Neonatal Med.
2022 Feb;29(1):36-45. 10.5385/nm.2022.29.1.36.
Neurodevelopmental Outcomes of Very-Low-Birth-Weight Infants without Severe Brain Lesions and Impact of Postnatal Steroid Use: A Single-Center Korean Study
- Affiliations
-
- 1Department of Pediatrics, Pusan National University Children’s Hospital, Yangsan, Korea
- 2Department of Pediatrics, Pusan National University Hospital, Busan, Korea
- 3Department of Pediatrics, Pusan National University School of Medicine, Yangsan, Korea
- 4Department of Statistics, Pusan National University, Busan, Korea
- KMID: 2527259
- DOI: http://doi.org/10.5385/nm.2022.29.1.36
Abstract
- Purpose
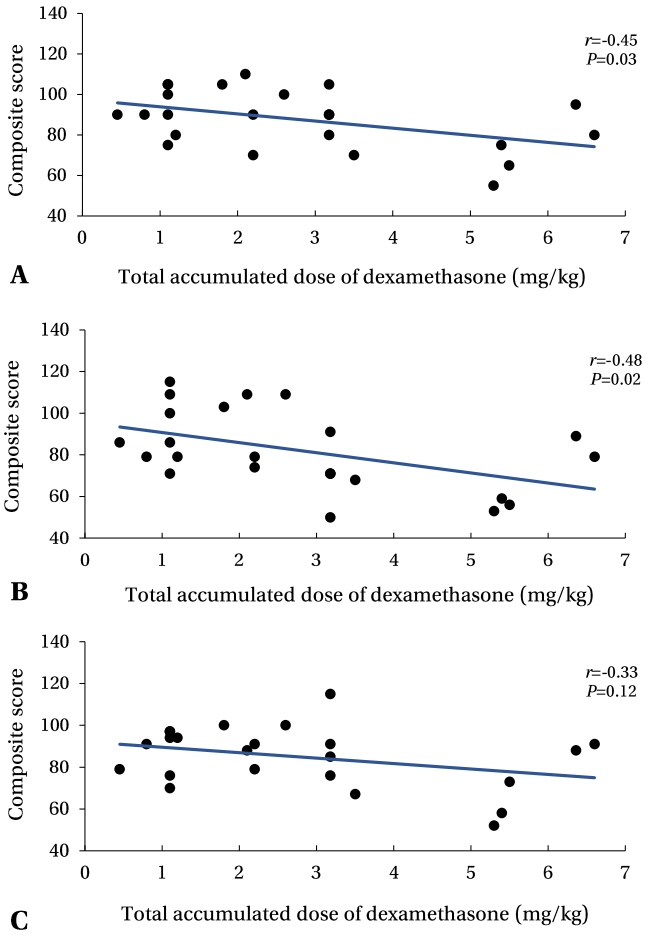
We used the Bayley Scales of Infant and Toddler Development (BSID)-III to analyze the incidence and risk factors of developmental delay in very-low-birth-weight infants without severe brain lesions. We further examined the correlation between the cumulative dexamethasone dose and developmental assessment results.
Methods
We retrospectively analyzed data of preterm infants (birth weight <1,500 g) admitted to our neonatal intensive care unit between January 2014 to December 2020. The BSID-III scores obtained between the corrected ages of 12 and 24 months and after 24 months were analyzed. Developmental delay was defined as a composite score of <85 for the cognition, language, and motor domains. Univariate and multivariate analyses of developmental delay risk factors and developmental changes from the first to second BSID-III were performed. Correlations between the accumulated dexamethasone dose used for bronchopulmonary dysplasia (BPD) and the first and second test scores were analyzed.
