J Korean Med Sci.
2022 Apr;37(13):e102. 10.3346/jkms.2022.37.e102.
Effectiveness and Safety of Regdanvimab in Patients With Mild-ToModerate COVID-19: A Retrospective Cohort Study
- Affiliations
-
- 1College of Pharmacy, Pusan National University, Busan, Korea
- 2Division of Pulmonology, Department of Internal Medicine, Busan Medical Center, Busan, Korea
- KMID: 2527829
- DOI: http://doi.org/10.3346/jkms.2022.37.e102
Abstract
- Background
Regdanvimab has decreased the time to clinical recovery from coronavirus disease 2019 (COVID-19) and lowered the rate of oxygen therapy according to the results from phase 2/3 randomized controlled trial. More information is needed about the effects and safety of regdanvimab.
Methods
We analyzed data for patients with high-risk mild or moderate COVID-19 being admitted to Busan Medical Center between December 1, 2020 and April 16, 2021. A propensity score (PS) matched analysis was conducted to compare patients treated with and without regdanvimab. The primary outcome was in-hospital death or disease aggravation which means the need for oxygen therapy (low- or high-flow oxygen therapy and mechanical ventilation) and secondary outcomes comprised the length of hospital stay and adverse reactions.
Results
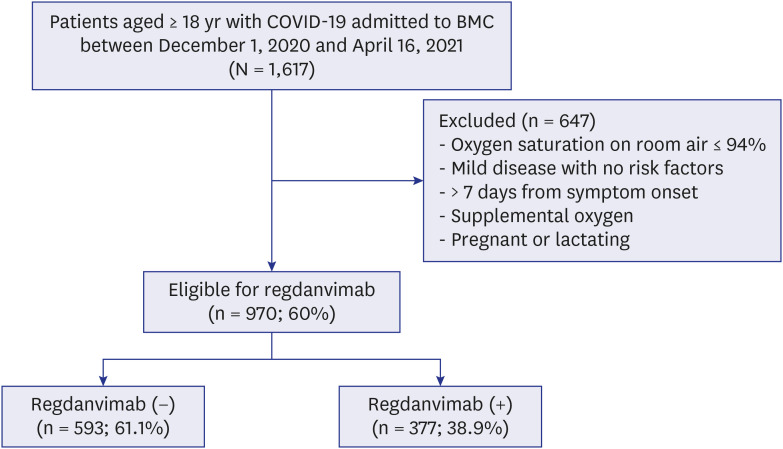
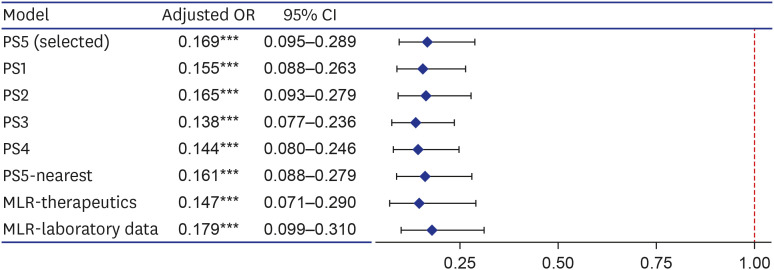
Among 1,617 selected patients, 970 (60.0%) were indicated for regdanvimab. Of these, 377 (38.9%) were administered with regdanvimab. Among a 1:1 PS-matched cohort of 377 patients each treated with and without regdanvimab, 19 (5%) and 81 (21.5%) reached the composite outcome of death, or disease aggravation, respectively (absolute risk difference, −16.4%; 95% confidence interval [CI], −21.1, −11.7; relative risk difference, 76.5%; P < 0.001). Regdanvimab significantly reduced the composite outcome of death, or disease aggravation in univariate (odds ratio [OR], 0.194; 95% CI, 0.112–0.320; P < 0.001) and multivariableadjusted analyses (OR, 0.169; 95% CI, 0.095–0.289; P < 0.001). The hospital stay was shorter for the group with than without regdanvimab. Some hematological adverse reactions were more frequent in the group without regdanvimab, but other adverse reactions did not significantly differ between the groups.
