Nutr Res Pract.
2022 Jun;16(3):330-343. 10.4162/nrp.2022.16.3.330.
Induction of apoptotic cell death in human bladder cancer cells by ethanol extract of Zanthoxylum schinifolium leaf, through ROS-dependent inactivation of the PI3K/Akt signaling pathway
- Affiliations
-
- 1Division of Basic Sciences, College of Liberal Studies, Dong-eui University, Busan 47340, Korea
- 2Anti-Aging Research Center, Dong-eui University, Busan 47340, Korea
- 3Department of Biochemistry, College of Korean Medicine, Dong-eui University, Busan 47227, Korea
- 4Korea Nanobiotechnology Center, Pusan National University, Busan 46241, Korea
- 5Nakdonggang National Institute of Biological Resources, Sangju 17104, Korea
- 6National Marine Biodiversity Institute of Korea, Seocheon 33662, Korea
- 7Department of Food and Nutrition, Chung-Ang University, Ansung 17546, Korea
- 8Department of Urology, College of Medicine, Chungbuk National University, Cheongju 28644, Korea
- 9Department of Marine Life Science, Jeju National University, Jeju 63243, Korea
- 10Department of Food and Nutrition, College of Nursing, Healthcare Sciences & Human Ecology, Dong-eui University, Busan 47340, Korea
- KMID: 2530394
- DOI: http://doi.org/10.4162/nrp.2022.16.3.330
Abstract
- BACKGROUND/OBJECTIVES
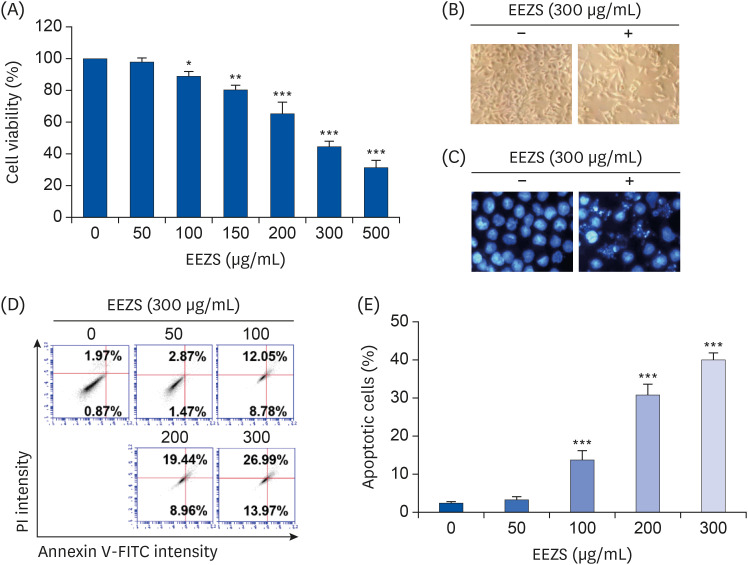
Zanthoxylum schinifolium is traditionally used as a spice for cooking in East Asian countries. This study was undertaken to evaluate the anti-proliferative potential of ethanol extracts of Z. schinifolium leaves (EEZS) against human bladder cancer T24 cells.
MATERIALS/METHODS
Subsequent to measuring the cytotoxicity of EEZS, the anti-cancer activity was measured by assessing apoptosis induction, reactive oxygen species (ROS) generation, and mitochondrial membrane potential (MMP). In addition, we determined the underlying mechanism of EEZS-induced apoptosis through various assays, including Western blot analysis.
