Endocrinol Metab.
2022 Jun;37(3):392-407. 10.3803/EnM.2021.1349.
Interplay of Vitamin D and CYP3A4 Polymorphisms in Endocrine Disorders and Cancer
- Affiliations
-
- 1Biomarker Discovery Laboratory, Translational Health Science and Technology Institute, Faridabad, India
- 2Institute of Inorganic and Analytical Chemistry, Justus Liebig University Giessen, Giessen, Germany
- 3Institute of Pharmaceutical Technology, Sri Padmavati Mahila Visvavidyalayam (Women’s University), Tirupati, India
- KMID: 2531383
- DOI: http://doi.org/10.3803/EnM.2021.1349
Abstract
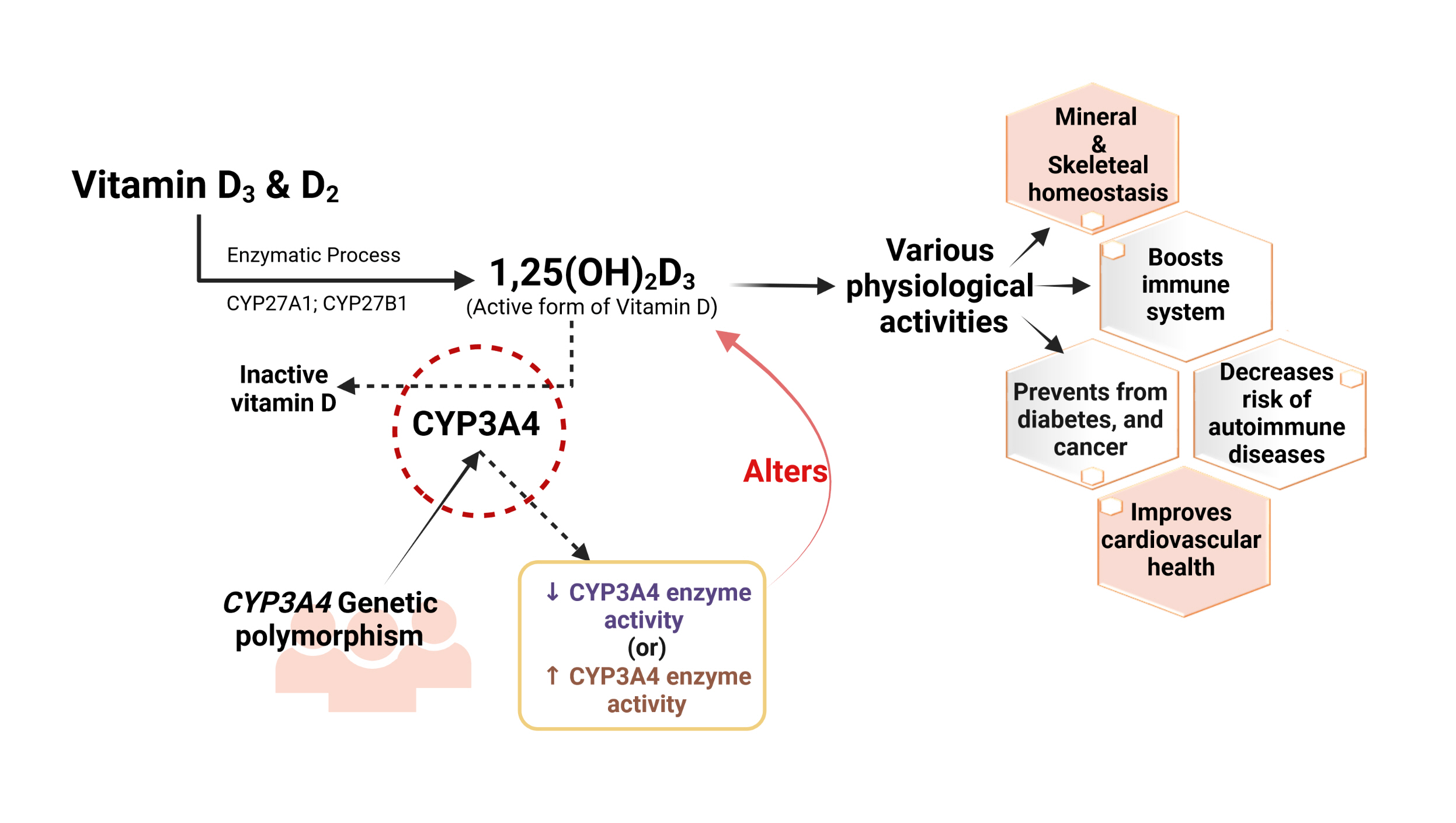
- Vitamin D has received considerable optimistic attention as a potentially important factor in many pathological states over the past few decades. However, the proportion of the active form of vitamin D metabolites responsible for biological activity is highly questionable in disease states due to flexible alterations in the enzymes responsible for their metabolism. For instance, CYP3A4 plays a crucial role in the biotransformation of vitamin D and other drug substances. Food-drug and/or drug-drug interactions, the disease state, genetic polymorphism, age, sex, diet, and environmental factors all influence CYP3A4 activity. Genetic polymorphisms in CYP450-encoding genes have received considerable attention in the past few decades due to their extensive impact on the pharmacokinetic and dynamic properties of drugs and endogenous substances. In this review, we focused on CYP3A4 polymorphisms and their interplay with vitamin D metabolism and summarized the role of vitamin D in calcium homeostasis, bone diseases, diabetes, cancer, other diseases, and drug substances. We also reviewed clinical observations pertaining to CYP3A4 polymorphisms among the aforementioned disease conditions. In addition, we highlighted the future perspectives of studying the pharmacogenetics of CYP3A4, which may have potential clinical significance for developing novel diagnostic genetic markers that will ascertain disease risk and progression.
