Korean J Physiol Pharmacol.
2023 Jan;27(1):95-103. 10.4196/kjpp.2023.27.1.95.
Effects of rosiglitazone, an antidiabetic drug, on Kv3.1 channels
- Affiliations
-
- 1Department of Pharmacology, Institute for Medical Sciences, Jeonbuk National University Medical School, Jeonju 54097, Korea
- 2Department of Physiology, Medical Research Center, College of Medicine, The Catholic University of Korea, Seoul 06591, Korea
- KMID: 2537507
- DOI: http://doi.org/10.4196/kjpp.2023.27.1.95
Abstract
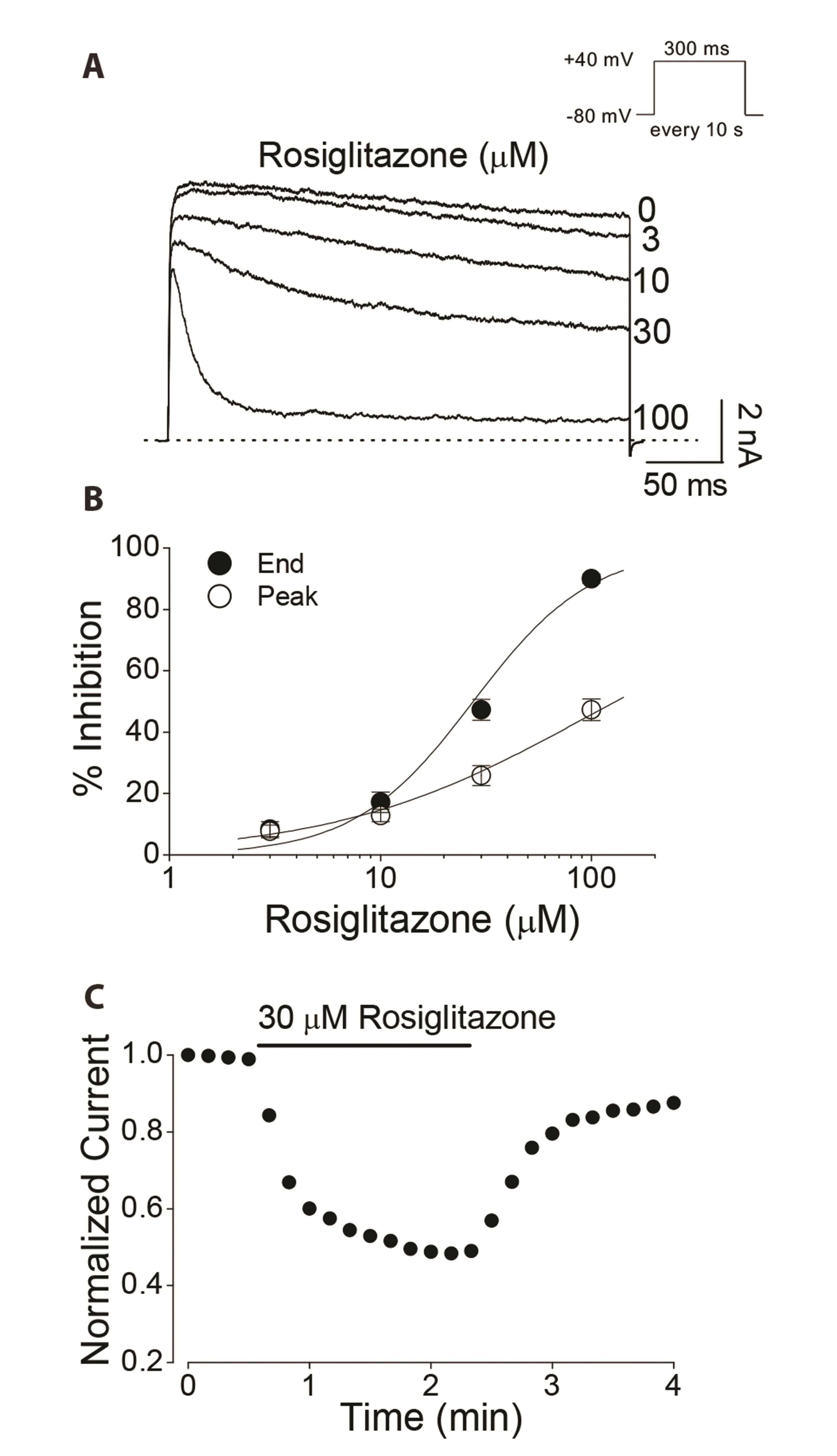
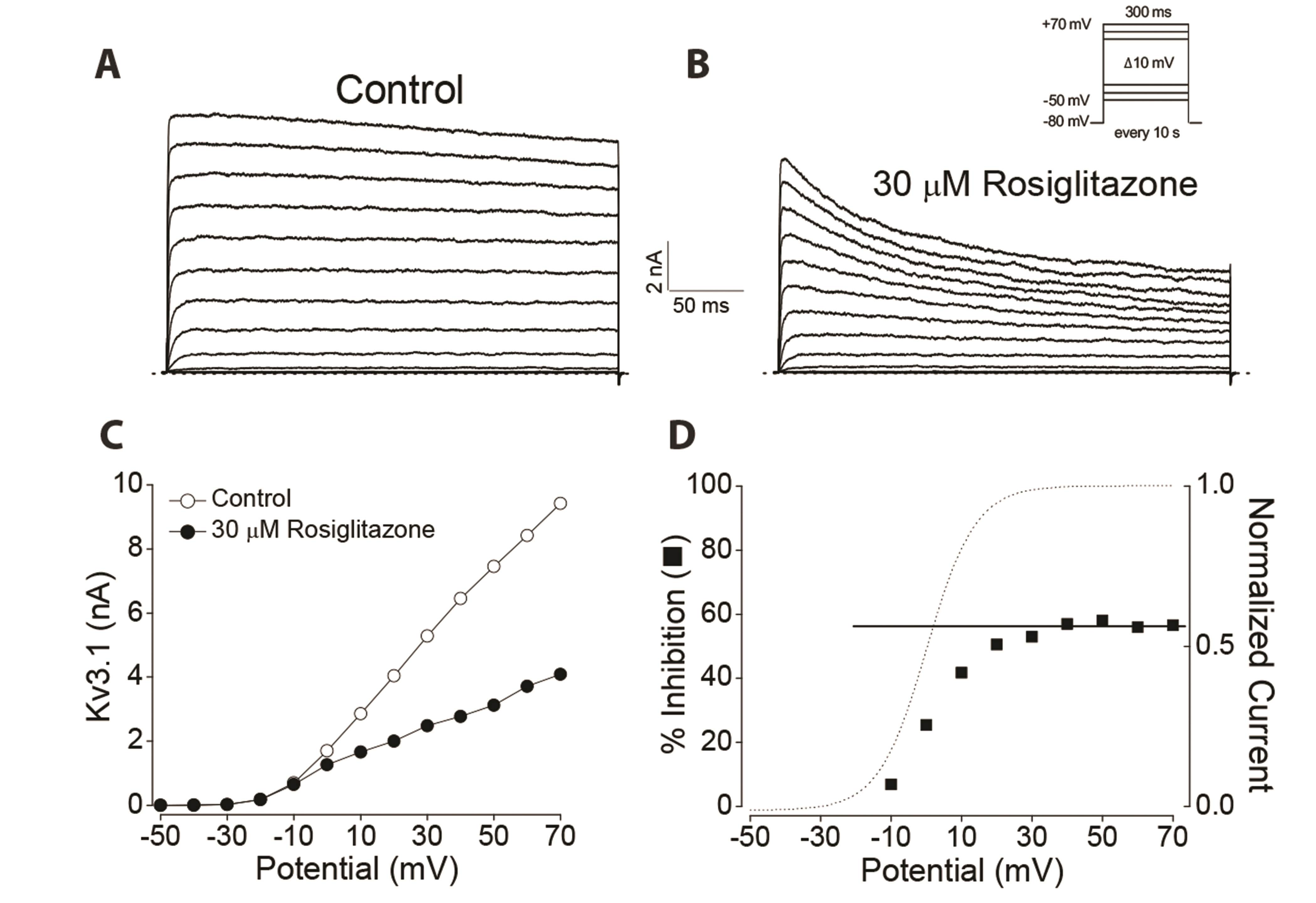
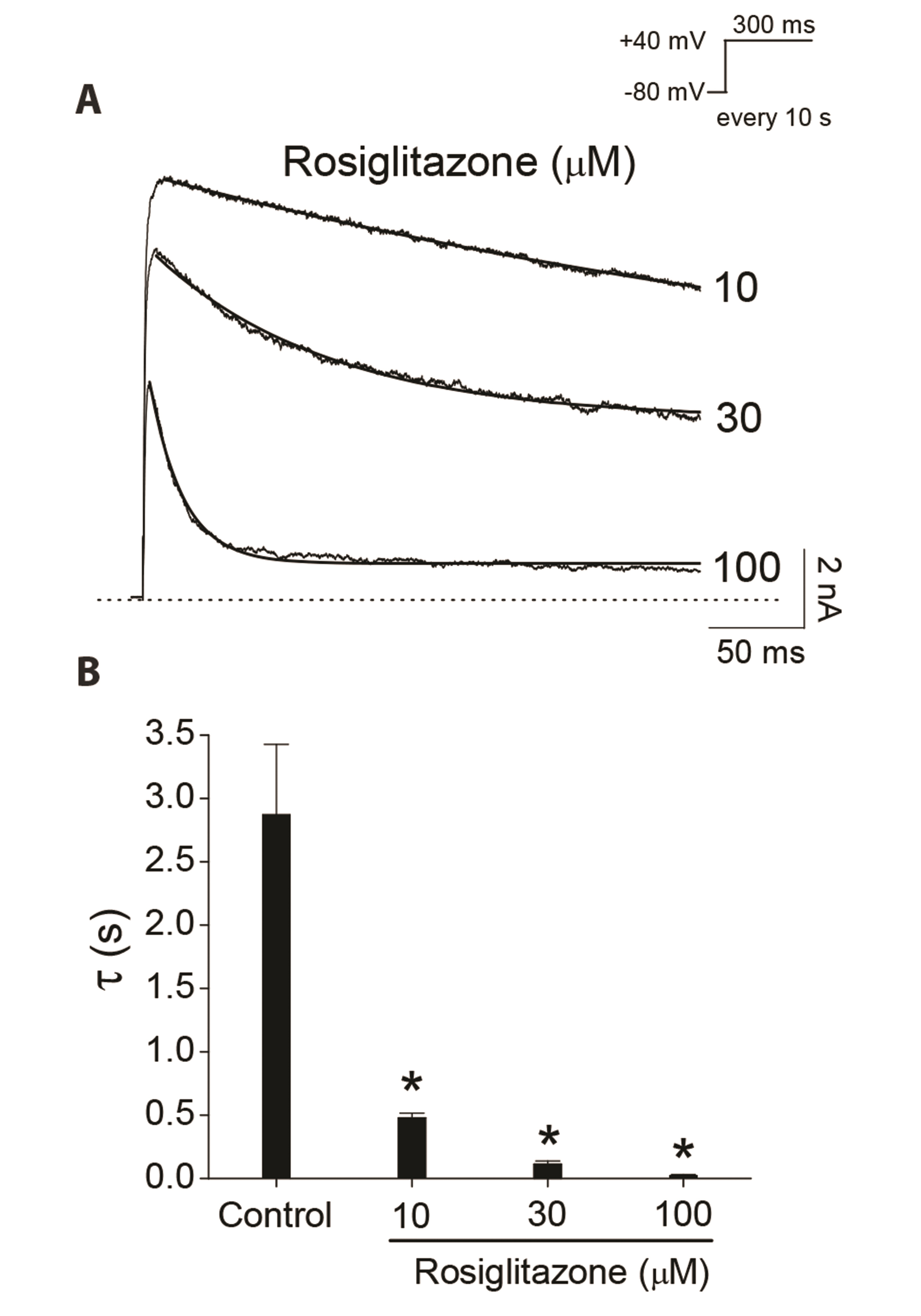
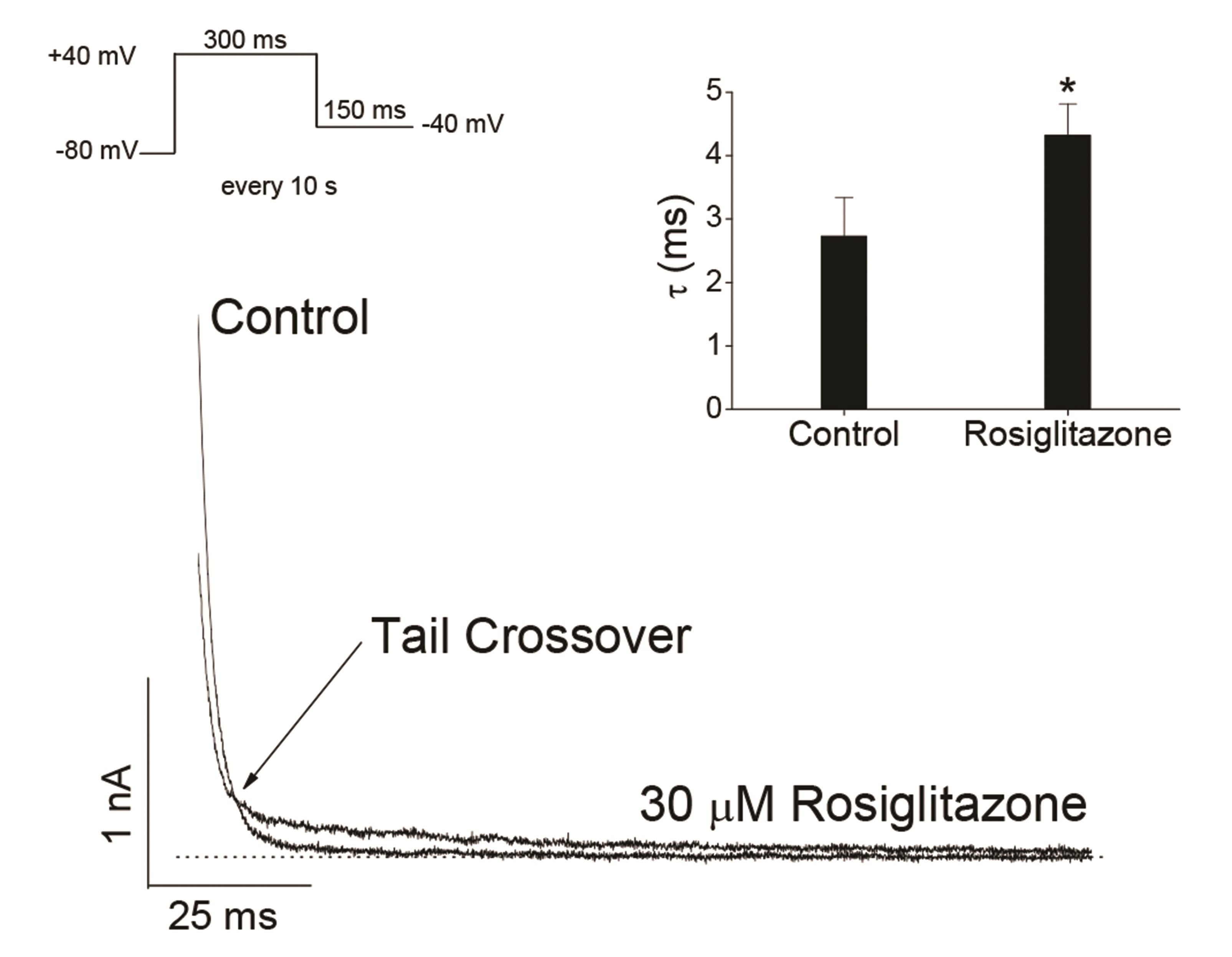
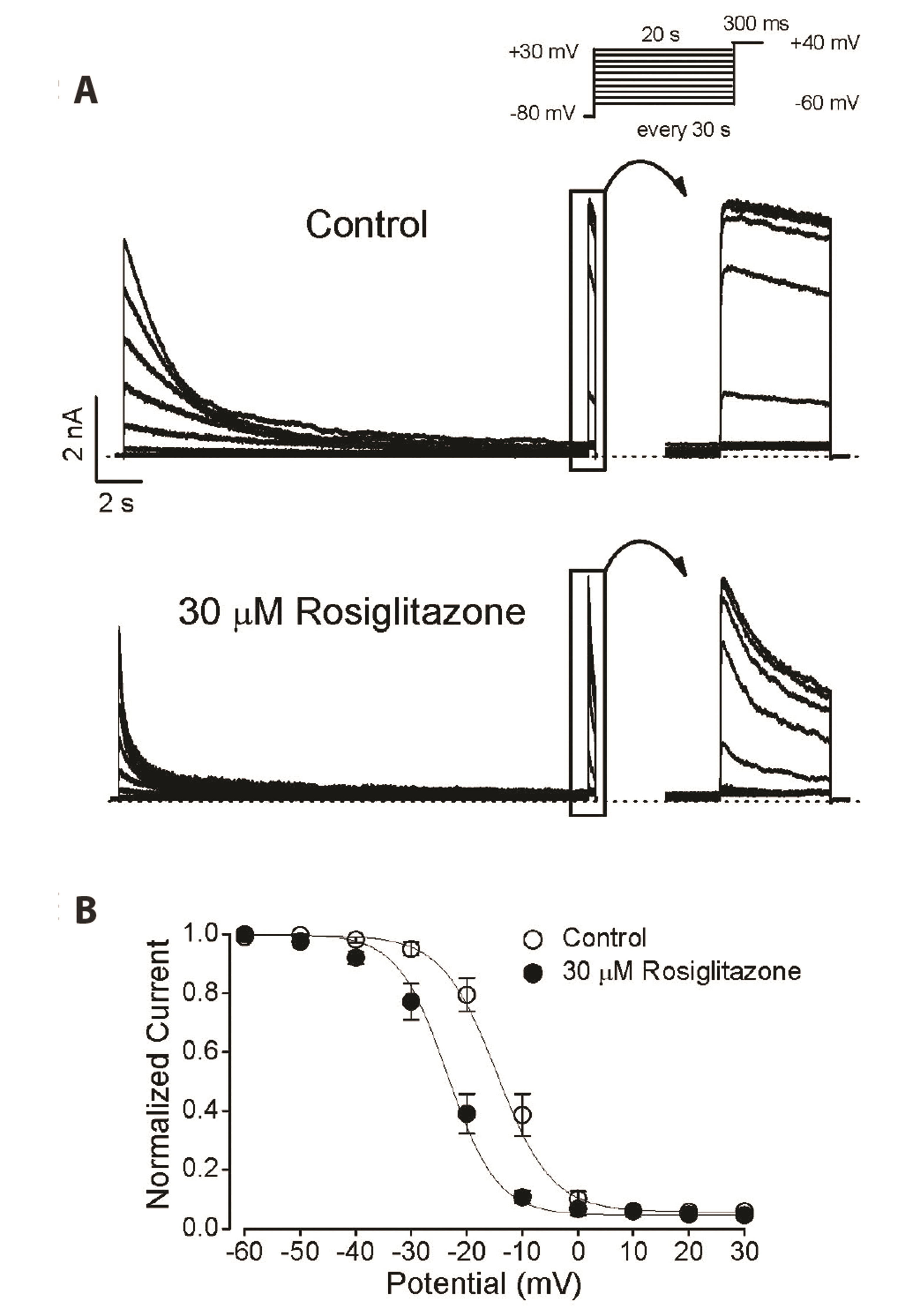
- Rosiglitazone is a thiazolidinedione-class antidiabetic drug that reduces blood glucose and glycated hemoglobin levels. We here investigated the interaction of rosiglitazone with Kv3.1 expressed in Chinese hamster ovary cells using the wholecell patch-clamp technique. Rosiglitazone rapidly and reversibly inhibited Kv3.1 currents in a concentration-dependent manner (IC 50 = 29.8 µM) and accelerated the decay of Kv3.1 currents without modifying the activation kinetics. The rosiglitazonemediated inhibition of Kv3.1 channels increased steeply in a sigmoidal pattern over the voltage range of –20 to +30 mV, whereas it was voltage-independent in the voltage range above +30 mV, where the channels were fully activated. The deactivation of Kv3.1 current, measured along with tail currents, was also slowed by the drug. In addition, the steady-state inactivation curve of Kv3.1 by rosiglitazone shifts to a negative potential without significant change in the slope value. All the results with the use dependence of the rosiglitazone-mediated blockade suggest that rosiglitazone acts on Kv3.1 channels as an open channel blocker.
Figure
Reference
-