RESULTS
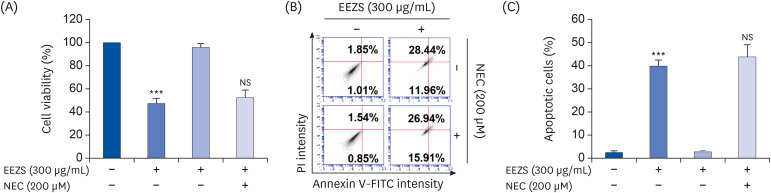
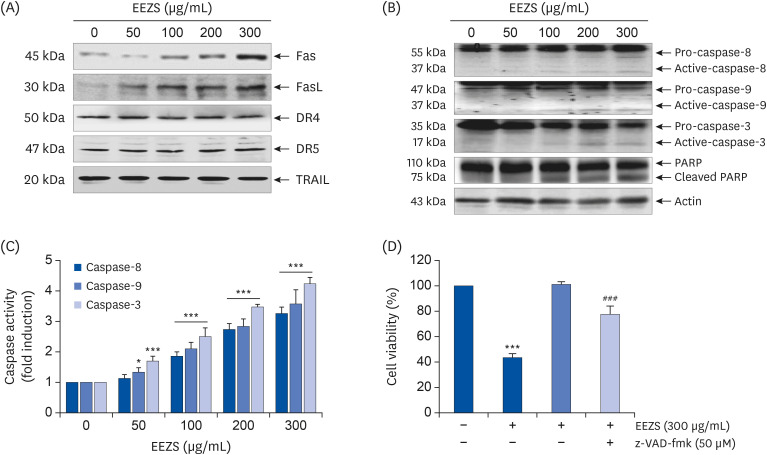
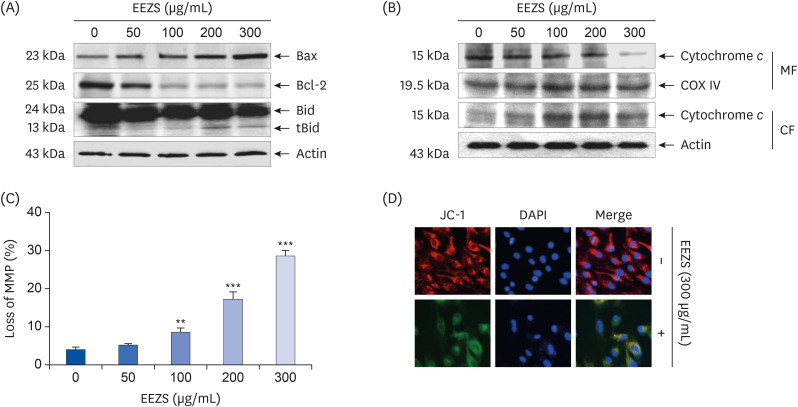
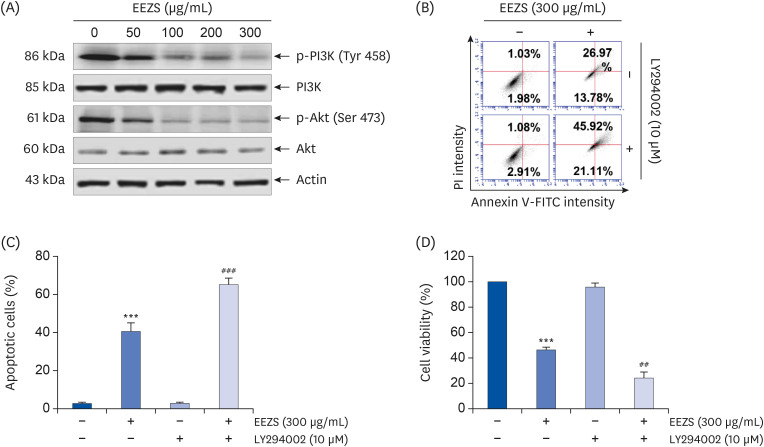
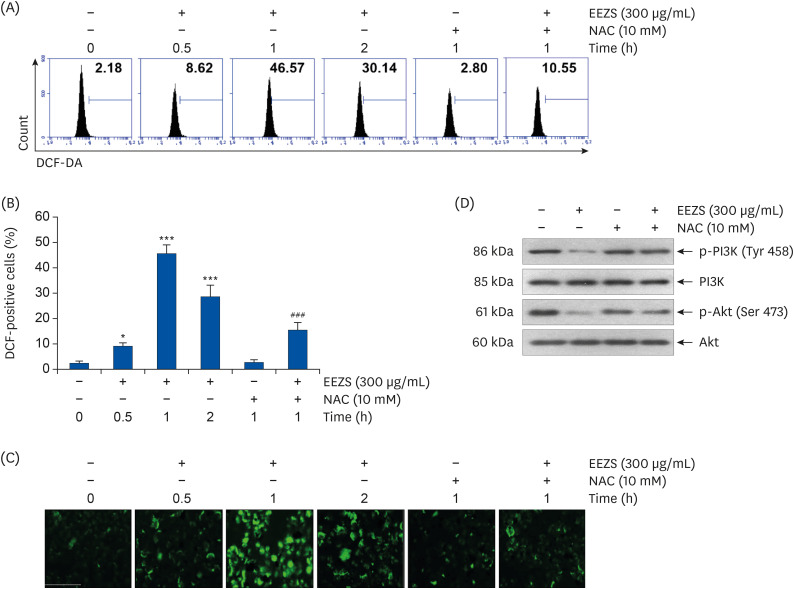
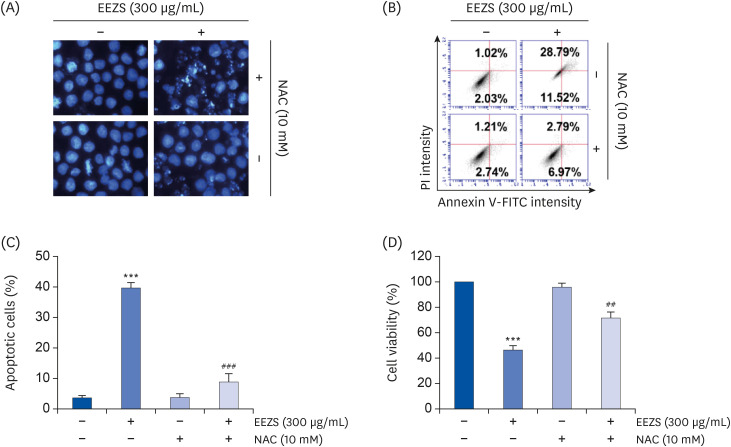
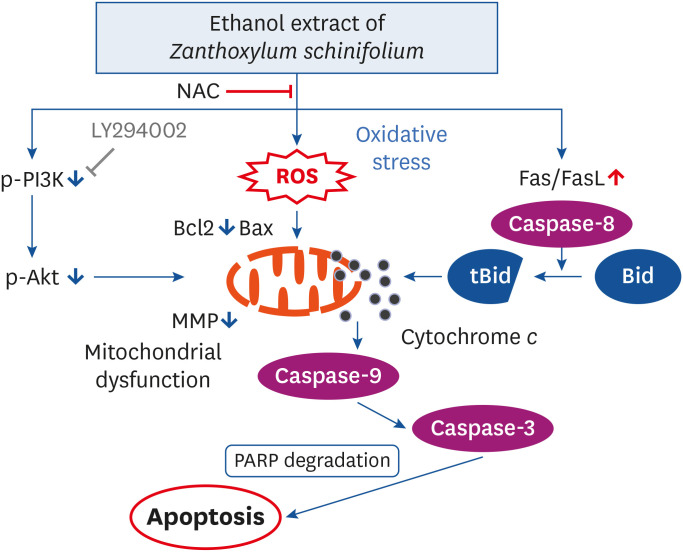
EEZS treatment concentration-dependently inhibited T24 cell survival, which is associated with apoptosis induction. Exposure to EEZS induced the expression of Fas and Fas-ligand, activated caspases, and subsequently resulted to cleavage of poly (ADPribose) polymerase. EEZS also enhanced the expression of cytochrome c in the cytoplasm by suppressing MMP, following increase in the ratio of Bax:Bcl-2 expression and truncation of Bid. However, EEZS-mediated growth inhibition and apoptosis were significantly diminished by a pan-caspase inhibitor. Moreover, EEZS inhibited activation of the phosphoinositide 3-kinase (PI3K)/Akt pathway, and the apoptosis-inducing potential of EEZS was promoted in the presence of PI3K/Akt inhibitor. In addition, EEZS enhanced the production of ROS, whereas N-acetyl cysteine (NAC), a ROS scavenger, markedly suppressed growth inhibition and inactivation of the PI3K/Akt signaling pathway induced by EEZS. Furthermore, NAC significantly attenuated the EEZS-induced apoptosis and reduction of cell viability.