Conclusion
Regdanvimab was associated with a significantly lower risk of disease aggravation without increasing adverse reactions.
Keyword
Figure
Reference
-
1. Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020; 579(7798):270–273. PMID: 32015507.
Article2. World Health Organization. Director-general's remarks at the media briefing on 2019-nCoV on 11 February 2020. Updated 2020. Accessed November 1, 2021. http://www.who.int/dg/speeches/detail/who-director-general-s-remarks-at-the-media-briefing-on-2019-ncov-on-11-february-2020 .3. Our World in Data. Statistics and research coronavirus (COVID-19) vaccinations. Updated 2021. Accessed November 1, 2021. https://ourworldindata.org/covid-vaccinations .4. World Health Organization. COVID-19 weekly epidemiological update. Updated 2021. Accessed November 1, 2021. https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19---26-october-2021 .5. Johns Hopkins Coronavirus Resource Center. COVID-19 dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU). Updated 2021. Accessed November 1, 2021. https://coronavirus.jhu.edu/map.html .6. World Health Organization. Tracking SARS-CoV-2 variants. Updated 2021. Accessed November 1, 2021. https://www.who.int/en/activities/tracking-SARS-CoV-2-variants/ .7. Yi S, Kim JM, Choe YJ, Hong S, Choi S, Ahn SB, et al. SARS-CoV-2 delta variant breakthrough infection and onward secondary transmission in household. J Korean Med Sci. 2022; 37(1):e12. PMID: 34981682.
Article8. Lee JJ, Choe YJ, Jeong H, Kim M, Kim S, Yoo H, et al. Importation and transmission of SARS-CoV-2 B.1.1.529 (Omicron) variant of concern in Korea, November 2021. J Korean Med Sci. 2021; 36(50):e346. PMID: 34962117.
Article9. Jia Z, Gong W. Will mutations in the spike protein of SARS-CoV-2 lead to the failure of COVID-19 vaccines? J Korean Med Sci. 2021; 36(18):e124. PMID: 33975397.
Article10. Ryu BH, Hong SI, Lim SJ, Cho Y, Hwang C, Kang H, et al. Clinical features of adult COVID-19 patients without risk factors before and after the nationwide SARS-CoV-2 B.1.617.2 (Delta)-variant outbreak in Korea: experience from Gyeongsangnam-do. J Korean Med Sci. 2021; 36(49):e341. PMID: 34931500.
Article11. World Health Organization. Therapeutics and COVID-19: living guideline. Updated 2022. Accessed November 1, 2021. https://www.who.int/publications/i/item/therapeutics-and-covid-19-living-guideline .12. National Institutes of Health. COVID-19 treatment guidelines panel. Coronavirus disease 2019 (COVID-19) treatment guidelines. Updated 2021. Accessed November 1, 2021. https://www.covid19treatmentguidelines.nih.gov/ .13. Wang C, Li W, Drabek D, Okba NM, van Haperen R, Osterhaus AD, et al. A human monoclonal antibody blocking SARS-CoV-2 infection. Nat Commun. 2020; 11(1):2251. PMID: 32366817.
Article14. Jahanshahlu L, Rezaei N. Monoclonal antibody as a potential anti-COVID-19. Biomed Pharmacother. 2020; 129:110337. PMID: 32534226.
Article15. Saphire EO, Schendel SL, Gunn BM, Milligan JC, Alter G. Antibody-mediated protection against Ebola virus. Nat Immunol. 2018; 19(11):1169–1178. PMID: 30333617.
Article16. Prabakaran P, Zhu Z, Xiao X, Biragyn A, Dimitrov AS, Broder CC, et al. Potent human monoclonal antibodies against SARS CoV, Nipah and Hendra viruses. Expert Opin Biol Ther. 2009; 9(3):355–368. PMID: 19216624.
Article17. Kim C, Ryu DK, Lee J, Kim YI, Seo JM, Kim YG, et al. A therapeutic neutralizing antibody targeting receptor binding domain of SARS-CoV-2 spike protein. Nat Commun. 2021; 12(1):288. PMID: 33436577.
