Korean Circ J.
2022 Jul;52(7):485-495. 10.4070/kcj.2022.0093.
Strategies for Renal Protection in Cardiovascular Interventions
- Affiliations
-
- 1The Heart Center, St. Francis Hospital, Roslyn, NY, USA
- 2DeMatteis Cardiovascular Institute, St. Francis Hospital, Roslyn, NY, USA
- 3Clinical Trials Center, Cardiovascular Research Foundation, New York, NY, USA
- 4Hospital Clinico San Carlos IDISSC and Universidad Complutense de Madrid, Madrid, Spain
- 5Institute of Cardiology, Jagiellonian University, Krakow, Poland
- 6Maria Cecilia Hospital, GVM Care & Research, Cotignola (RA), Ravenna, Italy
- 7Division of Nephrology, Department of Medicine, Vagelos College of Physicians & Surgeons, Columbia University, New York, NY, USA
- 8Heart Research Institute, Sydney, Australia
- 9Sydney Medical School, Faculty of Medicine and Health, The University of Sydney, Sydney, Australia
- KMID: 2530730
- DOI: http://doi.org/10.4070/kcj.2022.0093
Abstract
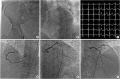
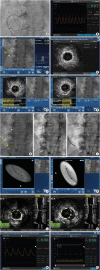
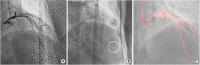
- Coronary artery disease is highly prevalent in chronic kidney disease (CKD) and is a risk factor for contrast-associated acute kidney injury (CA-AKI), a complication of cardiovascular procedures that require contrast administration (e.g., coronary angiography, percutaneous coronary intervention [PCI]). CA-AKI has a major impact on morbidity, mortality, and healthcare resource utilization. The incidence of CA-AKI is particularly high in patients with pre-existing CKD, advanced age and comorbidities that increase the likelihood of CKD. The focus of the present review is to provide a brief overview on the assessment of the risk for and prevention of CA-AKI in patients undergoing angiography and PCI, including recognition of the important patient- and procedure-related factors that may contribute to CA-AKI. Preventive and treatment strategies, the mainstay of which is volume repletion by normal saline, are briefly discussed. The main focus of the review is placed on technical details of contrast minimization techniques, including ultra-low contrast angiography and zerocontrast PCI. Operator competence in such techniques is important to ensure that procedural challenges in patients with CKD, like vessel calcification, multivessel disease and complex anatomical subsets, are effectively addressed by PCI while minimizing the risk of CA-AKI.
Keyword
Figure
Reference
-
1. Nash K, Hafeez A, Hou S. Hospital-acquired renal insufficiency. Am J Kidney Dis. 2002; 39:930–936. PMID: 11979336.
Article2. Mozaffarian D, Benjamin EJ, Go AS, et al. Executive summary: heart disease and stroke statistics--2016 update: a report from the American Heart Association. Circulation. 2016; 133:447–454. PMID: 26811276.3. Solomon R. Contrast media: are there differences in nephrotoxicity among contrast media? BioMed Res Int. 2014; 2014:934947. PMID: 24587997.
Article4. Weisbord SD, Palevsky PM. Contrast-induced acute kidney injury: short- and long-term implications. Semin Nephrol. 2011; 31:300–309. PMID: 21784279.
Article5. Tong J, Li H, Zhang H, et al. Neutrophil gelatinase-associated lipocalin in the prediction of contrast-induced nephropathy: a systemic review and meta-analysis. J Cardiovasc Pharmacol. 2015; 66:239–245. PMID: 25915514.
Article6. Chertow GM, Normand SL, McNeil BJ. “Renalism”: inappropriately low rates of coronary angiography in elderly individuals with renal insufficiency. J Am Soc Nephrol. 2004; 15:2462–2468. PMID: 15339996.
Article7. Section 2: AKI definition. Kidney Int Suppl (2011). 2012; 2:19–36. PMID: 25018918.8. van der Molen AJ, Reimer P, Dekkers IA, et al. Post-contrast acute kidney injury - Part 1: definition, clinical features, incidence, role of contrast medium and risk factors: recommendations for updated ESUR Contrast Medium Safety Committee guidelines. Eur Radiol. 2018; 28:2845–2855. PMID: 29426991.
Article9. Budano C, Levis M, D’Amico M, et al. Impact of contrast-induced acute kidney injury definition on clinical outcomes. Am Heart J. 2011; 161:963–971. PMID: 21570530.
Article10. Slocum NK, Grossman PM, Moscucci M, et al. The changing definition of contrast-induced nephropathy and its clinical implications: insights from the Blue Cross Blue Shield of Michigan Cardiovascular Consortium (BMC2). Am Heart J. 2012; 163:829–834. PMID: 22607861.
Article11. Mehran R, Aymong ED, Nikolsky E, et al. A simple risk score for prediction of contrast-induced nephropathy after percutaneous coronary intervention: development and initial validation. J Am Coll Cardiol. 2004; 44:1393–1399. PMID: 15464318.
Article12. Fähling M, Seeliger E, Patzak A, Persson PB. Understanding and preventing contrast-induced acute kidney injury. Nat Rev Nephrol. 2017; 13:169–180. PMID: 28138128.