Keyword
Figure
Reference
-
1. Trochoutsou AI, Kloukina V, Samitas K, Xanthou G. Vitamin-D in the immune system: genomic and non-genomic actions. Mini Rev Med Chem. 2015; 15:953–63.
Article2. Abai B. StatPearls. Treasure Island: StatPearls Publishing;2022. Chapter, Vitamin D deficiency. [cited 2022 May 30]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK532266.3. Nadeem S, Munim TF, Hussain HF, Hussain DF. Determinants of vitamin D deficiency in asymptomatic healthy young medical students. Pak J Med Sci. 2018; 34:1248–52.
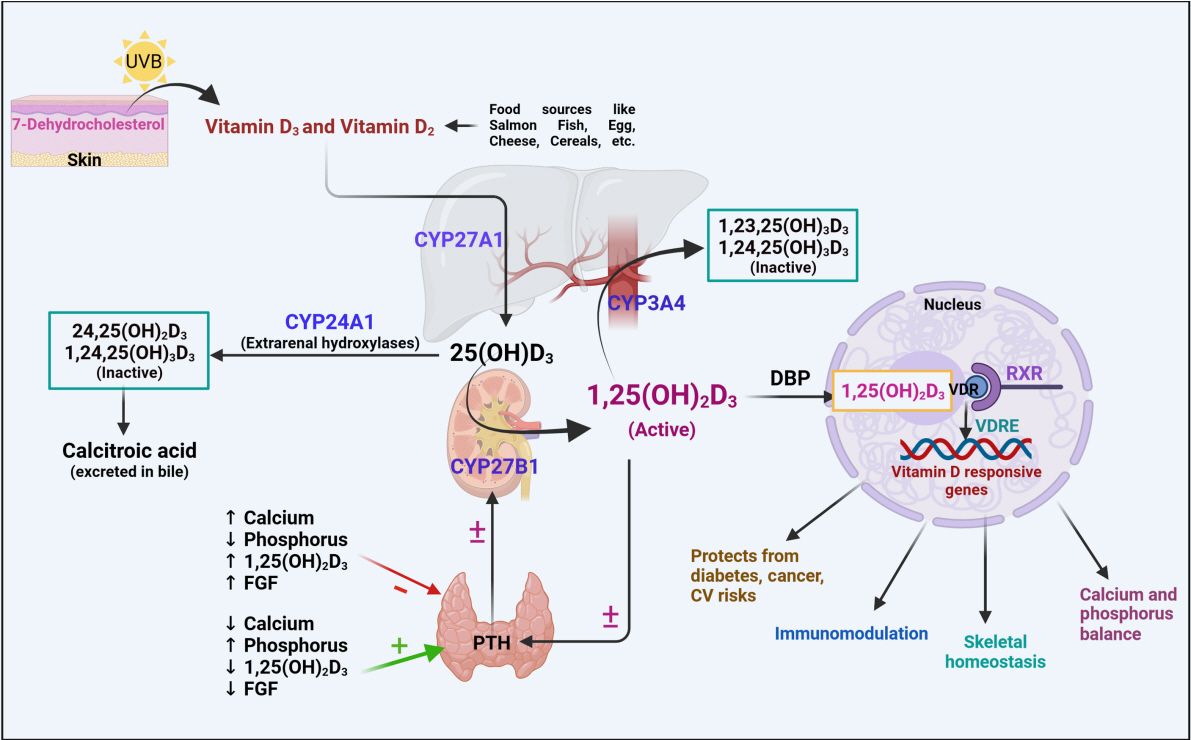
Article4. Jones G, Prosser DE, Kaufmann M. Cytochrome P450-mediated metabolism of vitamin D. J Lipid Res. 2014; 55:13–31.
Article5. Divanoglou N, Komninou D, Stea EA, Argiriou A, Papatzikas G, Tsakalof A, et al. Association of vitamin D receptor gene polymorphisms with serum vitamin D levels in a Greek rural population (Velestino Study). Lifestyle Genom. 2021; 14:81–90.
Article6. Mostafa WZ, Hegazy RA. Vitamin D and the skin: focus on a complex relationship: a review. J Adv Res. 2015; 6:793–804.
Article7. Wikvall K. Cytochrome P450 enzymes in the bioactivation of vitamin D to its hormonal form (review). Int J Mol Med. 2001; 7:201–9.
Article8. Omdahl JL, Bobrovnikova EA, Choe S, Dwivedi PP, May BK. Overview of regulatory cytochrome P450 enzymes of the vitamin D pathway. Steroids. 2001; 66:381–9.
Article9. Blomberg Jensen M, Andersen CB, Nielsen JE, Bagi P, Jorgensen A, Juul A, et al. Expression of the vitamin D receptor, 25-hydroxylases, 1alpha-hydroxylase and 24-hydroxylase in the human kidney and renal clear cell cancer. J Steroid Biochem Mol Biol. 2010; 121:376–82.10. Gottfried E, Rehli M, Hahn J, Holler E, Andreesen R, Kreutz M. Monocyte-derived cells express CYP27A1 and convert vitamin D3 into its active metabolite. Biochem Biophys Res Commun. 2006; 349:209–13.
