Endocrinol Metab.
2022 Jun;37(3):552-557. 10.3803/EnM.2022.1421.
Sestrin2 Regulates Beneficial β3-Adrenergic Receptor-Mediated Effects Observed in Inguinal White Adipose Tissue and Soleus Muscle
- Affiliations
-
- 1Division of Endocrinology and Metabolism, Department of Internal Medicine, Korea University College of Medicine, Seoul, Korea
- 2BK21 Graduate Program, Department of Biomedical Sciences and Department of Internal Medicine, Korea University College of Medicine, Seoul, Korea
- KMID: 2531399
- DOI: http://doi.org/10.3803/EnM.2022.1421
Abstract
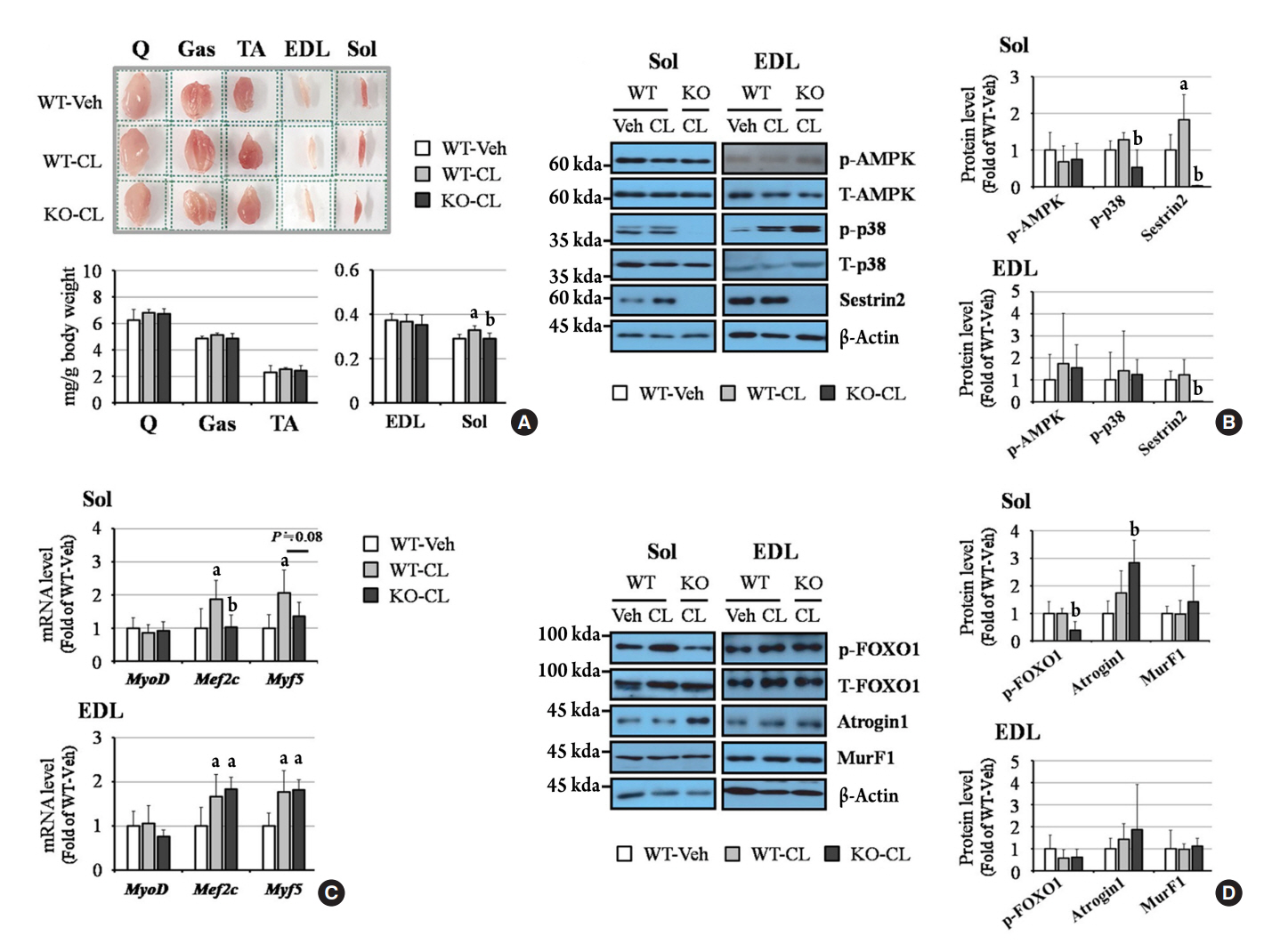
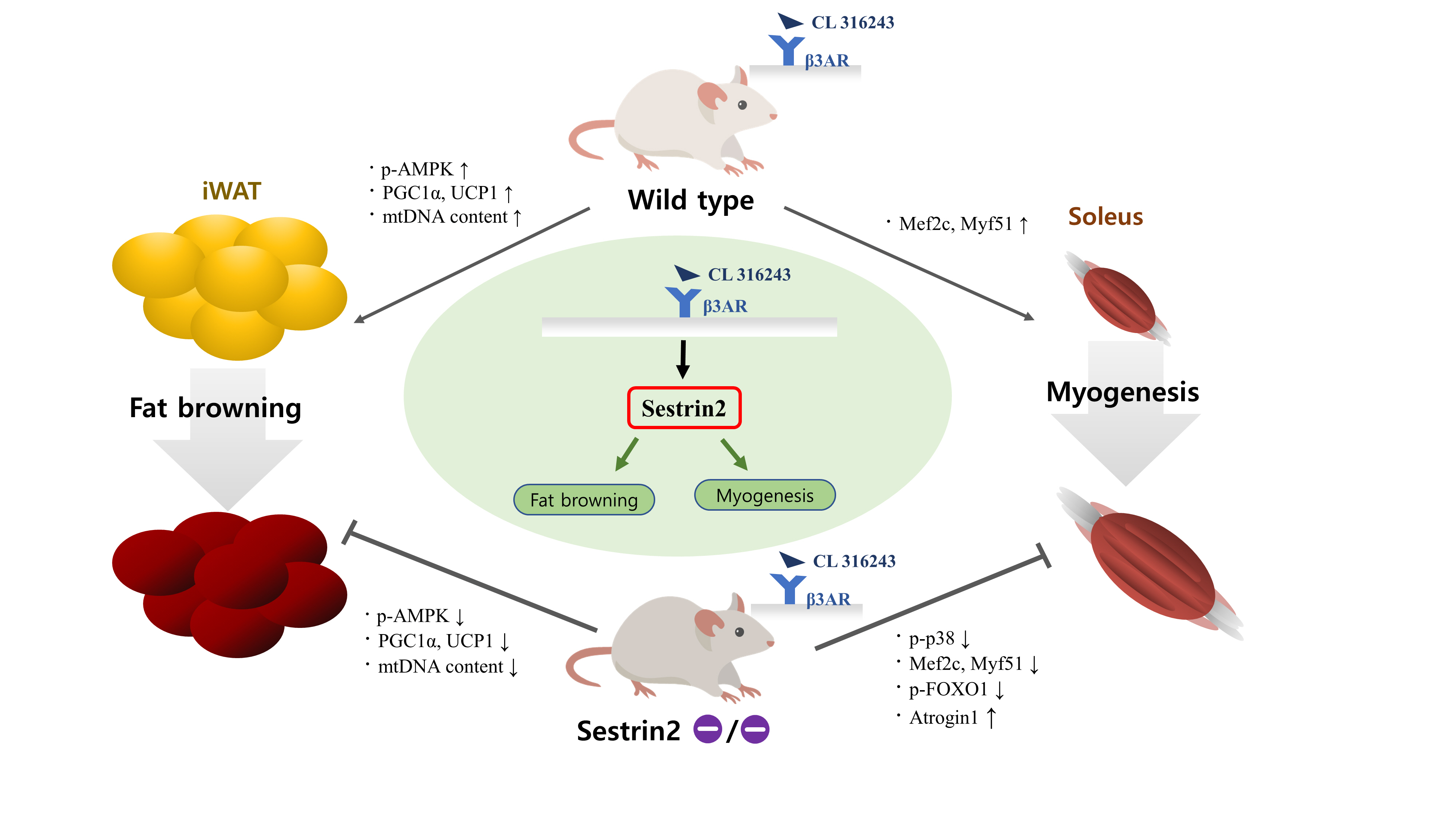
- Sestrin2, a well-known adenosine monophosphate-activated protein kinase (AMPK) regulator, plays a protective role against metabolic stress. The β3-adrenergic receptor (β3AR) induces fat browning and inhibits muscle atrophy in an AMPK-dependent manner. However, no prior research has examined the relationship of sestrin2 with β3AR in body composition changes. In this study, CL 316,243 (CL), a β3AR agonist, was administered to wild-type and sestrin2-knockout (KO) mice for 2 weeks, and fat and muscle tissues were harvested. CL induced AMPK phosphorylation, expression of brown-fat markers, and mitochondrial biogenesis, which resulted in the reduction of lipid droplet size in inguinal white adipose tissue (iWAT). These effects were not observed in sestrin2-KO mice. In CL-treated soleus muscle, sestrin2-KO was related to decreased myogenic gene expression and increased levels of muscle atrophy-related molecules. Our results suggest that sestrin2 is associated with beneficial β3AR-mediated changes in body composition, especially in iWAT and in the soleus.
Keyword
Figure
Reference
-
1. Zamboni M, Mazzali G, Fantin F, Rossi A, Di Francesco V. Sarcopenic obesity: a new category of obesity in the elderly. Nutr Metab Cardiovasc Dis. 2008; 18:388–95.