Results
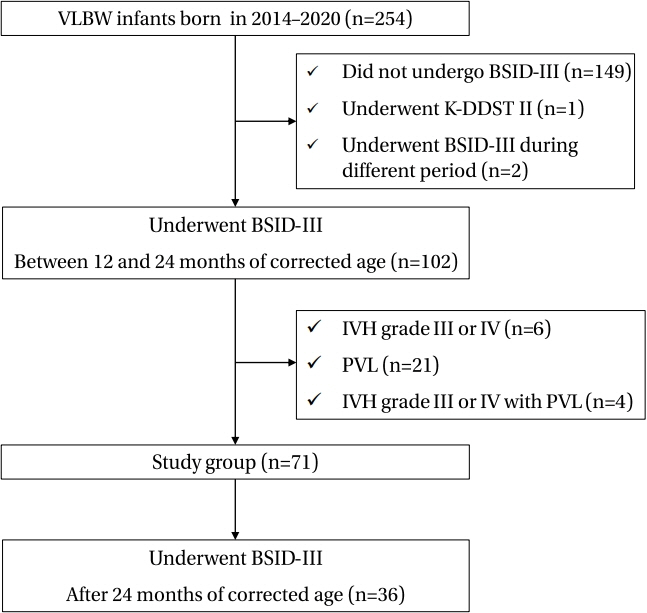
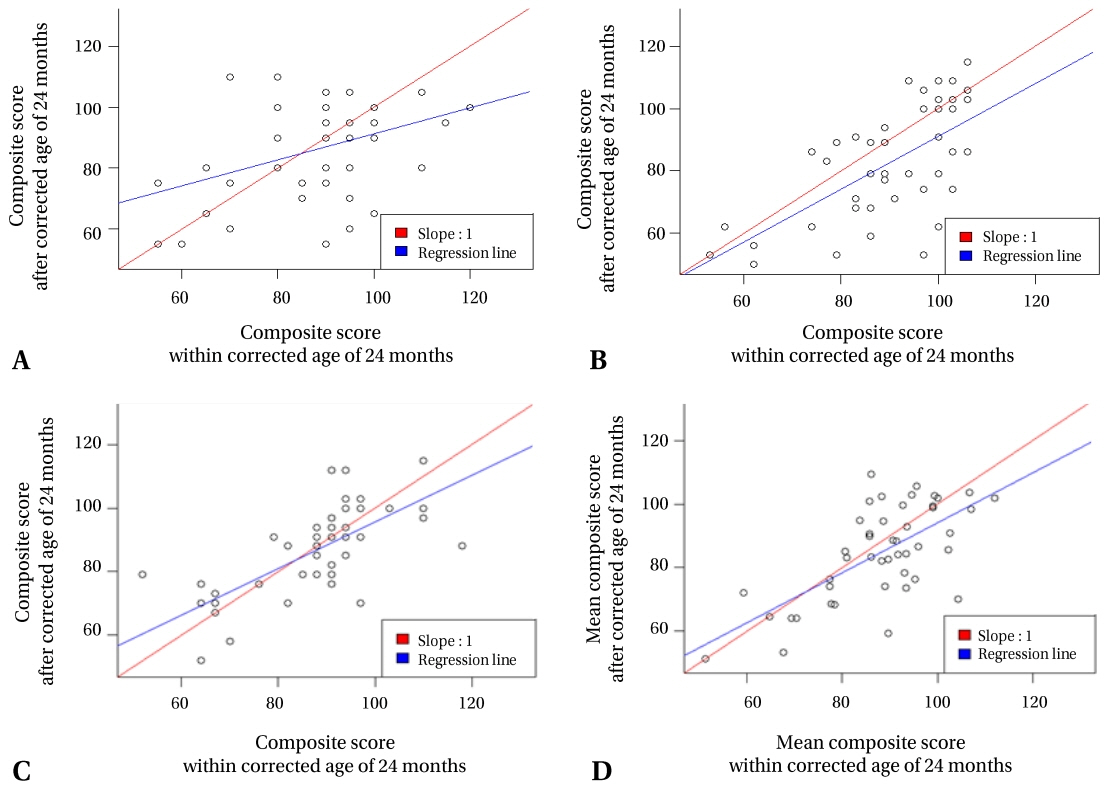
Seventy-one and thirty-six infants completed the first and second tests, respectively. In both tests, developmental delay was most commonly observed in the language domain (26.8%, 47.2%). In multivariate analysis, mild BPD was identified as a developmental delay risk factor (P<0.05), whereas prenatal steroid use reduced the developmental delay risk (P<0.05). All domain scores were lower in the second test than in the first test. The cognition and language domain scores in the second test decreased with increasing cumulative dexamethasone doses.
Conclusion
Very-low-birth-weight infants typically experience language delay, which can persist as they age.
