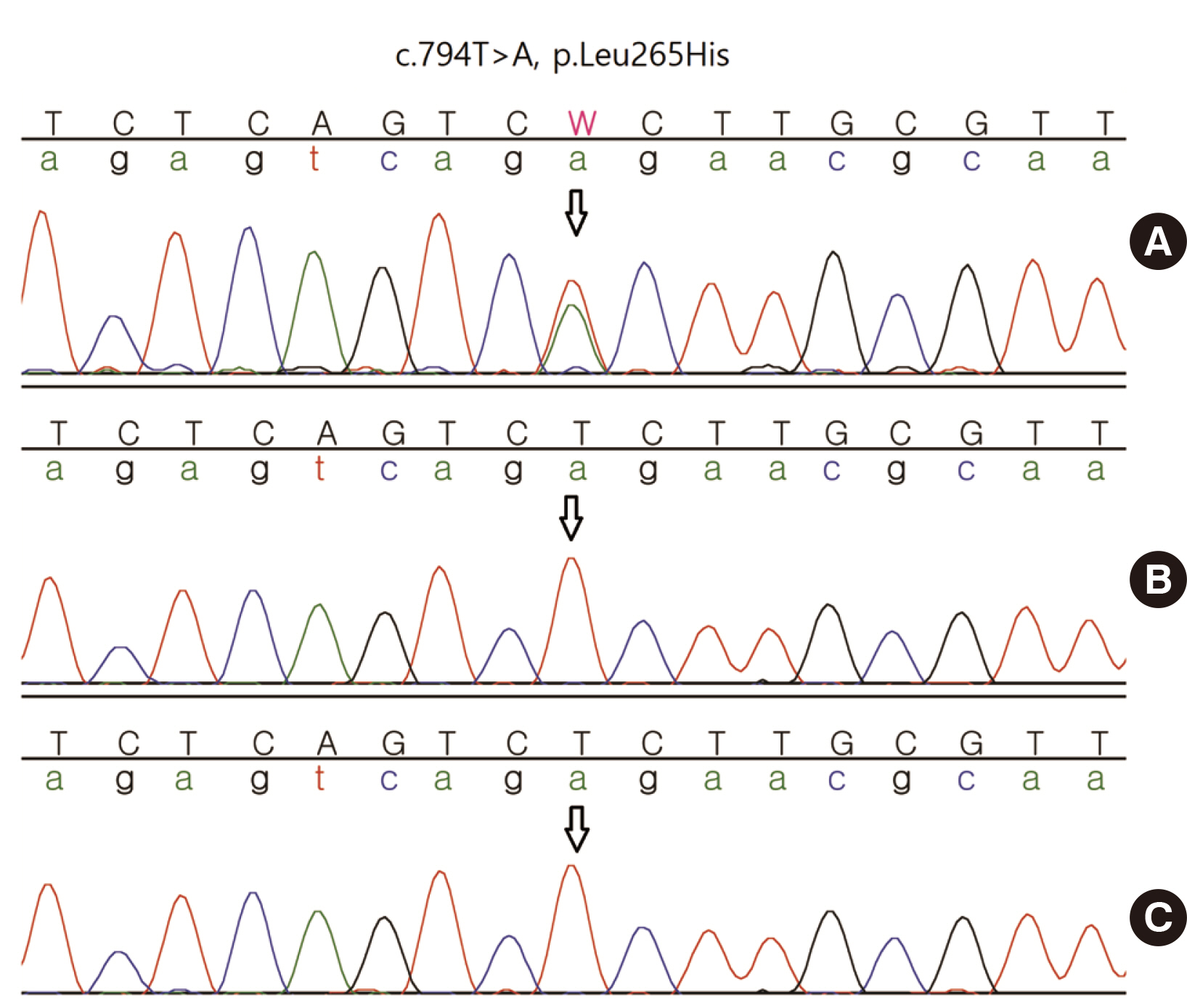
De novo HCN1 Mutation Identified by Next-Generation Sequencing in a Patient with Early Infantile Epileptic Encephalopathy: Case Report
- Affiliations
-
- 1Department of Laboratory Medicine, Yonsei University College of Medicine, Seoul, Korea
- 2Division of Pediatric Neurology, Department of Pediatrics, Yonsei University College of Medicine, Epilepsy Research Institute, Seoul, Korea
- KMID: 2538597
- DOI: http://doi.org/10.47429/lmo.2022.12.2.134
Abstract
- Early infantile epileptic encephalopathy (EIEE) is a group of brain disorders characterized by spontaneous seizures occurring at an early age. EIEE is caused by mutations in various genes, including HCN1. Here, we report a Korean patient with EIEE carrying a de novo mutation in the HCN1 gene. A female infant with unremarkable birth and family history showed status epilepticus four months after birth. Next-generation sequencing analysis revealed a heterozygous missense mutation, c.794T>A(p.Leu265His), in HCN1. The variant was not observed in any population control dataset, and in silico pathogenicity analyses predicted the variant to be pathogenic. Subsequent family testing by Sanger sequencing confirmed that the variant was a de novo mutation. These findings provide insights for predicting patient prognosis and the possibility of developing targeted therapy.
Keyword
Figure
Reference
-
1. Depienne C, Gourfinkel-An I, et al. Noebels JL, Avoli M, editors. 2012. Genes in infantile epileptic encephalopathies. Jasper’s basic mechanisms of the epilepsies. 4th ed. National Center for Biotechnology Information (US);Bethesda, MD: DOI: 10.1093/med/9780199746545.003.0062.