CONCLUSIONS
Taken together, our results indicate that exposure to EEZS exhibits anticancer activity in T24 bladder cancer cells through ROS-dependent induction of apoptosis and inactivation of the PI3K/Akt signaling pathway.
Keyword
Figure
Reference
-
1. Kiraz Y, Adan A, Kartal Yandim M, Baran Y. Major apoptotic mechanisms and genes involved in apoptosis. Tumour Biol. 2016; 37:8471–8486. PMID: 27059734.2. Pfeffer CM, Singh ATK. Apoptosis: a target for anticancer therapy. Int J Mol Sci. 2018; 19:448.3. Hassan M, Watari H, AbuAlmaaty A, Ohba Y, Sakuragi N. Apoptosis and molecular targeting therapy in cancer. Biomed Res Int. 2014; 2014:150845. PMID: 25013758.4. Birkinshaw RW, Czabotar PE. The BCL-2 family of proteins and mitochondrial outer membrane permeabilisation. Semin Cell Dev Biol. 2017; 72:152–162. PMID: 28396106.5. Edlich F. BCL-2 proteins and apoptosis: recent insights and unknowns. Biochem Biophys Res Commun. 2018; 500:26–34. PMID: 28676391.6. Faes S, Dormond O. PI3K and AKT: unfaithful partners in cancer. Int J Mol Sci. 2015; 16:21138–21152. PMID: 26404259.7. Lien EC, Dibble CC, Toker A. PI3K signaling in cancer: beyond AKT. Curr Opin Cell Biol. 2017; 45:62–71. PMID: 28343126.8. Mayer IA, Arteaga CL. The PI3K/AKT pathway as a target for cancer treatment. Annu Rev Med. 2016; 67:11–28. PMID: 26473415.9. Badrinath N, Yoo SY. Mitochondria in cancer: in the aspects of tumorigenesis and targeted therapy. Carcinogenesis. 2018; 39:1419–1430. PMID: 30357389.10. Moloney JN, Cotter TG. ROS signalling in the biology of cancer. Semin Cell Dev Biol. 2018; 80:50–64. PMID: 28587975.11. Mi Y, Xiao C, Du Q, Wu W, Qi G, Liu X. Momordin Ic couples apoptosis with autophagy in human hepatoblastoma cancer cells by reactive oxygen species (ROS)-mediated PI3K/Akt and MAPK signaling pathways. Free Radic Biol Med. 2016; 90:230–242. PMID: 26593748.12. Karimian A, Mir SM, Parsian H, Refieyan S, Mirza-Aghazadeh-Attari M, Yousefi B, Majidinia M. Crosstalk between phosphoinositide 3-kinase/Akt signaling pathway with DNA damage response and oxidative stress in cancer. J Cell Biochem. 2019; 120:10248–10272. PMID: 30592328.13. Song X, Wang Z, Liang H, Zhang W, Ye Y, Li H, Hu Y, Zhang Y, Weng H, Lu J, et al. Dioscin induces gallbladder cancer apoptosis by inhibiting ROS-mediated PI3K/AKT signalling. Int J Biol Sci. 2017; 13:782–793. PMID: 28656003.14. Sun X, Duan Z. Research progress of medicinal plants of Zanthoxylum linn. Yao Xue Xue Bao. 1996; 31:231–240. PMID: 9206270.15. Hu L, Wang K, Wang Z, Liu J, Wang K, Zhang J, Luo Z, Xue Y, Zhang Y, Zhang Y. A new megastigmane sesquiterpenoid from Zanthoxylum schinifolium Sieb. et Zucc. Molecules. 2016; 21:383. PMID: 27007360.16. Liu ZL, Chu SS, Jiang GH. Feeding deterrents from Zanthoxylum schinifolium against two stored-product insects. J Agric Food Chem. 2009; 57:10130–10133. PMID: 19886679.17. Tsai IL, Lin WY, Teng CM, Ishikawa T, Doong SL, Huang MW, Chen YC, Chen IS. Coumarins and antiplatelet constituents from the root bark of Zanthoxylum schinifolium . Planta Med. 2000; 66:618–623. PMID: 11105565.18. Cao LH, Lee YJ, Kang DG, Kim JS, Lee HS. Effect of Zanthoxylum schinifolium on TNF-alpha-induced vascular inflammation in human umbilical vein endothelial cells. Vascul Pharmacol. 