1. Deeks ED, Keam SJ. 2007; Rosiglitazone: a review of its use in type 2 diabetes mellitus. Drugs. 67:2747–2779. DOI: 10.2165/00003495-200767180-00008. PMID: 18062722.2. Wagstaff AJ, Goa KL. 2002; Rosiglitazone: a review of its use in the management of type 2 diabetes mellitus. Drugs. 62:1805–1837. DOI: 10.2165/00003495-200262120-00007. PMID: 12149047. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0036343675&origin=inward.3. Lebovitz HE. 2019; Thiazolidinediones: the Forgotten Diabetes Medications. Curr Diab Rep. 19:151. DOI: 10.1007/s11892-019-1270-y. PMID: 31776781. PMCID: PMC6881429. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85075677686&origin=inward.4. Perney TM, Marshall J, Martin KA, Hockfield S, Kaczmarek LK. 1992; Expression of the mRNAs for the Kv3.1 potassium channel gene in the adult and developing rat brain. J Neurophysiol. 68:756–766. DOI: 10.1152/jn.1992.68.3.756. PMID: 1432046. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0026744145&origin=inward.5. Wang LY, Gan L, Forsythe ID, Kaczmarek LK. 1998; Contribution of the Kv3.1 potassium channel to high-frequency firing in mouse auditory neurones. J Physiol. 509(Pt 1):183–194. DOI: 10.1111/j.1469-7793.1998.183bo.x. PMID: 9547392. PMCID: PMC2230948. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0032523747&origin=inward.6. Kaczmarek LK, Zhang Y. 2017; Kv3 channels: enablers of rapid firing, neurotransmitter release, and neuronal endurance. Physiol Rev. 97:1431–1468. DOI: 10.1152/physrev.00002.2017. PMID: 28904001. PMCID: PMC6151494. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85029827375&origin=inward.7. Kanemasa T, Gan L, Perney TM, Wang LY, Kaczmarek LK. 1995; Electrophysiological and pharmacological characterization of a mammalian Shaw channel expressed in NIH 3T3 fibroblasts. J Neurophysiol. 74:207–217. DOI: 10.1152/jn.1995.74.1.207. PMID: 7472324. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0029086692&origin=inward.8. McDonald AJ, Mascagni F. 2006; Differential expression of Kv3.1b and Kv3.2 potassium channel subunits in interneurons of the basolateral amygdala. Neuroscience. 138:537–547. DOI: 10.1016/j.neuroscience.2005.11.047. PMID: 16413129. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=32844467747&origin=inward.9. Chow A, Erisir A, Farb C, Nadal MS, Ozaita A, Lau D, Welker E, Rudy B. 1999; K+ channel expression distinguishes subpopulations of parvalbumin- and somatostatin-containing neocortical interneurons. J Neurosci. 19:9332–9345. DOI: 10.1523/JNEUROSCI.19-21-09332.1999. PMID: 10531438. PMCID: PMC6782929. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0033232517&origin=inward.10. Sekirnjak C, Martone ME, Weiser M, Deerinck T, Bueno E, Rudy B, Ellisman M. 1997; Subcellular localization of the K+ channel subunit Kv3.1b in selected rat CNS neurons. Brain Res. 766:173–187. DOI: 10.1016/S0006-8993(97)00527-1. PMID: 9359601. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0030924116&origin=inward.11. Beghi E, Giussani G, Sander JW. 2015; The natural history and prognosis of epilepsy. Epileptic Disord. 17:243–253. DOI: 10.1684/epd.2015.0751. PMID: 26234761. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84942238130&origin=inward.12. Thijs RD, Surges R, O'Brien TJ, Sander JW. 2019; Epilepsy in adults. Lancet. 393:689–701. DOI: 10.1016/S0140-6736(18)32596-0. PMID: 30686584. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85061398477&origin=inward.13. Pack AM. 2019; Epilepsy overview and revised classification of seizures and epilepsies. Continuum (Minneap Minn). 25:306–321. DOI: 10.1212/CON.0000000000000707. PMID: 30921011. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85063892994&origin=inward.14. Muona M, Berkovic SF, Dibbens LM, Oliver KL, Maljevic S, Bayly MA, Joensuu T, Canafoglia L, Franceschetti S, Michelucci R, Markkinen S, Heron SE, Hildebrand MS, Andermann E, Andermann F, Gambardella A, Tinuper P, Licchetta L, Scheffer IE, Criscuolo C, et al. 2015; A recurrent de novo mutation in KCNC1 causes progressive myoclonus epilepsy. Nat Genet. 47:39–46. DOI: 10.1038/ng.3144. PMID: 25401298. PMCID: PMC4281260. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84930946843&origin=inward.15. Zahn RK, Tolner EA, Derst C, Gruber C, Veh RW, Heinemann U. 2008; Reduced ictogenic potential of 4-aminopyridine in the perirhinal and entorhinal cortex of kainate-treated chronic epileptic rats. Neurobiol Dis. 29:186–200. DOI: 10.1016/j.nbd.2007.08.013. PMID: 17942314. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=38149025433&origin=inward.16. Lee SM, Kim JE, Sohn JH, Choi HC, Lee JS, Kim SH, Kim MJ, Choi IG, Kang TC. 2009; Down-regulation of delayed rectifier K+ channels in the hippocampus of seizure sensitive gerbils. Brain Res Bull. 80:433–442. DOI: 10.1016/j.brainresbull.2009.07.016. PMID: 19665528. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=70349973889&origin=inward.17. Eto K, Ohya Y, Nakamura Y, Abe I, Fujishima M. 2001; Comparative actions of insulin sensitizers on ion channels in vascular smooth muscle. Eur J Pharmacol. 423:1–7. DOI: 10.1016/S0014-2999(01)01047-0. PMID: 11438300.18. Knock GA, Mishra SK, Aaronson PI. 1999; Differential effects of insulin-sensitizers troglitazone and rosiglitazone on ion currents in rat vascular myocytes. Eur J Pharmacol. 368:103–109. DOI: 10.1016/S0014-2999(99)00020-5. PMID: 10096775. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0033004825&origin=inward.19. Ahn HS, Kim SE, Jang HJ, Kim MJ, Rhie DJ, Yoon SH, Jo YH, Kim MS, Sung KW, Kim SY, Hahn SJ. 2007; Open channel block of Kv1.3 by rosiglitazone and troglitazone: Kv1.3 as the pharmacological target for rosiglitazone. Naunyn Schmiedebergs Arch Pharmacol. 374:305–309. DOI: 10.1007/s00210-006-0118-6. PMID: 17119927. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=33846221869&origin=inward.20. Jeong I, Choi BH, Hahn SJ. 2011; Rosiglitazone inhibits Kv4.3 potassium channels by open-channel block and acceleration of closed-state inactivation. Br J Pharmacol. 163:510–520. DOI: 10.1111/j.1476-5381.2011.01210.x. PMID: 21232039. PMCID: PMC3101614. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=79955911273&origin=inward.21. Lee HM, Hahn SJ, Choi BH. 2022; The antidiabetic drug rosiglitazone blocks Kv1.5 potassium channels in an open state. Korean J Physiol Pharmacol. 26:135–144. DOI: 10.4196/kjpp.2022.26.2.135. PMID: 35203063. PMCID: PMC8890944. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85127537308&origin=inward.22. Hahn SJ, Wang LY, Kaczmarek LK. 1996; Inhibition by nystatin of Kv1.3 channels expressed in Chinese hamster ovary cells. Neuropharmacology. 35:895–901. DOI: 10.1016/0028-3908(96)00094-9. PMID: 8938720. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0029909579&origin=inward.23. Luneau CJ, Williams JB, Marshall J, Levitan ES, Oliva C, Smith JS, Antanavage J, Folander K, Stein RB, Swanson R. 1991; Alternative splicing contributes to K+ channel diversity in the mammalian central nervous system. Proc Natl Acad Sci U S A. 88:3932–3936. DOI: 10.1073/pnas.88.9.3932. PMID: 2023941. PMCID: PMC51567. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0025764319&origin=inward.24. Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ. 1981; Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 391:85–100. DOI: 10.1007/BF00656997. PMID: 6270629. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0019441262&origin=inward.25. Choi BH, Choi JS, Jeong SW, Hahn SJ, Yoon SH, Jo YH, Kim MS. 2000; Direct block by bisindolylmaleimide of rat Kv1.5 expressed in Chinese hamster ovary cells. J Pharmacol Exp Ther. 293:634–640. PMID: 10773038. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0343527303&origin=inward.26. Lee HM, Chai OH, Hahn SJ, Choi BH. 2018; Antidepressant drug paroxetine blocks the open pore of Kv3.1 potassium channel. Korean J Physiol Pharmacol. 22:71–80. DOI: 10.4196/kjpp.2018.22.1.71. PMID: 29302214. PMCID: PMC5746514. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85041181251&origin=inward.27. Sung MJ, Ahn HS, Hahn SJ, Choi BH. 2008; Open channel block of Kv3.1 currents by fluoxetine. J Pharmacol Sci. 106:38–45. DOI: 10.1254/jphs.FP0070759. PMID: 18187934. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=38549093981&origin=inward.28. Valenzuela C, Delpón E, Franqueza L, Gay P, Pérez O, Tamargo J, Snyders DJ. 1996; Class III antiarrhythmic effects of zatebradine. Time-, state-, use-, and voltage-dependent block of hKv1.5 channels. Circulation. 94:562–570. DOI: 10.1161/01.CIR.94.3.562. PMID: 8759103. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0029790924&origin=inward.29. Delpón E, Valenzuela C, Gay P, Franqueza L, Snyders DJ, Tamargo J. 1997; Block of human cardiac Kv1.5 channels by loratadine: voltage-, time- and use-dependent block at concentrations above therapeutic levels. Cardiovasc Res. 35:341–350. DOI: 10.1016/S0008-6363(97)00121-1. PMID: 9349397. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0030768949&origin=inward.30. Franqueza L, Valenzuela C, Delpón E, Longobardo M, Caballero R, Tamargo J. 1998; Effects of propafenone and 5-hydroxy-propafenone on hKv1.5 channels. Br J Pharmacol. 125:969–978. DOI: 10.1038/sj.bjp.0702129. PMID: 9846634. PMCID: PMC1565661. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0031787052&origin=inward.31. Sung MJ, Hahn SJ, Choi BH. 2009; Effect of psoralen on the cloned Kv3.1 currents. Arch Pharm Res. 32:407–412. DOI: 10.1007/s12272-009-1314-y. PMID: 19387585. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=65449186304&origin=inward.32. Choi BH, Choi JS, Yoon SH, Rhie DJ, Min DS, Jo YH, Kim MS, Hahn SJ. 2001; Effects of norfluoxetine, the major metabolite of fluoxetine, on the cloned neuronal potassium channel Kv3.1. Neuropharmacology. 41:443–453. DOI: 10.1016/S0028-3908(01)00088-0. PMID: 11543764. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0034866127&origin=inward.33. Lee HM, Hahn SJ, Choi BH. 2010; Open channel block of Kv1.5 currents by citalopram. Acta Pharmacol Sin. 31:429–435. DOI: 10.1038/aps.2010.14. PMID: 20228830. PMCID: PMC4007671. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=77952119359&origin=inward.34. Park J, Cho KH, Lee HJ, Choi JS, Rhie DJ. 2020; Open channel block of Kv1.4 potassium channels by aripiprazole. Korean J Physiol Pharmacol. 24:545–553. DOI: 10.4196/kjpp.2020.24.6.545. PMID: 33093275. PMCID: PMC7585592. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85095843104&origin=inward.35. Lee HM, Hahn SJ, Choi BH. 2016; Blockade of Kv1.5 by paroxetine, an antidepressant drug. Korean J Physiol Pharmacol. 20:75–82. DOI: 10.4196/kjpp.2016.20.1.75. PMID: 26807026. PMCID: PMC4722194. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84956956934&origin=inward.36. Lee HM, Hahn SJ, Choi BH. 2016; Blockade of Kv1.5 channels by the antidepressant drug sertraline. Korean J Physiol Pharmacol. 20:193–200. DOI: 10.4196/kjpp.2016.20.2.193. PMID: 26937216. PMCID: PMC4770110. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84959931160&origin=inward.37. Kolte BL, Raut BB, Deo AA, Bagool MA, Shinde DB. 2003; Liquid chromatographic method for the determination of rosiglitazone in human plasma. J Chromatogr B Analyt Technol Biomed Life Sci. 788:37–44. DOI: 10.1016/S1570-0232(02)01011-5. PMID: 12668069. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0037420712&origin=inward.38. Wang Z, Fermini B, Nattel S. 1995; Effects of flecainide, quinidine, and 4-aminopyridine on transient outward and ultrarapid delayed rectifier currents in human atrial myocytes. J Pharmacol Exp Ther. 272:184–196. PMID: 7815332. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0028838977&origin=inward.39. Jeong I, Choi BH, Hahn SJ. 2013; Pergolide block of the cloned Kv1.5 potassium channels. Naunyn Schmiedebergs Arch Pharmacol. 386:125–133. DOI: 10.1007/s00210-012-0776-5. PMID: 22763615. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84873735597&origin=inward.40. Jeong I, Choi BH, Hahn SJ. 2010; Effects of lobeline, a nicotinic receptor ligand, on the cloned Kv1.5. Pflugers Arch. 460:851–862. DOI: 10.1007/s00424-010-0868-3. PMID: 20734202. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=77957925870&origin=inward.41. Lacerda AE, Roy ML, Lewis EW, Rampe D. 1997; Interactions of the nonsedating antihistamine loratadine with a Kv1.5-type potassium channel cloned from human heart. Mol Pharmacol. 52:314–322. Erratum. DOI: 10.1124/mol.52.2.314. PMID: 9271355. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0030852667&origin=inward.42. Butterworth JF 4th, Strichartz GR. 1990; Molecular mechanisms of local anesthesia: a review. Anesthesiology. 72:711–734. DOI: 10.1097/00000542-199004000-00022. PMID: 2157353. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0025263891&origin=inward.43. Chapelsky MC, Thompson-Culkin K, Miller AK, Sack M, Blum R, Freed MI. 2003; Pharmacokinetics of rosiglitazone in patients with varying degrees of renal insufficiency. J Clin Pharmacol. 43:252–259. DOI: 10.1177/0091270002250602. PMID: 12638393. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0344453817&origin=inward.44. Hruska MW, Frye RF. 2004; Simplified method for determination of rosiglitazone in human plasma. J Chromatogr B Analyt Technol Biomed Life Sci. 803:317–320. DOI: 10.1016/j.jchromb.2004.01.010. PMID: 15063342. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=1842479290&origin=inward.45. Erisir A, Lau D, Rudy B, Leonard CS. 1999; Function of specific K+ channels in sustained high-frequency firing of fast-spiking neocortical interneurons. J Neurophysiol. 82:2476–2489. Erratum. DOI: 10.1152/jn.1999.82.5.2476. PMID: 10561420.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The antidiabetic drug rosiglitazone blocks Kv1.5 potassium channels in an open state
- Antidepressant drug paroxetine blocks the open pore of Kv3.1 potassium channel
- Review of Cardiovascular Effects of Antidiabetic Drugs and Recent Cardiovascular Outcome Trials
- Open Channel Block of Kv3.1 Currents by Genistein, a Tyrosine Kinase Inhibitor
- Clinical Trial for Antidiabetic Drugs: FDA Guidance for Diabetes Mellitus-Evaluation of Cardiovascular Risk in New Antidiabetic Therapies