Article11. Bosworth C, de Boer IH. Impaired vitamin D metabolism in CKD. Semin Nephrol. 2013; 33:158–68.
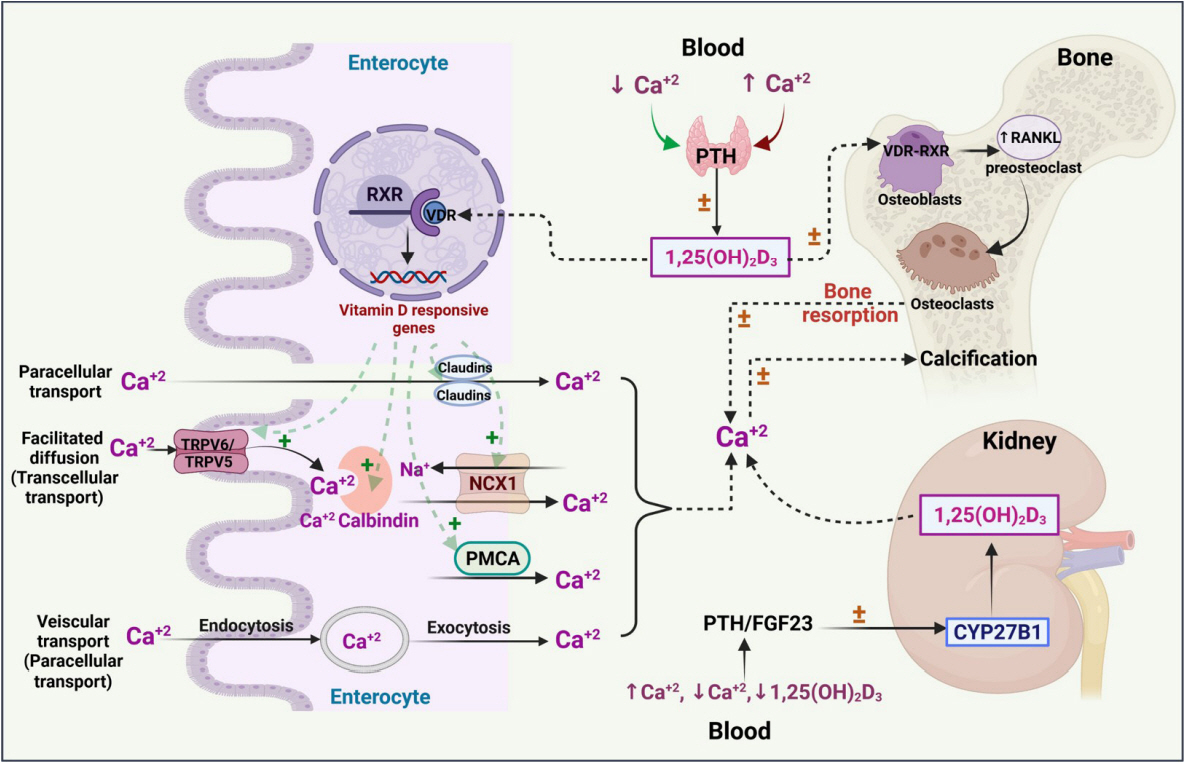
Article12. Christakos S, Dhawan P, Verstuyf A, Verlinden L, Carmeliet G. Vitamin D: metabolism, molecular mechanism of action, and pleiotropic effects. Physiol Rev. 2016; 96:365–408.
Article13. Piotrowska A, Wierzbicka J, Rybarczyk A, Tuckey RC, Slominski AT, Zmijewski MA. Vitamin D and its low calcemic analogs modulate the anticancer properties of cisplatin and dacarbazine in the human melanoma A375 cell line. Int J Oncol. 2019; 54:1481–95.
Article14. Wang Z, Lin YS, Zheng XE, Senn T, Hashizume T, Scian M, et al. An inducible cytochrome P450 3A4-dependent vitamin D catabolic pathway. Mol Pharmacol. 2012; 81:498–509.
Article15. Antunes MV, Timm TA, de Oliveira V, Staudt DE, Raymundo S, Gossling G, et al. Influence of CYP2D6 and CYP3A4 phenotypes, drug interactions, and vitamin D status on tamoxifen biotransformation. Ther Drug Monit. 2015; 37:733–44.
Article16. Kasarla SS, Dodoala S, Sampathi S, Talluri NK. Impact of chrysin on vitamin D and bone health: preclinical studies. bioRxiv. 2020; Nov. 20. [Preprint]. https://doi.org/10.1101/2020.11.19.390757.
Article17. Chang YT, Hernandez D, Alonso S, Gao M, Su M, Ghiaur G, et al. Role of CYP3A4 in bone marrow microenvironment-mediated protection of FLT3/ITD AML from tyrosine kinase inhibitors. Blood Adv. 2019; 3:908–16.
Article18. Wang Z, Lin YS, Dickmann LJ, Poulton EJ, Eaton DL, Lampe JW, et al. Enhancement of hepatic 4-hydroxylation of 25-hydroxyvitamin D3 through CYP3A4 induction in vitro and in vivo: implications for drug-induced osteomalacia. J Bone Miner Res. 2013; 28:1101–16.
Article19. Xu Y, Hashizume T, Shuhart MC, Davis CL, Nelson WL, Sakaki T, et al. Intestinal and hepatic CYP3A4 catalyze hydroxylation of 1alpha,25-dihydroxyvitamin D(3): implications for drug-induced osteomalacia. Mol Pharmacol. 2006; 69:56–65.
