J Pathol Transl Med.
2022 Nov;56(6):334-341. 10.4132/jptm.2022.08.22.
Usefulness of BRAF VE1 immunohistochemistry in non–small cell lung cancers: a multi-institutional study by 15 pathologists in Korea
- Affiliations
-
- 1Department of Pathology, Inje University Ilsan Paik Hospital, Goyang, Korea
- 2Department of Pathology and Translational Genomics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
- 3Department of Pathology, Yonsei University College of Medicine, Seoul, Korea
- 4Department of Pathology, National Cancer Center, Goyang, Korea
- 5Department of Pathology, Gachon University Gil Medical Center, Gachon University College of Medicine, Incheon, Korea
- KMID: 2535853
- DOI: http://doi.org/10.4132/jptm.2022.08.22
Abstract
- Background
Next-generation sequencing (NGS) is an approved test to select patients for BRAF V600E targeted therapy in Korea. However, the high cost, long turnaround times, and the need for sophisticated equipment and skilled personnel limit the use of NGS in daily practice. Immunohistochemistry (IHC) is a rapid and relatively inexpensive assay available in most laboratories. Therefore, in this study, we evaluate the usefulness of BRAF VE1 IHC in terms of predictive value and interobserver agreement in non–small cell lung cancers (NSCLCs).
Methods
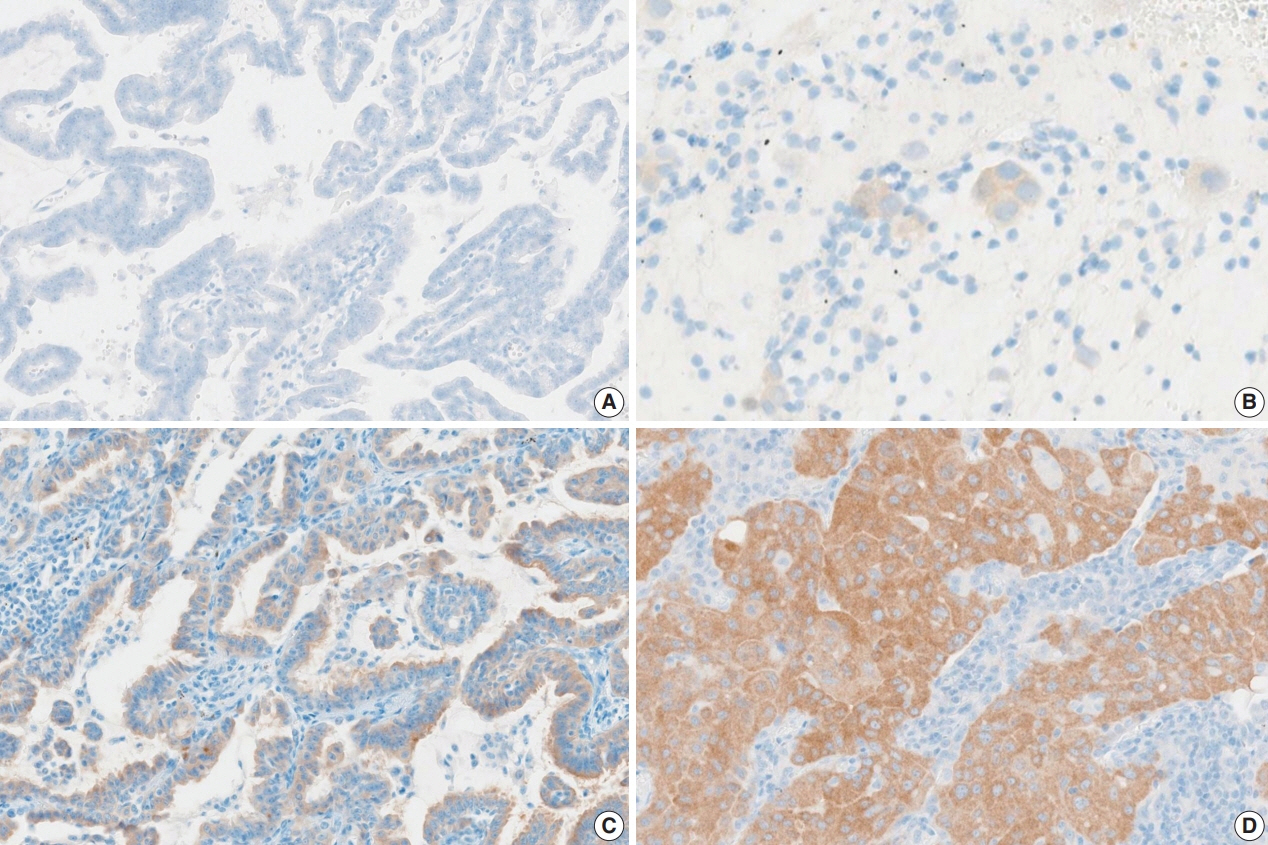
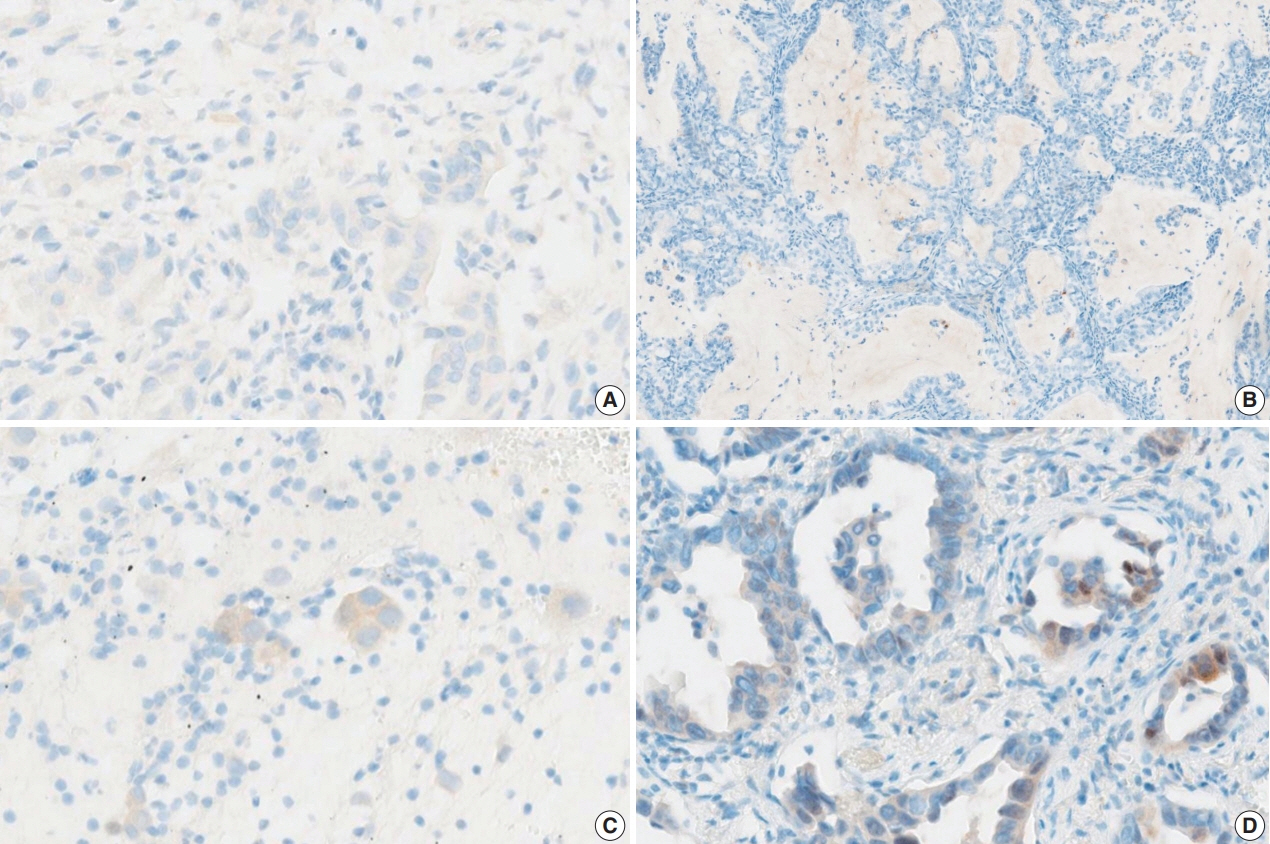
A total of 30 cases with known BRAF mutation status were selected, including 20 cases of lung adenocarcinomas, six cases of colorectal adenocarcinomas, and four cases of papillary thyroid carcinomas. IHC for BRAF V600E was carried out using the VE1 antibody. Fifteen pathologists independently scored both the staining intensity and the percentage of tumor cell staining on whole slide images.