2009; 50:200–207. PMID: 19563733.19. Choi EO, Park C, Shin SS, Cho EJ, Kim BW, Hwang JA, Hwang HJ, Choi YH. Zanthoxylum schinifolium leaf ethanol extract inhibits adipocyte differentiation through inactivation of the extracellular signal regulated kinase and phosphoinositide 3-kinase/Akt signaling pathways in 3T3-L1 pre-adipocytes. Mol Med Rep. 2015; 12:1314–1320. PMID: 25760758.20. Nguyen PH, Zhao BT, Kim O, Lee JH, Choi JS, Min BS, Woo MH. Anti-inflammatory terpenylated coumarins from the leaves of Zanthoxylum schinifolium with α-glucosidase inhibitory activity. J Nat Med. 2016; 70:276–281. PMID: 26753624.21. Kim KK, Kim TW, Kang YH, Kim DJ, Choe M. Lipid-lowering effects of Zanthoxylum schinifolium Siebold & Zucc. seed oil (ZSO) in hyperlipidemic rats and lipolytic effects in 3T3-L1 adipocytes. Food Sci Biotechnol. 2016; 25:1427–1436. PMID: 30263426.22. Lee HY, Park YM, Lee YH, Kang YG, Lee HM, Park DS, Yang HJ, Kim MJ, Lee YR. Immunostimulatory effect of Zanthoxylum schinifolium-based complex oil prepared by supercritical fluid extraction in splenocytes and cyclophosphamide-induced immunosuppressed rats. Evid Based Complement Alternat Med. 2018; 2018:8107326. PMID: 30402134.23. Lee SW, Lim JM, Mohan H, Seralathan KK, Park YJ, Lee JH, Oh BT. Enhanced bioactivity of Zanthoxylum schinifolium fermented extract: Anti-inflammatory, anti-bacterial, and anti-melanogenic activity. J Biosci Bioeng. 2020; 129:638–645. PMID: 31926815.24. Paik SY, Koh KH, Beak SM, Paek SH, Kim JA. The essential oils from Zanthoxylum schinifolium pericarp induce apoptosis of HepG2 human hepatoma cells through increased production of reactive oxygen species. Biol Pharm Bull. 2005; 28:802–807. PMID: 15863882.25. Jun DY, Kim JS, Park HS, Han CR, Fang Z, Woo MH, Rhee IK, Kim YH. Apoptogenic activity of auraptene of Zanthoxylum schinifolium toward human acute leukemia Jurkat T cells is associated with ER stress-mediated caspase-8 activation that stimulates mitochondria-dependent or -independent caspase cascade. Carcinogenesis. 2007; 28:1303–1313. PMID: 17301064.26. Min BK, Hyun DG, Jeong SY, Kim YH, Ma ES, Woo MH. A new cytotoxic coumarin, 7-[(E)-3′,7′-dimethyl-6′-oxo-2′,7′-octadienyl] oxy coumarin, from the leaves of Zanthoxylum schinifolium . Arch Pharm Res. 2011; 34:723–726. PMID: 21656356.27. Li W, Sun YN, Yan XT, Yang SY, Kim EJ, Kang HK, Kim YH. Coumarins and lignans from Zanthoxylum schinifolium and their anticancer activities. J Agric Food Chem. 2013; 61:10730–10740. PMID: 24144361.28. Choi YH. Trans-cinnamaldehyde protects C2C12 myoblasts from DNA damage, mitochondrial dysfunction and apoptosis caused by oxidative stress through inhibiting ROS production. Genes Genomics. 