Article20. Prytula A, Cransberg K, Raes A. CYP3A4 is a crosslink between vitamin D and calcineurin inhibitors in solid organ transplant recipients: implications for bone health. Pharmacogenomics J. 2017; 17:481–7.
Article21. Hohmann N, Kocheise F, Carls A, Burhenne J, Weiss J, Haefeli WE, et al. Dose-dependent bioavailability and CYP3A inhibition contribute to non-linear pharmacokinetics of voriconazole. Clin Pharmacokinet. 2016; 55:1535–45.
Article22. Pravina P, Sayaji D, Avinash M. Calcium and its role in human body. Int J Res Pharm Biomed Sci. 2013; 4:659–68.23. Veldurthy V, Wei R, Oz L, Dhawan P, Jeon YH, Christakos S. Vitamin D, calcium homeostasis and aging. Bone Res. 2016; 4:16041.
Article24. Yang LP, Dong YP, Luo WT, Zhu T, Li QW, Zhang LJ, et al. Tissue-specific regulatory effects of vitamin D and its receptor on calbindin-D28K and calbindin-D9K. Biochem Mol Biol J. 2018; 4:23.25. Goltzman D, Mannstadt M, Marcocci C. Physiology of the calcium-parathyroid hormone-vitamin D axis. Front Horm Res. 2018; 50:1–13.
Article26. Comar CL, Bronner F; Calcium physiology. New York: Academic Press;1969. Chapter 7, Parathormone, calcitonin, and calcium homeostasis. p. 453–513.27. Cormier C. Genetic hypercalcemia. Joint Bone Spine. 2019; 86:459–66.
Article28. Khazai N, Judd SE, Tangpricha V. Calcium and vitamin D: skeletal and extraskeletal health. Curr Rheumatol Rep. 2008; 10:110–7.
Article29. Fleet JC. The role of vitamin D in the endocrinology controlling calcium homeostasis. Mol Cell Endocrinol. 2017; 453:36–45.
Article30. Reid IR, Bolland MJ. Controversies in medicine: the role of calcium and vitamin D supplements in adults. Med J Aust. 2019; 211:468–73.
Article31. Thacher TD, Fischer PR, Pettifor JM. Vitamin D treatment in calcium-deficiency rickets: a randomised controlled trial. Arch Dis Child. 2014; 99:807–11.
Article32. Rodan GA. Bone homeostasis. Proc Natl Acad Sci U S A. 1998; 95:13361–2.
Article33. Cashman KD. Diet, nutrition, and bone health. J Nutr. 2007; 137(11 Suppl):2507S–12S.
Article34. Bikle DD. Vitamin D and bone. Curr Osteoporos Rep. 2012; 10:151–9.
Article35. Bikle DD. Vitamin D metabolism, mechanism of action, and clinical applications. Chem Biol. 2014; 21:319–29.
Article36. St-Arnaud R. The direct role of vitamin D on bone homeostasis. Arch Biochem Biophys. 2008; 473:225–30.
Article37. Yoshizawa T, Handa Y, Uematsu Y, Takeda S, Sekine K, Yoshihara Y, et al. Mice lacking the vitamin D receptor exhibit impaired bone formation, uterine hypoplasia and growth retardation after weaning. Nat Genet. 1997; 16:391–6.
Article38. Takeyama K, Yamamoto Y, Kato S. VDR knockout mice and bone mineralization disorders. Clin Calcium. 2007; 17:1560–6.39. Burt LA, Billington EO, Rose MS, Raymond DA, Hanley DA, Boyd SK. Effect of high-dose vitamin D supplementation on volumetric bone density and bone strength: a randomized clinical trial. JAMA. 2019; 322:736–45.
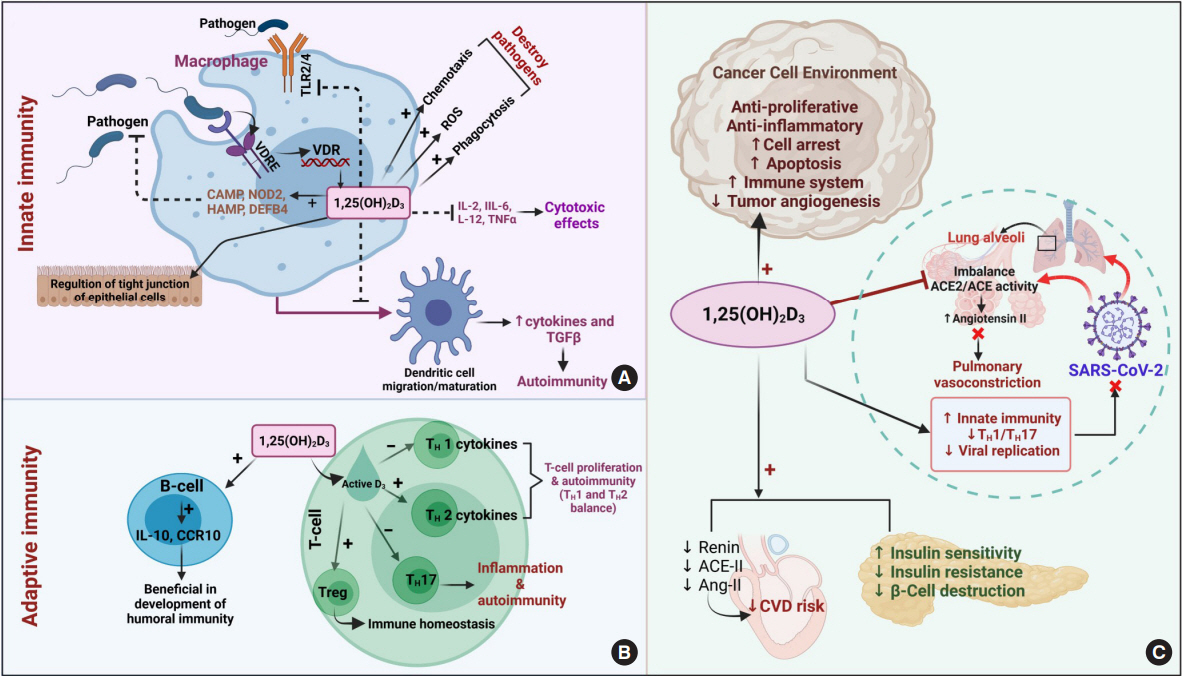
Article40. Aranow C. Vitamin D and the immune system. J Investig Med. 2011; 59:881–6.