Results
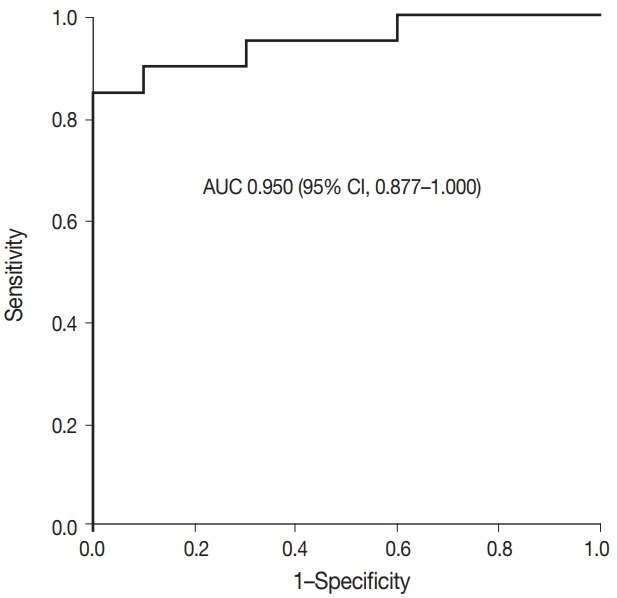
In the lung adenocarcinoma subset, interobserver agreement for the percentage of tumor cell staining and staining intensity was good (percentage of tumor cell staining, intraclass correlation coefficient = 0.869; staining intensity, kappa = 0.849). The interobserver agreement for the interpretation using the cutoff of 40% was almost perfect in the entire study group and the lung adenocarcinoma subset (kappa = 0.815). Sensitivity, specificity, positive predictive value, and negative predictive value of BRAF VE1 IHC were 80.0%, 90.0%, 88.9%, and 81.8%, respectively.
Conclusions
BRAF VE1 IHC could be a screening test for the detection of BRAF V600E mutation in NSCLC. However, further studies are needed to optimize the protocol and to establish and validate interpretation criteria for BRAF VE1 IHC.
Keyword
Figure
Reference
-
References
1. Chang S, Shim HS, Kim TJ, et al. Molecular biomarker testing for non-small cell lung cancer: consensus statement of the Korean Cardiopulmonary Pathology Study Group. J Pathol Transl Med. 2021; 55:181–91.
Article2. Sepulveda AR, Hamilton SR, Allegra CJ, et al. Molecular biomarkers for the evaluation of colorectal cancer: guideline from the American Society for Clinical Pathology, College of American Pathologists, Association for Molecular Pathology, and American Society of Clinical Oncology. Arch Pathol Lab Med. 2017; 141:625–57.
Article3. Dvorak K, Higgins A, Palting J, Cohen M, Brunhoeber P. Immunohistochemistry with anti-BRAF V600E (VE1) mouse monoclonal antibody is a sensitive method for detection of the BRAF V600E mutation in colon cancer: evaluation of 120 cases with and without KRAS mutation and literature review. Pathol Oncol Res. 2019; 25:349–59.4. Seto K, Haneda M, Masago K, et al. Negative reactions of BRAF mutation-specific immunohistochemistry to non-V600E mutations of BRAF. Pathol Int. 2020; 70:253–61.
Article5. Sasaki H, Shimizu S, Tani Y, et al. Usefulness of immunohistochemistry for the detection of the BRAF V600E mutation in Japanese lung adenocarcinoma. Lung Cancer. 2013; 82:51–4.
Article6. Ilie M, Long E, Hofman V, et al. Diagnostic value of immunohistochemistry for the detection of the BRAFV600E mutation in primary lung adenocarcinoma Caucasian patients. Ann Oncol. 2013; 24:742–8.7. Gow CH, Hsieh MS, Lin YT, Liu YN, Shih JY. Validation of immunohistochemistry for the detection of BRAF V600E-mutated lung adenocarcinomas. Cancers (Basel). 2019; 11:886.8. Hwang I, Choi YL, Lee H, et al. Selection strategies and practical application of BRAF V600E-mutated non-small cell lung carcinoma. Cancer Res Treat. 2022; 54:782–92.
Article9. O’Brien O, Lyons T, Murphy S, Feeley L, Power D, Heffron C. BRAF V600 mutation detection in melanoma: a comparison of two laboratory testing methods. J Clin Pathol. 2017; 70:935–40.10. Anwar MA, Murad F, Dawson E, Abd Elmageed ZY, Tsumagari K, Kandil E. Immunohistochemistry as a reliable method for detection of BRAF-V600E mutation in melanoma: a systematic review and meta-analysis of current published literature. J Surg Res. 2016; 203:407–15.11. Pyo JS, Sohn JH, Kang G. BRAF immunohistochemistry using clone VE1 is strongly concordant with BRAF(V600E) mutation test in papillary thyroid carcinoma. Endocr Pathol. 2015; 26:211–7.
Article12. Schafroth C, Galvan JA, Centeno I, et al. VE1 immunohistochemistry predicts BRAF V600E mutation status and clinical outcome in colorectal cancer. Oncotarget. 2015; 6:41453–63.