Figure
Reference
-
1. Kim JK, Chang YS, Sung S, Ahn SY, Yoo HS, Park WS. Trends in survival and incidence of bronchopulmonary dysplasia in extremely preterm infants at 23-26 weeks gestation. J Korean Med Sci. 2016; 31:423–9.2. Stoll BJ, Hansen NI, Bell EF, Walsh MC, Carlo WA, Shankaran S, et al. Trends in care practices, morbidity, and mortality of ex tremely preterm neonates, 1993-2012. JAMA. 2015; 314:1039–51.3. Woodward LJ, Anderson PJ, Austin NC, Howard K, Inder TE. Neonatal MRI to predict neurodevelopmental outcomes in preterm infants. N Engl J Med. 2006; 355:685–94.4. Jeng SF, Hsu CH, Tsao PN, Chou HC, Lee WT, Kao HA, et al. Bronchopulmonary dysplasia predicts adverse developmental and clinical outcomes in very-low-birthweight infants. Dev Med Child Neurol. 2008; 50:51–7.5. Grier DG, Halliday HL. Corticosteroids in the prevention and management of bronchopulmonary dysplasia. Semin Neonatol. 2003; 8:83–91.6. Sharp M, DeMauro SB. Counterbalanced comparison of the BSID-II and Bayley-III at eighteen to twenty-two months corrected age. J Dev Behav Pediatr. 2017; 38:322–9.7. Johnson S, Moore T, Marlow N. Using the Bayley-III to assess neurodevelopmental delay: which cut-off should be used? Pediatr Res. 2014; 75:670–4.8. Ehrenkranz RA, Dusick AM, Vohr BR, Wright LL, Wrage LA, Poole WK. Growth in the neonatal intensive care unit influences neurodevelopmental and growth outcomes of extremely low birth weight infants. Pediatrics. 2006; 117:1253–61.9. Jobe AH, Bancalari E. Bronchopulmonary dysplasia. Am J Respir Crit Care Med. 2001; 163:1723–9.10. Ahn SH, Kim SA. Assessment of preterm infants using the Bayley-III scales in Korea. Ann Rehabil Med. 2017; 41:843–50.11. Guerra CC, Barros MC, Goulart AL, Fernandes LV, Kopelman BI, Santos AM. Premature infants with birth weights of 1500-1999 g exhibit considerable delays in several developmental areas. Acta Paediatr. 2014; 103:e1–6.12. Chang YS, Ahn SY, Park WS. The establishment of the Korean Neonatal Network (KNN). Neonatal Med. 2013; 20:169–78.13. The Executive Committee of Korean Neonatal Network. 2016 Korean Neonatal Network Annual Report. Cheongju: Korean Centers for Disease Control and Prevention;2017. p. 4–63.14. The Executive Committee of Korean Neonatal Network. 2020 Korean Neonatal Network Annual Report. Cheongju: Korean Centers for Disease Control and Prevention;2011. p. 4–68.15. The Executive Committee of Korean Neonatal Network. 2017 Korean Neonatal Network Annual Report. Cheongju: Korean Centers for Disease Control and Prevention;2018. p. 4–65.16. The Executive Committee of Korean Neonatal Network. 2018 Korean Neonatal Network Annual Report. Cheongju: Korean Centers for Disease Control and Prevention;2021. p. 4–68.17. The Executive Committee of Korean Neonatal Network. 2019 Korean Neonatal Network Annual Report. Cheongju: Korean Centers for Disease Control and Prevention;2021. p. 4–68.18. Cha JH, Choi N, Kim YJ, Lee HJ, Kim CR, Park HK. Developmental outcome of very-low-birth-weight infants without major brain injuries based on data from the Korean Neonatal Network: a nationwide cohort study. Neonatal Med. 2020; 27:151–8.19. Sansavini A, Guarini A, Justice LM, Savini S, Broccoli S, Alessandroni R, et al. Does preterm birth increase a child's risk for language impairment? Early Hum Dev. 2010; 86:765–72.20. Kiechl-Kohlendorfer U, Ralser E, Pupp Peglow U, Reiter G, Trawoger R. Adverse neurodevelopmental outcome in preterm infants: risk factor profiles for different gestational ages. Acta Paediatr. 2009; 98:792–6.21. Kerstjens JM, Bocca-Tjeertes IF, de Winter AF, Reijneveld SA, Bos AF. Neonatal morbidities and developmental delay in moderately preterm-born children. Pediatrics. 2012; 130:e265–72.22. Twilhaar ES, Wade RM, de Kieviet JF, van Goudoever JB, van Elburg RM, Oosterlaan J. Cognitive outcomes of children born extremely or very preterm since the 1990s and associated risk factors: a meta-analysis and meta-regression. JAMA Pediatr. 