Article41. Baeke F, Takiishi T, Korf H, Gysemans C, Mathieu C. Vitamin D: modulator of the immune system. Curr Opin Pharmacol. 2010; 10:482–96.
Article42. Yamamoto EA, Nguyen JK, Liu J, Keller E, Campbell N, Zhang CJ, et al. Low levels of vitamin D promote memory B cells in lupus. Nutrients. 2020; 12:291.
Article43. Cantorna MT, Snyder L, Lin YD, Yang L. Vitamin D and 1,25(OH)2D regulation of T cells. Nutrients. 2015; 7:3011–21.
Article44. Prietl B, Treiber G, Pieber TR, Amrein K. Vitamin D and immune function. Nutrients. 2013; 5:2502–21.
Article45. Lagishetty V, Liu NQ, Hewison M. Vitamin D metabolism and innate immunity. Mol Cell Endocrinol. 2011; 347:97–105.
Article46. Kawasaki T, Kawai T. Toll-like receptor signaling pathways. Front Immunol. 2014; 5:461.
Article47. Martinez-Moreno J, Hernandez JC, Urcuqui-Inchima S. Effect of high doses of vitamin D supplementation on dengue virus replication, Toll-like receptor expression, and cytokine profiles on dendritic cells. Mol Cell Biochem. 2020; 464:169–80.
Article48. van Splunter M, Perdijk O, Fick-Brinkhof H, Feitsma AL, Floris-Vollenbroek EG, Meijer B, et al. Bovine lactoferrin enhances TLR7-mediated responses in plasmacytoid dendritic cells in elderly women: results from a nutritional intervention study with bovine lactoferrin, GOS and vitamin D. Front Immunol. 2018; 9:2677.
Article49. Jeon SM, Shin EA. Exploring vitamin D metabolism and function in cancer. Exp Mol Med. 2018; 50:1–14.
Article50. Colston K, Colston MJ, Feldman D. 1,25-Dihydroxyvitamin D3 and malignant melanoma: the presence of receptors and inhibition of cell growth in culture. Endocrinology. 1981; 108:1083–6.
Article51. Mondul AM, Weinstein SJ, Layne TM, Albanes D. Vitamin D and cancer risk and mortality: state of the science, gaps, and challenges. Epidemiol Rev. 2017; 39:28–48.
Article52. Giammanco M, Di Majo D, La Guardia M, Aiello S, Crescimannno M, Flandina C, et al. Vitamin D in cancer chemoprevention. Pharm Biol. 2015; 53:1399–434.
Article53. Feldman D, Krishnan AV, Swami S, Giovannucci E, Feldman BJ. The role of vitamin D in reducing cancer risk and progression. Nat Rev Cancer. 2014; 14:342–57.
Article54. Sun M, Zhang Q, Yang X, Qian SY, Guo B. Vitamin D enhances the efficacy of irinotecan through miR-627-mediated inhibition of intratumoral drug metabolism. Mol Cancer Ther. 2016; 15:2086–95.
Article55. Gupta RP, Hollis BW, Patel SB, Patrick KS, Bell NH. CYP3A4 is a human microsomal vitamin D 25-hydroxylase. J Bone Miner Res. 2004; 19:680–8.
Article56. Chen J, Zhao KN, Chen C. The role of CYP3A4 in the biotransformation of bile acids and therapeutic implication for cholestasis. Ann Transl Med. 2014; 2:7.57. Hernandez LM, Blazer DG; Institute of Medicine. Genes, behavior, and the social environment: moving beyond the nature/nurture debate. Washington, DC: National Academies Press;2006.
Article58. Tracy TS, Chaudhry AS, Prasad B, Thummel KE, Schuetz EG, Zhong XB, et al. Interindividual variability in cytochrome P450-mediated drug metabolism. Drug Metab Dispos. 2016; 44:343–51.
Article59. Kang YS, Park SY, Yim CH, Kwak HS, Gajendrarao P, Krishnamoorthy N, et al. The CYP3A4*18 genotype in the cytochrome P450 3A4 gene, a rapid metabolizer of sex steroids, is associated with low bone mineral density. Clin Pharmacol Ther. 2009; 85:312–8.