Article18. Eom JS, Ison M, Streinu-Cercel A, Săndulescu O, Preotescu LL, Kim YS, et al. Efficacy and safety of CT-P59 plus standard of care: a phase 2/3 randomized, double-blind, placebo-controlled trial in outpatients with mild-to-moderate SARS-CoV-2 infection. Res Sq. DOI: 10.21203/rs.3.rs-296518/v1.
Article19. Ministry of Food and Drug Safety. Approval of Regkirona® Inj., an antibody treatment for COVID-19. Updated 2021. Accessed November 1, 2021. https://www.mfds.go.kr/brd/m_99/view.do?seq=45029&srchFr=&srchTo=&srchWord=&srchTp=0&itm_seq_1=0&itm_seq_2=0&multi_itm_seq=0&company_cd=&company_nm=&Data_stts_gubun=C1009&page=2 .20. Ministry of Food and Drug Safety. Pharmaceutical product information: Regkirona® Inj. 960 mg (regdanvimab). Updated 2021. Accessed November 1, 2021. https://nedrug.mfds.go.kr/pbp/CCBBB01/getItemDetail?itemSeq=202101124 .21. European Medicines Agency. EMA issues advice on use of regdanvimab for treating COVID-19. Updated 2021. Accessed November 1, 2021. https://www.ema.europa.eu/en/news/ema-issues-advice-use-regdanvimab-treating-covid-19 .22. Ministry of Food and Drug Safety. Official approval of Regkirona® Inj., an antibody treatment for COVID-19. Updated 2021. Accessed November 1, 2021. https://www.mfds.go.kr/brd/m_99/view.do?seq=45778&srchFr=&srchTo=&srchWord=&srchTp=0&itm_seq_1=0&itm_seq_2=0&multi_itm_seq=0&company_cd=&company_nm=&Data_stts_gubun=C1009&page=1 .23. Kim SB, Ryoo S, Huh K, Joo EJ, Kim YJ, Choi WS, et al. Revised Korean Society of Infectious Diseases/National Evidence-based Healthcarea collaborating agency guidelines on the treatment of patients with COVID-19. Infect Chemother. 2021; 53(1):166–219. PMID: 34409790.
Article24. Lam KW, Chow KW, Vo J, Hou W, Li H, Richman PS, et al. Continued in-hospital angiotensin-converting enzyme inhibitor and angiotensin ii receptor blocker use in hypertensive COVID-19 patients is associated with positive clinical outcome. J Infect Dis. 2020; 222(8):1256–1264. PMID: 32702098.
Article25. Fosbøl EL, Butt JH, Østergaard L, Andersson C, Selmer C, Kragholm K, et al. Association of angiotensin-converting enzyme inhibitor or angiotensin receptor blocker use with COVID-19 diagnosis and mortality. JAMA. 2020; 324(2):168–177. PMID: 32558877.
Article26. Gupta A, Madhavan MV, Poterucha TJ, DeFilippis EM, Hennessey JA, Redfors B, et al. Association between antecedent statin use and decreased mortality in hospitalized patients with COVID-19. Nat Commun. 2021; 12(1):1325. PMID: 33637713.
Article27. Daniels LB, Sitapati AM, Zhang J, Zou J, Bui QM, Ren J, et al. Relation of statin use prior to admission to severity and recovery among COVID-19 inpatients. Am J Cardiol. 2020; 136:149–155. PMID: 32946859.
Article28. Osborne TF, Veigulis ZP, Arreola DM, Mahajan SM, Röösli E, Curtin CM. Association of mortality and aspirin prescription for COVID-19 patients at the Veterans Health Administration. PLoS One. 2021; 16(2):e0246825. PMID: 33571280.
Article29. Chow JH, Khanna AK, Kethireddy S, Yamane D, Levine A, Jackson AM, et al. Aspirin use is associated with decreased mechanical ventilation, intensive care unit admission, and in-hospital mortality in hospitalized patients with coronavirus disease 2019. Anesth Analg. 2021; 132(4):930–941. PMID: 33093359.