Article13. Tziakas D, Chalikias G, Stakos D, et al. Validation of a new risk score to predict contrast-induced nephropathy after percutaneous coronary intervention. Am J Cardiol. 2014; 113:1487–1493. PMID: 24630389.
Article14. Gurm HS, Seth M, Dixon SR, et al. Contemporary use of and outcomes associated with ultra-low contrast volume in patients undergoing percutaneous coronary interventions. Catheter Cardiovasc Interv. 2019; 93:222–230. PMID: 30144337.
Article15. Ranucci M, Castelvecchio S, Menicanti L, Frigiola A, Pelissero G. Risk of assessing mortality risk in elective cardiac operations: age, creatinine, ejection fraction, and the law of parsimony. Circulation. 2009; 119:3053–3061. PMID: 19506110.
Article16. Liu YH, Liu Y, Zhou YL, et al. Comparison of different risk scores for predicting contrast induced nephropathy and outcomes after primary percutaneous coronary intervention in patients with ST elevation myocardial infarction. Am J Cardiol. 2016; 117:1896–1903. PMID: 27161818.
Article17. Mohebi R, Karimi Galougahi K, Garcia JJ, et al. Long-term clinical impact of contrast-associated acute kidney injury following PCI: an ADAPT-DES Substudy. JACC Cardiovasc Interv. 2022; 15:753–766. PMID: 35305904.
Article18. Isaka Y, Hayashi H, Aonuma K, et al. Guideline on the use of iodinated contrast media in patients with kidney disease 2018. Clin Exp Nephrol. 2020; 24:1–44.
Article19. Brar SS, Aharonian V, Mansukhani P, et al. Haemodynamic-guided fluid administration for the prevention of contrast-induced acute kidney injury: the POSEIDON randomised controlled trial. Lancet. 2014; 383:1814–1823. PMID: 24856027.
Article20. Khwaja A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract. 2012; 120:c179–c184. PMID: 22890468.
Article21. Ali ZA, Karimi Galougahi K, Nazif T, et al. Imaging- and physiology-guided percutaneous coronary intervention without contrast administration in advanced renal failure: a feasibility, safety, and outcome study. Eur Heart J. 2016; 37:3090–3095. PMID: 26957421.
Article22. Nyman U, Björk J, Aspelin P, Marenzi G. Contrast medium dose-to-GFR ratio: a measure of systemic exposure to predict contrast-induced nephropathy after percutaneous coronary intervention. Acta Radiol. 2008; 49:658–667. PMID: 18568558.
Article23. Rahim HM, Flattery E, Gkargkoulas F, et al. Ultra-low-contrast angiography in patients with advanced chronic kidney disease and previous coronary artery bypass surgery. Coron Artery Dis. 2019; 30:346–351. PMID: 31094895.
Article24. Bhatti N, Rahim H, Chen S, et al. Ultra-low contrast coronary angiography in patients with advanced chronic kidney disease: feasibility and outcomes compared with conventional angiography. J Am Coll Cardiol. 2019; 74:B30.25. Karimi Galougahi K, Zalewski A, Leon MB, Karmpaliotis D, Ali ZA. Optical coherence tomography-guided percutaneous coronary intervention in pre-terminal chronic kidney disease with no radio-contrast administration. Eur Heart J. 2016; 37:1059. PMID: 26685137.
Article26. Rahim H, Flattery E, Gkargkoulas F, et al. Clinical outcomes of imaging- and physiology-guided PCI without contrast administration in advanced renal failure. J Am Coll Cardiol. 2019; 74:B32.27. Karimi Galougahi K, Mintz GS, Karmpaliotis D, Ali ZA. Zero-contrast percutaneous coronary intervention on calcified lesions facilitated by rotational atherectomy. Catheter Cardiovasc Interv. 2017; 90:E85–E89. PMID: 28303645.
Article28. Karimi Galougahi K, Patel S, Shlofmitz RA, et al. Calcific plaque modification by acoustic shockwaves - intravascular lithotripsy in coronary interventions. Circ Cardiovasc Interv. 2021; 14:e009354. PMID: 32907343.29. Hatem R, Finn MT, Riley RF, et al. Zero contrast retrograde chronic total occlusions percutaneous coronary intervention: a case series. Eur Heart J Case Rep. 2018; 2:1–5.
Article30. Madhavan MV, Prasad M, Fall KN, Rahim H, Ali ZA. Zero-contrast multivessel revascularization for acute coronary syndrome in a patient with chronic kidney disease. JACC Case Rep. 2019; 1:774–780. PMID: 34316930.
Article31. Parviz Y, Fall K, Stone GW, et al. Imaging and physiology to guide venous graft interventions without contrast administration in advanced renal failure. J Invasive Cardiol. 2017; 29:E163–E165. PMID: 29086735.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Adequacy of Ambulatory Hemodynamic Assessments for Reducing All-Cause Mortality in Individuals With Heart Failure
- Bioengineering the Heart: Harnessing Biomaterials for Innovative Cardiovascular Interventions
- The Role of Distal Protection Devices for Cardiovascular Intervention
- Vascular Access: The Alpha and Omega of Cardiovascular Intervention
- RSV Prevention Strategies in Korean Children: A Review of Current Approaches and Emerging Options