Article13. Rusu S, Verocq C, Trepant AL, et al. Immunohistochemistry as an accurate tool for the assessment of BRAF V600E and TP53 mutations in primary and metastatic melanoma. Mol Clin Oncol. 2021; 15:270.14. Marin C, Beauchet A, Capper D, et al. Detection of BRAF p.V600E mutations in melanoma by immunohistochemistry has a good interobserver reproducibility. Arch Pathol Lab Med. 2014; 138:71–5.15. Fisher KE, Cohen C, Siddiqui MT, Palma JF, Lipford EH, Longshore JW. Accurate detection of BRAF p.V600E mutations in challenging melanoma specimens requires stringent immunohistochemistry scoring criteria or sensitive molecular assays. Hum Pathol. 2014; 45:2281–93.16. Bledsoe JR, Kamionek M, Mino-Kenudson M. BRAF V600E immunohistochemistry is reliable in primary and metastatic colorectal carcinoma regardless of treatment status and shows high intratumoral homogeneity. Am J Surg Pathol. 2014; 38:1418–28.17. Toon CW, Walsh MD, Chou A, et al. BRAFV600E immunohistochemistry facilitates universal screening of colorectal cancers for Lynch syndrome. Am J Surg Pathol. 2013; 37:1592–602.18. Rossle M, Sigg M, Ruschoff JH, et al. Ultra-deep sequencing confirms immunohistochemistry as a highly sensitive and specific method for detecting BRAF V600E mutations in colorectal carcinoma. Virchows Arch. 2013; 463:623–31.19. Capper D, Voigt A, Bozukova G, et al. BRAF V600E-specific immunohistochemistry for the exclusion of Lynch syndrome in MSI-H colorectal cancer. Int J Cancer. 2013; 133:1624–30.20. Affolter K, Samowitz W, Tripp S, Bronner MP. BRAF V600E mutation detection by immunohistochemistry in colorectal carcinoma. Genes Chromosomes Cancer. 2013; 52:748–52.
Article21. Hofman V, Benzaquen J, Heeke S, et al. Real-world assessment of the BRAF status in non-squamous cell lung carcinoma using VE1 immunohistochemistry: a single laboratory experience (LPCE, Nice, France). Lung Cancer. 2020; 145:58–62.
Article22. Karbel HA, Ejam SS, Naji AZ. Immunohistochemical study using monoclonal VE1 antibody can substitute the molecular tests for apprehension of BRAF V600E mutation in patients with non-smallcell lung carcinoma. Anal Cell Pathol (Amst). 2019; 2019:2315673.23. NordiQC. Assessment Run 62 2021 BRAF (BRAF V600E) [Internet]. Aalborg: NordiQC;2021. [cited 2021 Nov 30]. Available from: https://www.nordiqc.org/downloads/assessments/146_113.pdf.24. Sinicrope FA, Smyrk TC, Tougeron D, et al. Mutation-specific antibody detects mutant BRAFV600E protein expression in human colon carcinomas. Cancer. 2013; 119:2765–70.
Article25. Adackapara CA, Sholl LM, Barletta JA, Hornick JL. Immunohistochemistry using the BRAF V600E mutation-specific monoclonal antibody VE1 is not a useful surrogate for genotyping in colorectal adenocarcinoma. Histopathology. 2013; 63:187–93.
Article26. Kuan SF, Navina S, Cressman KL, Pai RK. Immunohistochemical detection of BRAF V600E mutant protein using the VE1 antibody in colorectal carcinoma is highly concordant with molecular testing but requires rigorous antibody optimization. Hum Pathol. 2014; 45:464–72.27. Lasota J, Kowalik A, Wasag B, et al. Detection of the BRAF V600E mutation in colon carcinoma: critical evaluation of the imunohistochemical approach. Am J Surg Pathol. 2014; 38:1235–41.28. Bullock M, O’Neill C, Chou A, et al. Utilization of a MAB for BRAF(V600E) detection in papillary thyroid carcinoma. Endocr Relat Cancer. 2012; 19:779–84.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Sensitivity and Usefulness of VE1 Immunohistochemical Staining in Acral Melanomas with BRAF Mutation
- Selection Strategies and Practical Application of BRAF V600E-Mutated Non–Small Cell Lung Carcinoma
- Utility of BRAF VE1 Immunohistochemistry as a Screening Tool for Colorectal Cancer Harboring BRAF V600E Mutation
- Evaluation of the VE1 Antibody in Thyroid Cytology Using Ex Vivo Papillary Thyroid Carcinoma Specimens
- Interobserver Reproducibility of PD-L1 Biomarker in Non-small Cell Lung Cancer: A Multi-Institutional Study by 27 Pathologists