2021; 43:303–312. PMID: 32851512.29. Pham TNA, Le B, Yang SH. Anticancer activity of the potential Pyropia yezoensis galactan fractionated in human prostate cancer cells. Biotechnol Bioprocess Eng. 2021; 26:63–70.30. Park S, Kim M, Hong Y, Lee H, Tran Q, Kim C, Kwon SH, Park J, Park J, Kim SH. Myristoylated TMEM39AS41, a cell-permeable peptide, causes lung cancer cell death. Toxicol Res. 2020; 36:123–130. PMID: 32257924.31. Liang Y, Kong D, Zhang Y, Li S, Li Y, Ramamoorthy S, Ma J. Fisetin inhibits cell proliferation and induces apoptosis via JAK/STAT3 signaling pathways in human thyroid TPC 1 cancer cells. Biotechnol Bioprocess Eng. 2020; 25:197–205.32. Hwangbo H, Kim SY, Lee H, Park SH, Hong SH, Park C, Kim GY, Leem SH, Hyun JW, Cheong J, et al. Auranofin enhances sulforaphane-mediated apoptosis in hepatocellular carcinoma Hep3B cells through inactivation of the PI3K/Akt signaling pathway. Biomol Ther (Seoul). 2020; 28:443–455. PMID: 32856616.33. Bae CS, Lee CM, Ahn T. Encapsulation of apoptotic proteins in lipid nanoparticles to induce death of cancer cells. Biotechnol Bioprocess Eng. 2020; 25:264–271.34. Yin XM. Bid, a BH3-only multi-functional molecule, is at the cross road of life and death. Gene. 2006; 369:7–19. PMID: 16446060.35. Billen LP, Shamas-Din A, Andrews DW. Bid: a Bax-like BH3 protein. Oncogene. 2008; 27(Suppl 1):S93–104. PMID: 19641510.36. Kantari C, Walczak H. Caspase-8 and Bid: caught in the act between death receptors and mitochondria. Biochim Biophys Acta. 2011; 1813:558–563. PMID: 21295084.37. Houédé N, Pourquier P. Targeting the genetic alterations of the PI3K-AKT-mTOR pathway: its potential use in the treatment of bladder cancers. Pharmacol Ther. 2015; 145:1–18. PMID: 24929024.38. Sathe A, Nawroth R. Targeting the PI3K/AKT/mTOR pathway in bladder cancer. Methods Mol Biol. 2018; 1655:335–350. PMID: 28889395.39. Song G, Ouyang G, Bao S. The activation of Akt/PKB signaling pathway and cell survival. J Cell Mol Med. 2005; 9:59–71. PMID: 15784165.40. Manning BD, Cantley LC. AKT/PKB signaling: navigating downstream. Cell. 2007; 129:1261–1274. PMID: 17604717.41. Galadari S, Rahman A, Pallichankandy S, Thayyullathil F. Reactive oxygen species and cancer paradox: to promote or to suppress? Free Radic Biol Med. 2017; 104:144–164. PMID: 28088622.42. Lee YJ, Yoon JJ, Lee SM, Kim JS, Kang DG, Lee HS. Inhibitory effect of Zanthoxylum schinifolium on vascular smooth muscle proliferation. Immunopharmacol Immunotoxicol. 2012; 34:354–361. PMID: 22268651.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Erratum: Induction of apoptotic cell death in human bladder cancer cells by ethanol extract of Zanthoxylum schinifolium leaf, through ROSdependent inactivation of the PI3K/ Akt signaling pathway
- Induction of Apoptosis Scutellaria baicalensis Georgi Root Extract by Inactivation of the Phosphatidyl Inositol 3-kinase/Akt Signaling Pathway in Human Leukemia U937 Cells
- Induction of Hepatocellular Carcinoma Cell Cycle Arrest and Apoptosis by Dendropanax morbifera Leveille Leaf Extract via the PI3K/AKT/mTOR Pathway
- beta-lapachone-Induced Apoptosis of Human Gastric Carcinoma AGS Cells Is Caspase-Dependent and Regulated by the PI3K/Akt Pathway
- Myricetin Inhibits Angiogenesis by Inducing Apoptosis and Suppressing PI3K/Akt/mTOR Signaling in Endothelial Cells