Article30. Li X, Xu S, Yu M, Wang K, Tao Y, Zhou Y, et al. Risk factors for severity and mortality in adult COVID-19 inpatients in Wuhan. J Allergy Clin Immunol. 2020; 146(1):110–118. PMID: 32294485.
Article31. Pastor-Nieto M, Checa-Díaz P, González-Muñoz P, Martín-Fuentes A, Vergara-Sánchez A, Sánchez-Herreros C, et al. Prior treatment with immunosuppressants among COVID-19 inpatients at one hospital in Spain. J Eur Acad Dermatol Venereol. 2020; 34(12):e760–e762. PMID: 32594619.32. Scully EP, Haverfield J, Ursin RL, Tannenbaum C, Klein SL. Considering how biological sex impacts immune responses and COVID-19 outcomes. Nat Rev Immunol. 2020; 20(7):442–447. PMID: 32528136.
Article33. Gebhard C, Regitz-Zagrosek V, Neuhauser HK, Morgan R, Klein SL. Impact of sex and gender on COVID-19 outcomes in Europe. Biol Sex Differ. 2020; 11(1):29. PMID: 32450906.
Article34. Moon Hj, Kim K, Kang EK, Yang HJ, Lee E. Prediction of COVID-19-related mortality and 30-day and 60-day survival probabilities using a nomogram. J Korean Med Sci. 2021; 36(35):e248. PMID: 34490756.
Article35. Clinical Trials Arena. Celltrion's COVID-19 drug regdanvimab meets Phase III endpoints. Updated 2021. Accessed November 1, 2021. https://www.clinicaltrialsarena.com/news/celltrion-regdanvimab-phaseiii-data/ .36. Monoclonal regdanvimab cuts COVID progression risk in placebo trial. IDWeek, September 29-October 3, 2021. Updated 2021. Accessed November 1, 2021. https://www.natap.org/2021/IDWeek/IDWeek_43.htm .37. An EUA for bamlanivimab and etesevimab for COVID-19. Med Lett Drugs Ther. 2021; 63(1621):49–50. PMID: 33830966.38. FDA. Fact sheet for health care providers. Emergency Use Authorization (EUA) of bamlanivimab and etesevimab. Updated 2021. Accessed November 1, 2021. https://bit.ly/3qfS6DN .39. Weinreich DM, Sivapalasingam S, Norton T, Ali S, Gao H, Bhore R, et al. REGN-COV2, a neutralizing antibody cocktail, in outpatients with COVID-19. N Engl J Med. 2021; 384(3):238–251. PMID: 33332778.
Article40. An EUA for casirivimab and imdevimab for COVID-19. Med Lett Drugs Ther. 2020; 62(1614):201–202. PMID: 33451174.41. FDA. Fact sheet for health care providers. Emergency Use Authorization (EUA) of casirivimab and imdevimab. Updated 2021. Accessed November 1, 2021. http://e-lactancia.org/media/papers/casirivimabimdevimab-DS-FDA-Regeneron2020.pdf .42. Orders M. An EUA for sotrovimab for treatment of COVID-19. Med Lett Drugs Ther. 2021; 63(1627):97–xx98. PMID: 34181630.43. FDA. Fact sheet for health care providers. Emergency Use Authorization (EUA) of sotrovimab. Updated 2021. Accessed November 1, 2021. https://www.fda.gov/media/149534/download .44. Hurt AC, Wheatley AK. Neutralizing antibody therapeutics for COVID-19. Viruses. 2021; 13(4):628. PMID: 33916927.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Real World Experience with Regdanvimab Treatment of Mild-toModerate Coronavirus Disease-19 in a COVID-19 Designated Hospital of Korea
- Effectiveness of Regdanvimab at Preventing the Need for Oxygen Therapy in Patients with Mild-to-Moderate COVID-19: A Retrospective Cohort Study
- Effectiveness of regdanvimab on mortality in COVID-19 infected patients on hemodialysis
- Use of the Monoclonal Antibody Regdanvimab to Treat Patients Hospitalized with COVID-19: Real-World Data during the Delta Variant Predominance
- Early Oxygen Requirement in Patients with Mild-to-Moderate COVID-19 Who Received Regdanvimab after DeltaVariant Outbreak