Article60. Kondratyeva EI, Zakharova IN, Ilenkova NA, Klimov LY, Petrova NV, Zodbinova AE, et al. Vitamin D status in Russian children and adolescents: contribution of genetic and exogenous factors. Front Pediatr. 2020; 8:583206.
Article61. Jin T, Yang H, Zhang J, Yunus Z, Sun Q, Geng T, et al. Polymorphisms and phenotypic analysis of cytochrome P450 3A4 in the Uygur population in northwest China. Int J Clin Exp Pathol. 2015; 8:7083–91.62. Nylen H, Habtewold A, Makonnen E, Yimer G, Bertilsson L, Burhenne J, et al. Prevalence and risk factors for efavirenz-based antiretroviral treatment-associated severe vitamin D deficiency: a prospective cohort study. Medicine (Baltimore). 2016; 95:e4631.63. Dally H, Edler L, Jager B, Schmezer P, Spiegelhalder B, Dienemann H, et al. The CYP3A4*1B allele increases risk for small cell lung cancer: effect of gender and smoking dose. Pharmacogenetics. 2003; 13:607–18.
Article64. Fohner AE, Dalton R, Skagen K, Jackson K, Claw KG, Hopkins SE, et al. Characterization of CYP3A pharmacogenetic variation in American Indian and Alaska Native communities, targeting CYP3A4*1G allele function. Clin Transl Sci. 2021; 14:1292–302.
Article65. Amrein K, Scherkl M, Hoffmann M, Neuwersch-Sommeregger S, Kostenberger M, Tmava Berisha A, et al. Vitamin D deficiency 2.0: an update on the current status worldwide. Eur J Clin Nutr. 2020; 74:1498–513.
Article66. Grant WB. Epidemiology of disease risks in relation to vitamin D insufficiency. Prog Biophys Mol Biol. 2006; 92:65–79.
Article67. Roizen JD, Li D, O’Lear L, Javaid MK, Shaw NJ, Ebeling PR, et al. CYP3A4 mutation causes vitamin D-dependent rickets type 3. J Clin Invest. 2018; 128:1913–8.
Article68. Robien K, Butler LM, Wang R, Beckman KB, Walek D, Koh WP, et al. Genetic and environmental predictors of serum 25-hydroxyvitamin D concentrations among middle-aged and elderly Chinese in Singapore. Br J Nutr. 2013; 109:493–502.
Article69. Zeigler-Johnson C, Friebel T, Walker AH, Wang Y, Spangler E, Panossian S, et al. CYP3A4, CYP3A5, and CYP3A43 genotypes and haplotypes in the etiology and severity of prostate cancer. Cancer Res. 2004; 64:8461–7.70. Reyes-Hernandez OD, Vega L, Jimenez-Rios MA, Martinez-Cervera PF, Lugo-Garcia JA, Hernandez-Cadena L, et al. The PXR rs7643645 polymorphism is associated with the risk of higher prostate-specific antigen levels in prostate cancer patients. PLoS One. 2014; 9:e99974.
Article71. Gervasini G, Garcia-Martin E, Ladero JM, Pizarro R, Sastre J, Martinez C, et al. Genetic variability in CYP3A4 and CYP3A5 in primary liver, gastric and colorectal cancer patients. BMC Cancer. 2007; 7:118.72. Liu X, Huang X, Zhang S, Niu F, Ouyang Y, Shou Z, et al. Correlations between CYP3A4 polymorphism and susceptibility to breast cancer in Chinese Han population. Int J Clin Oncol. 2019; 24:179–88.
Article73. Fiszer-Maliszewska L, Laczmanski L, Dolinska A, Jagas M, Kolodziejska E, Jankowska M, et al. Polymorphisms of ABCB1, CYP3A4 and CYP3A5 genes in ovarian cancer and treatment response in poles. Anticancer Res. 2018; 38:1455–9.74. Chau CH, Price DK, Till C, Goodman PJ, Chen X, Leach RJ, et al. Finasteride concentrations and prostate cancer risk: results from the Prostate Cancer Prevention Trial. PLoS One. 2015; 10:e0126672.
Article75. He BX, Shi L, Qiu J, Tao L, Li R, Yang L, et al. A functional polymorphism in the CYP3A4 gene is associated with increased risk of coronary heart disease in the Chinese Han population. Basic Clin Pharmacol Toxicol. 2011; 108:208–13.
Article76. Jeovanio-Silva AL, Monteiro TP, El-Jaick KB, do Brasil PE, Rolla VC, de Castro L. Unique CYP3A4 genetic variant in Brazilian tuberculosis patients with/without HIV. Mol Med Rep. 2012; 5:153–61.77. Baker SD, Verweij J, Cusatis GA, van Schaik RH, Marsh S, Orwick SJ, et al. Pharmacogenetic pathway analysis of docetaxel elimination. Clin Pharmacol Ther. 2009; 85:155–63.
Article78. Zhou XY, Hu XX, Wang CC, Lu XR, Chen Z, Liu Q, et al. Enzymatic activities of CYP3A4 allelic variants on quinine 3-hydroxylation in vitro. Front Pharmacol. 2019; 10:591.
Article79. Zhou LP, Yao F, Luan H, Wang YL, Dong XH, Zhou WW, et al. CYP3A4*1B polymorphism and cancer risk: a HuGE review and meta-analysis. Tumour Biol. 2013; 34:649–60.
Article80. Kuehl P, Zhang J, Lin Y, Lamba J, Assem M, Schuetz J, et al. Sequence diversity in CYP3A promoters and characterization of the genetic basis of polymorphic CYP3A5 expression. Nat Genet. 2001; 27:383–91.