2018; 172:361–7.23. Gentle SJ, Carlo WA, Tan S, Gargano M, Ambalavanan N, Chawla S, et al. Association of antenatal corticosteroids and magnesium sulfate therapy with neurodevelopmental outcome in extremely preterm children. Obstet Gynecol. 2020; 135:1377–86.24. Srinivas Jois R. Neurodevelopmental outcome of late-preterm infants: a pragmatic review. Aust J Gen Pract. 2018; 47:776–81.25. Malavolti AM, Bassler D, Arlettaz-Mieth R, Faldella G, Latal B, Natalucci G. Bronchopulmonary dysplasia-impact of severity and timing of diagnosis on neurodevelopment of preterm infants: a retrospective cohort study. BMJ Paediatr Open. 2018; 2:e000165.26. Wood NS, Costeloe K, Gibson AT, Hennessy EM, Marlow N, Wilkinson AR, et al. The EPICure study: associations and antecedents of neurological and developmental disability at 30 months of age following extremely preterm birth. Arch Dis Child Fetal Neonatal Ed. 2005; 90:F134–40.27. Wilson-Costello D, Walsh MC, Langer JC, Guillet R, Laptook AR, Stoll BJ, et al. Impact of postnatal corticosteroid use on neurodevelopment at 18 to 22 months' adjusted age: effects of dose, timing, and risk of bronchopulmonary dysplasia in extremely low birth weight infants. Pediatrics. 2009; 123:e430–7.28. Davidovitch M, Kuint J, Lerner-Geva L, Zaslavsky-Paltiel I, Rotem RS, Chodick G, et al. Postnatal steroid therapy is associated with autism spectrum disorder in children and adolescents of very low birth weight infants. Pediatr Res. 2020; 87:1045–51.29. Doyle LW, Halliday HL, Ehrenkranz RA, Davis PG, Sinclair JC. An update on the impact of postnatal systemic corticosteroids on mortality and cerebral palsy in preterm infants: effect modification by risk of bronchopulmonary dysplasia. J Pediatr. 2014; 165:1258–60.30. Doyle LW, Cheong JL, Ehrenkranz RA, Halliday HL. Late (> 7 days) systemic postnatal corticosteroids for prevention of bronchopulmonary dysplasia in preterm infants. Cochrane Database Syst Rev. 2017; 10:CD001145.31. Wood NS, Marlow N, Costeloe K, Gibson AT, Wilkinson AR. Neurologic and developmental disability after extremely preterm birth. EPICure Study Group. N Engl J Med. 2000; 343:378–84.32. Maalouf EF, Duggan PJ, Rutherford MA, Counsell SJ, Fletcher AM, Battin M, et al. Magnetic resonance imaging of the brain in a cohort of extremely preterm infants. J Pediatr. 1999; 135:351–7.33. Volpe JJ. Cerebral white matter injury of the premature infantmore common than you think. Pediatrics. 2003; 112(1 Pt 1):176–80.34. de Bruine FT, van den Berg-Huysmans AA, Leijser LM, Rijken M, Steggerda SJ, van der Grond J, et al. Clinical implications of MR imaging findings in the white matter in very preterm infants: a 2-year follow-up study. Radiology. 2011; 261:899–906.35. Brostrom L, Bolk J, Padilla N, Skiold B, Eklof E, Martensson G, et al. Clinical implications of diffuse excessive high signal intensity (DEHSI) on neonatal MRI in school age children born extremely preterm. PLoS One. 2016; 11:e0149578.36. Murner-Lavanchy IM, Kidokoro H, Thompson DK, Doyle LW, Cheong J, Hunt RW, et al. Thirteen-year outcomes in very preterm children associated with diffuse excessive high signal intensity on neonatal magnetic resonance imaging. J Pediatr. 2019; 206:66–71.37. Rath CP, Desai S, Rao SC, Patole S. Diffuse excessive high signal intensity on term equivalent MRI does not predict disability: a systematic review and meta-analysis. Arch Dis Child Fetal Neonatal Ed. 2021; 106:9–16.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Neurodevelopmental outcomes of preterm infants
- Growth patterns of preterm infants in Korea
- Clinical Evaluation of Postnatal Weight Gain in Premature and Low Birth Weight Infants
- Neurodevelopmental Outcomes of Extremely Preterm Infants
- Neurodevelopmental outcomes of very low birth weight infants and extremely low birth weight infants in Korea, 1984-2008