Article2. Veeramah KR, Johnstone L, Karafet TM, Wolf D, Sprissler R, Salogiannis J, et al. 2013; Exome sequencing reveals new causal mutations in children with epileptic encephalopathies. Epilepsia. 54:1270–81. DOI: 10.1111/epi.12201. PMID: 23647072. PMCID: PMC3700577.
Article3. Kase D, Imoto K. 2012; The role of HCN channels on membrane excitability in the nervous system. J Signal Transduct. 2012:619747. DOI: 10.1155/2012/619747. PMID: 22934165. PMCID: PMC3425855.
Article4. Tanguay J, Callahan KM, D'Avanzo N. 2019; Characterization of drug binding within the HCN1 channel pore. Sci Rep. 9:465. DOI: 10.1038/s41598-018-37116-2. PMID: 30679654. PMCID: PMC6345760.
Article5. Santoro B, Chen S, Luthi A, Pavlidis P, Shumyatsky GP, Tibbs GR, et al. 2000; Molecular and functional heterogeneity of hyperpolarization-activated pacemaker channels in the mouse CNS. J Neurosci. 20:5264–75. DOI: 10.1523/JNEUROSCI.20-14-05264.2000. PMID: 10884310. PMCID: PMC6772310.
Article6. Notomi T, Shigemoto R. 2004; Immunohistochemical localization of Ih channel subunits, HCN1-4, in the rat brain. J Comp Neurol. 471:241–76. DOI: 10.1002/cne.11039. PMID: 14991560.
Article7. Kole MH, Bräuer AU, Stuart GJ. 2007; Inherited cortical HCN1 channel loss amplifies dendritic calcium electrogenesis and burst firing in a rat absence epilepsy model. J Physiol. 578:507–25. DOI: 10.1113/jphysiol.2006.122028. PMID: 17095562. PMCID: PMC2075144.
Article8. Santoro B, Lee JY, Englot DJ, Gildersleeve S, Piskorowski RA, Siegelbaum SA, et al. 2010; Increased seizure severity and seizure-related death in mice lacking HCN1 channels. Epilepsia. 51:1624–7. DOI: 10.1111/j.1528-1167.2010.02554.x. PMID: 20384728. PMCID: PMC2952649.
Article9. Nava C, Dalle C, Rastetter A, Striano P, de Kovel CG, Nabbout R, et al. 2014; De novo mutations in HCN1 cause early infantile epileptic encephalopathy. Nat Genet. 46:640–5. DOI: 10.1038/ng.2952. PMID: 24747641.
Article10. Poduri A. 2014; HCN1 gain-of-function mutations - A new cause of epileptic encephalopathy. Epilepsy Curr. 14:348–9. DOI: 10.5698/1535-7597-14.6.348. PMID: 25678871. PMCID: PMC4325594.
Article11. Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. 2015; Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 17:405–24. DOI: 10.1038/gim.2015.30. PMID: 25741868. PMCID: PMC4544753.
Article12. Noam Y, Bernard C, Baram TZ. 2011; Towards an integrated view of HCN channel role in epilepsy. Curr Opin Neurobiol. 21:873–9. DOI: 10.1016/j.conb.2011.06.013. PMID: 21782415. PMCID: PMC3235400.
Article13. Herrmann S, Stieber J, Ludwig A. 2007; Pathophysiology of HCN channels. Pflugers Arch. 454:517–22. DOI: 10.1007/s00424-007-0224-4. PMID: 17549513.
Article14. Aggarwal SK, Mackinnon R. 1996; Contribution of the S4 segment to gating charge in the Shaker K+ channel. Neuron. 16:1169–77. DOI: 10.1016/S0896-6273(00)80143-9. PMID: 8663993.
Article15. Postea O, Biel M. 2011; Exploring HCN channels as novel drug targets. Nat Rev Drug Discov. 10:903–14. DOI: 10.1038/nrd3576. PMID: 22094868.
Article16. Surges R, Freiman TM, Feuerstein TJ. 2003; Gabapentin increases the hyperpolarization-activated cation current Ih in rat CA1 pyramidal cells. Epilepsia. 44:150–6. DOI: 10.1046/j.1528-1157.2003.36802.x. PMID: 12558567.
Article17. Poolos NP, Migliore M, Johnston D. 2002; Pharmacological upregulation of h-channels reduces the excitability of pyramidal neuron dendrites. Nat Neurosci. 5:767–74. DOI: 10.1038/nn891. PMID: 12118259.
Article18. Kitayama M, Miyata H, Yano M, Saito N, Matsuda Y, Yamauchi T, et al. 2003; Ih blockers have a potential of antiepileptic effects. Epilepsia. 44:20–4. DOI: 10.1046/j.1528-1157.2003.22702.x. PMID: 12581225.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A case of early infantile epileptic encephalopathy
- Ketogenic Diet in Infants with Early-Onset Epileptic Encephalopathy and SCN2A Mutation
- Early-onset epileptic encephalopathies and the diagnostic approach to underlying causes
- A Neonate with a PIGA c.1234C>T Mutation as a Novel Cause of Neonatal Early Infantile Epileptic Encephalopathy
- Identification of a de novo ANK1 Variant in a Patient with Hereditary Spherocytosis on Multi-gene Panel Testing