Article81. Hamzeiy H, Vahdati-Mashhadian N, Edwards HJ, Goldfarb PS. Mutation analysis of the human CYP3A4 gene 5’ regulatory region: population screening using non-radioactive SSCP. Mutat Res. 2002; 500:103–10.
Article82. Huang Y, Wen G, Lu Y, Wen J, Ji Y, Xing X, et al. CYP3A4*1G and CYP3A5*3 genetic polymorphisms alter the antihypertensive efficacy of amlodipine in patients with hypertension following renal transplantation. Int J Clin Pharmacol Ther. 2017; 55:109–18.
Article83. Gao Y, Zhang LR, Fu Q. CYP3A4*1G polymorphism is associated with lipid-lowering efficacy of atorvastatin but not of simvastatin. Eur J Clin Pharmacol. 2008; 64:877–82.
Article84. Fukushima-Uesaka H, Saito Y, Watanabe H, Shiseki K, Saeki M, Nakamura T, et al. Haplotypes of CYP3A4 and their close linkage with CYP3A5 haplotypes in a Japanese population. Hum Mutat. 2004; 23:100.
Article85. Liu L, Chang Y, Du S, Shi X, Yang H, Kang L, et al. Genetic analysis of drug metabolizing phase-I enzymes CYP3A4 in Tibetan populations. J Genet. 2017; 96:219–25.
Article86. Miyazaki M, Nakamura K, Fujita Y, Guengerich FP, Horiuchi R, Yamamoto K. Defective activity of recombinant cytochromes P450 3A4.2 and 3A4.16 in oxidation of midazolam, nifedipine, and testosterone. Drug Metab Dispos. 2008; 36:2287–91.
Article87. Guttman Y, Nudel A, Kerem Z. Polymorphism in cytochrome P450 3A4 is ethnicity related. Front Genet. 2019; 10:224.
Article88. Lamba JK, Lin YS, Thummel K, Daly A, Watkins PB, Strom S, et al. Common allelic variants of cytochrome P4503A4 and their prevalence in different populations. Pharmacogenetics. 2002; 12:121–32.
Article89. Fang P, Tang PF, Xu RA, Zheng X, Wen J, Bao SS, et al. Functional assessment of CYP3A4 allelic variants on lidocaine metabolism in vitro. Drug Des Devel Ther. 2017; 11:3503–10.90. Tang PF, Zheng X, Hu XX, Yang CC, Chen Z, Qian JC, et al. Functional measurement of CYP2C9 and CYP3A4 allelic polymorphism on sildenafil metabolism. Drug Des Devel Ther. 2020; 14:5129–41.91. Wang A, Yu BN, Luo CH, Tan ZR, Zhou G, Wang LS, et al. Ile118Val genetic polymorphism of CYP3A4 and its effects on lipid-lowering efficacy of simvastatin in Chinese hyperlipidemic patients. Eur J Clin Pharmacol. 2005; 60:843–8.
Article92. Hsieh KP, Lin YY, Cheng CL, Lai ML, Lin MS, Siest JP, et al. Novel mutations of CYP3A4 in Chinese. Drug Metab Dispos. 2001; 29:268–73.93. Varenhorst C, Eriksson N, Johansson A, Barratt BJ, Hagstrom E, Akerblom A, et al. Effect of genetic variations on ticagrelor plasma levels and clinical outcomes. Eur Heart J. 2015; 36:1901–12.
Article94. Liao Q, Chen DJ, Zhang F, Li L, Hu R, Tang YZ, et al. Effect of CYP3A4*18B polymorphisms and interactions with OPRM1 A118G on postoperative fentanyl requirements in patients undergoing radical gastrectomy. Mol Med Rep. 2013; 7:901–8.
Article95. Dai D, Tang J, Rose R, Hodgson E, Bienstock RJ, Mohrenweiser HW, et al. Identification of variants of CYP3A4 and characterization of their abilities to metabolize testosterone and chlorpyrifos. J Pharmacol Exp Ther. 2001; 299:825–31.96. Apellaniz-Ruiz M, Inglada-Perez L, Naranjo ME, Sanchez L, Mancikova V, Curras-Freixes M, et al. High frequency and founder effect of the CYP3A4*20 loss-of-function allele in the Spanish population classifies CYP3A4 as a polymorphic enzyme. Pharmacogenomics J. 2015; 15:288–92.
Article97. Teft WA, Gong IY, Dingle B, Potvin K, Younus J, Vandenberg TA, et al. CYP3A4 and seasonal variation in vitamin D status in addition to CYP2D6 contribute to therapeutic endoxifen level during tamoxifen therapy. Breast Cancer Res Treat. 2013; 139:95–105.
Article98. Holmberg MT, Tornio A, Paile-Hyvarinen M, Tarkiainen EK, Neuvonen M, Neuvonen PJ, et al. CYP3A4*22 impairs the elimination of ticagrelor, but has no significant effect on the bioactivation of clopidogrel or prasugrel. Clin Pharmacol Ther. 2019; 105:448–57.99. Hu GX, Dai DP, Wang H, Huang XX, Zhou XY, Cai J, et al. Systematic screening for CYP3A4 genetic polymorphisms in a Han Chinese population. Pharmacogenomics. 2017; 18:369–79.