Article2. Hornyak V, Brach JS, Wert DM, Hile E, Studenski S, VanSwearingen JM. What is the relation between fear of falling and physical activity in older adults? Arch Phys Med Rehabil. 2013; 94:2529–34.
Article3. Bartelt A, Heeren J. Adipose tissue browning and metabolic health. Nat Rev Endocrinol. 2014; 10:24–36.
Article4. Mottillo EP, Desjardins EM, Crane JD, Smith BK, Green AE, Ducommun S, et al. Lack of adipocyte AMPK exacerbates insulin resistance and hepatic steatosis through brown and beige adipose tissue function. Cell Metab. 2016; 24:118–29.
Article5. Finlin BS, Memetimin H, Zhu B, Confides AL, Vekaria HJ, El Khouli RH, et al. The β3-adrenergic receptor agonist mirabegron improves glucose homeostasis in obese humans. J Clin Invest. 2020; 130:2319–31.
Article6. Lee JH, Budanov AV, Talukdar S, Park EJ, Park HL, Park HW, et al. Maintenance of metabolic homeostasis by Sestrin2 and Sestrin3. Cell Metab. 2012; 16:311–21.
Article7. Park HW, Park H, Ro SH, Jang I, Semple IA, Kim DN, et al. Hepatoprotective role of Sestrin2 against chronic ER stress. Nat Commun. 2014; 5:4233.
Article8. Ro SH, Nam M, Jang I, Park HW, Park H, Semple IA, et al. Sestrin2 inhibits uncoupling protein 1 expression through suppressing reactive oxygen species. Proc Natl Acad Sci U S A. 2014; 111:7849–54.
Article9. Segales J, Perdiguero E, Serrano AL, Sousa-Victor P, Ortet L, Jardi M, et al. Sestrin prevents atrophy of disused and aging muscles by integrating anabolic and catabolic signals. Nat Commun. 2020; 11:189.
Article10. Budanov AV, Karin M. p53 target genes sestrin1 and sestrin2 connect genotoxic stress and mTOR signaling. Cell. 2008; 134:451–60.
Article11. Wu L, Zhang L, Li B, Jiang H, Duan Y, Xie Z, et al. AMP-activated protein kinase (AMPK) regulates energy metabolism through modulating thermogenesis in adipose tissue. Front Physiol. 2018; 9:122.
Article12. Nedergaard J, Cannon B. The browning of white adipose tissue: some burning issues. Cell Metab. 2014; 20:396–407.
Article13. Ricquier D. Uncoupling protein 1 of brown adipocytes, the only uncoupler: a historical perspective. Front Endocrinol (Lausanne). 2011; 2:85.
Article14. Zuriaga MA, Fuster JJ, Gokce N, Walsh K. Humans and mice display opposing patterns of “browning” gene expression in visceral and subcutaneous white adipose tissue depots. Front Cardiovasc Med. 2017; 4:27.
Article15. Gonzalez-Hurtado E, Lee J, Choi J, Wolfgang MJ. Fatty acid oxidation is required for active and quiescent brown adipose tissue maintenance and thermogenic programing. Mol Metab. 2018; 7:45–56.
Article16. Evans W. Functional and metabolic consequences of sarcopenia. J Nutr. 1997; 127(5 Suppl):998S–1003S.17. Li H, Liu S, Yuan H, Niu Y, Fu L. Sestrin 2 induces autophagy and attenuates insulin resistance by regulating AMPK signaling in C2C12 myotubes. Exp Cell Res. 2017; 354:18–24.
Article18. Lenhare L, Crisol BM, Silva VR, Katashima CK, Cordeiro AV, Pereira KD, et al. Physical exercise increases Sestrin 2 protein levels and induces autophagy in the skeletal muscle of old mice. Exp Gerontol. 2017; 97:17–21.
Article19. Soukup T, Zacharova G, Smerdu V. Fibre type composition of soleus and extensor digitorum longus muscles in normal female inbred Lewis rats. Acta Histochem. 2002; 104:399–405.
Article20. Kim J, Grotegut CA, Wisler JW, Li T, Mao L, Chen M, et al. β-Arrestin 1 regulates β2-adrenergic receptor-mediated skeletal muscle hypertrophy and contractility. Skelet Muscle. 2018; 8:39.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Deletion of adipose triglyceride lipase abolishes blood flow increase after β3-adrenergic stimulation in visceral adipose tissue of mice
- A study of alpha adrenergic receptors in human and rabbit corpus cavernosum tissue
- The Role of beta-Adrenergic Receptor in the Seminal Vesicle Contraction
- Bile Acid Receptor Farnesoid X Receptor: A Novel Therapeutic Target for Metabolic Diseases
- Swim training improves leptin receptor deficiency-induced obesity and lipid disorder by activating uncoupling proteins