Article100. Scheibner A, Remmel R, Schladt D, Oetting WS, Guan W, Wu B, et al. Tacrolimus elimination in four patients with a CYP3A5*3/*3 CYP3A4*22/*22 genotype combination. Pharmacotherapy. 2018; 38:e46–52.101. Zhou Q, Yu X, Shu C, Cai Y, Gong W, Wang X, et al. Analysis of CYP3A4 genetic polymorphisms in Han Chinese. J Hum Genet. 2011; 56:415–22.
Article102. Hawkes CP, Li D, Hakonarson H, Meyers KE, Thummel KE, Levine MA. CYP3A4 induction by rifampin: an alternative pathway for vitamin D inactivation in patients with CYP24A1 mutations. J Clin Endocrinol Metab. 2017; 102:1440–6.
Article103. Bozina N, Bradamante V, Lovric M. Genetic polymorphism of metabolic enzymes P450 (CYP) as a susceptibility factor for drug response, toxicity, and cancer risk. Arh Hig Rada Toksikol. 2009; 60:217–42.
Article104. Stone A, Ratnasinghe LD, Emerson GL, Modali R, Lehman T, Runnells G, et al. CYP3A43 Pro(340)Ala polymorphism and prostate cancer risk in African Americans and Caucasians. Cancer Epidemiol Biomarkers Prev. 2005; 14:1257–61.
Article105. Wilson RT, Masters LD, Barnholtz-Sloan JS, Salzberg AC, Hartman TJ. Ancestry-adjusted vitamin D metabolite concentrations in association with cytochrome P450 3A polymorphisms. Am J Epidemiol. 2018; 187:754–66.
Article106. Fohner A, Muzquiz LI, Austin MA, Gaedigk A, Gordon A, Thornton T, et al. Pharmacogenetics in American Indian populations: analysis of CYP2D6, CYP3A4, CYP3A5, and CYP2C9 in the Confederated Salish and Kootenai Tribes. Pharmacogenet Genomics. 2013; 23:403–14.107. Saiz-Rodriguez M, Almenara S, Navares-Gomez M, Ochoa D, Roman M, Zubiaur P, et al. Effect of the most relevant CYP3A4 and CYP3A5 polymorphisms on the pharmacokinetic parameters of 10 CYP3A substrates. Biomedicines. 2020; 8:94.
Article108. Espindola LM, Lopez MJ, Flores AU, Espinosa LR, Granados J, Pacheco JL, et al. Genetic polymorphism of CYP3A4 is associated with poor response to ifosfamide treatment in children with solid embryonic tumors. Arch Med Sci. 2019; 17:1766–71.109. Sidiropoulou P, Docea AO, Nikolaou V, Katsarou MS, Spandidos DA, Tsatsakis A, et al. Unraveling the roles of vitamin D status and melanin during COVID-19 (Review). Int J Mol Med. 2021; 47:92–100.
Article110. Somasundaram NP, Ranathunga I, Ratnasamy V, Wijewickrama PS, Dissanayake HA, Yogendranathan N, et al. The impact of SARS-Cov-2 virus infection on the endocrine system. J Endocr Soc. 2020; 4:bvaa082.
Article111. Salem Hareedy M, Rashad SM, Hetta HF, Hassanien SM, Abdellatif H, Hassanien M. CYP2D6 and CYP3A4 variants influence the risk and outcome of COVID-19 infection among rheumatoid arthritis patients maintained on hydroxychloroquine. Drug Metab Pers Ther. 2021; Mar. 26. [Epub]. https://doi.org/10.1515/dmdi-2020-0164.
Article112. Wang J, Ji H, Jia H, Guan D. Association between CYP3A4 gene rs4646437 polymorphism and the risk of hypertension in Chinese population: a case-control study. Biosci Rep. 2019; 39:BSR20190296.
Article113. Gao N, Tang H, Gao L, Tu G, Luo H, Xia Y. CYP3A4 and CYP11A1 variants are risk factors for ischemic stroke: a case control study. BMC Neurol. 2020; 20:77.
Article114. Liu R, Zhou ZY, Chen YB, Li JL, Yu WB, Chen XM, et al. Associations of CYP3A4, NR1I2, CYP2C19 and P2RY12 polymorphisms with clopidogrel resistance in Chinese patients with ischemic stroke. Acta Pharmacol Sin. 2016; 37:882–8.
Article115. Zhang W, Chang YZ, Kan QC, Zhang LR, Li ZS, Lu H, et al. CYP3A4*1G genetic polymorphism influences CYP3A activity and response to fentanyl in Chinese gynecologic patients. Eur J Clin Pharmacol. 2010; 66:61–6.
Article116. Sapkota BR, Hopkins R, Bjonnes A, Ralhan S, Wander GS, Mehra NK, et al. Genome-wide association study of 25(OH) vitamin D concentrations in Punjabi Sikhs: results of the Asian Indian diabetic heart study. J Steroid Biochem Mol Biol. 2016; 158:149–56.
Article117. Fujita K. Food-drug interactions via human cytochrome P450 3A (CYP3A). Drug Metabol Drug Interact. 2004; 20:195–217.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A review of vitamin D deficiency and vitamin D receptor polymorphisms in endocrine-related disorders
- Premature Ejaculation and Endocrine Disorders: A Literature Review
- Vitamin D Status and Vitamin D Receptor Gene Polymorphisms Are Associated with Pelvic Floor Disorders in Women
- Vitamin D receptor gene polymorphisms in breast cancer
- Endocrine disorders and the neurologic manifestations