Endocrinol Metab.
2021 Dec;36(6):1298-1306. 10.3803/EnM.2021.1226.
Clinical Value of Serum Mitochondria-Inhibiting Substances in Assessing Renal Hazards: A Community-Based Prospective Study in Korea
- Affiliations
-
- 1Department of Internal Medicine, Kangwon National University School of Medicine, Chuncheon, Korea
- 2Department of Internal Medicine, Nowon Eulji Medical Center, Eulji University, Uijeongbu, Korea
- 3Department of Physiology, College of Medicine, Kyung Hee University, Seoul, Korea
- 4Department of Internal Medicine, Uijeongbu Eulji Medical Center, Eulji University, Uijeongbu, Korea
- KMID: 2523498
- DOI: http://doi.org/10.3803/EnM.2021.1226
Abstract
- Background
Mitochondrial dysfunction is strongly associated with several kidney diseases. However, no studies have evaluated the potential renal hazards of serum mitochondria-inhibiting substance (MIS) and aryl hydrocarbon receptor ligand (AhRL) levels.
Methods
We used serum level of MIS and AhRL and clinical renal outcomes from 1,511 participants of a prospective community-based cohort in Ansung. MIS was evaluated based on intracellular adenosine triphosphate (MIS-ATP) or reactive oxygen species (MIS-ROS) generation measured using cell-based assays.
Results
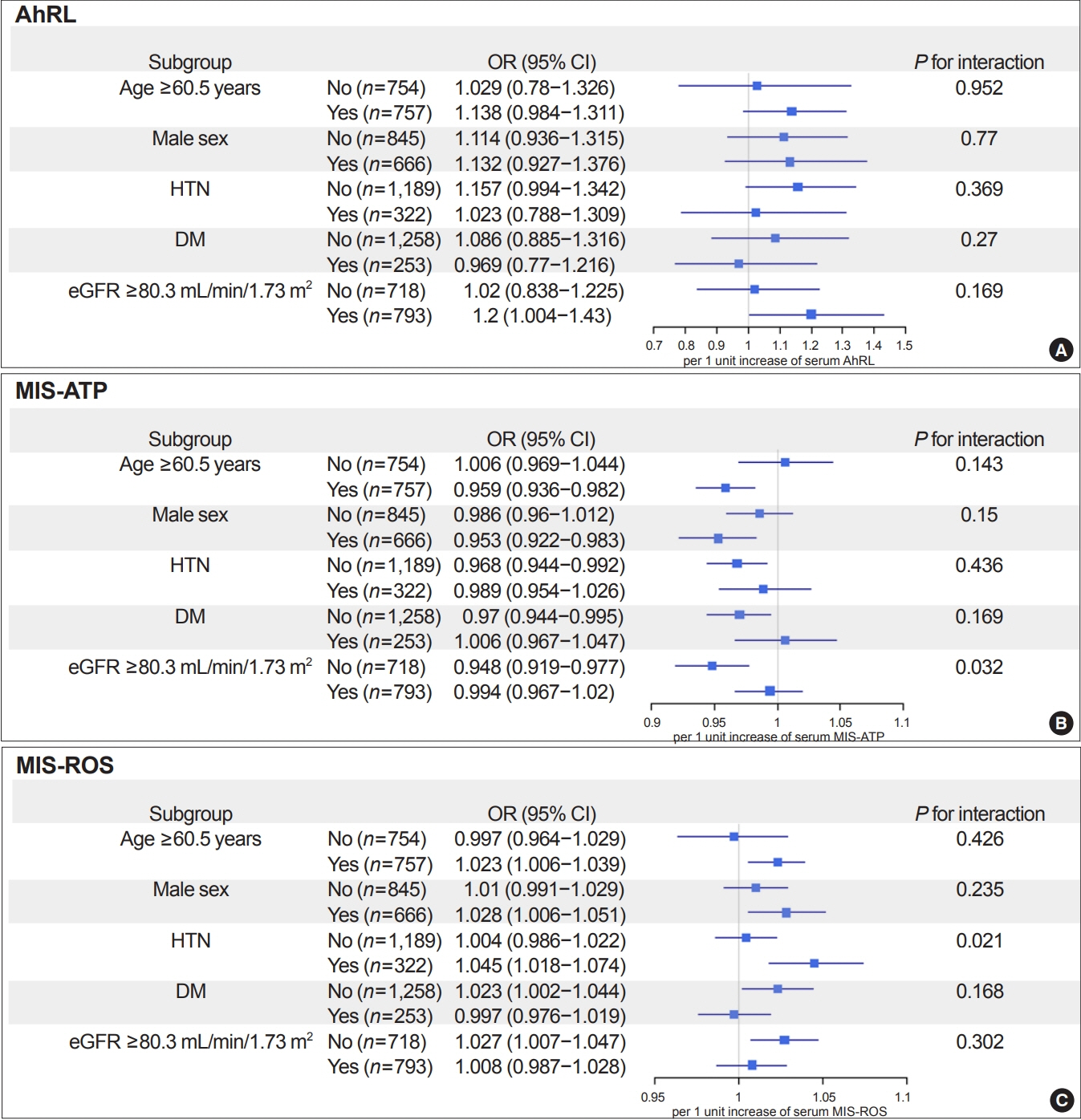
During a mean 6.9-year follow-up, 84 participants (5.6%) developed a rapid decline in kidney function. In the lowest quartile group of MIS-ATP, patients were older and had metabolically deleterious parameters. In multivariate logistic regression analysis, higher MIS-ATP was associated with decreased odds for rapid decline: the odds ratio (OR) of 1% increase was 0.977 (95% confidence interval [CI], 0.957 to 0.998; P=0.031), while higher MIS-ROS was marginally associated with increased odds for rapid decline (OR, 1.014; 95% CI, 0.999 to 1.028; P=0.055). However, serum AhRL was not associated with the rapid decline in kidney function. In subgroup analysis, the renal hazard of MIS was particularly evident in people with hypertension and low baseline kidney function.
Conclusion
Serum MIS was independently associated with a rapid decline in kidney function, while serum AhRL was not. The clinical implication of renal hazard on serum MIS requires further evaluation in future studies.
Keyword
Figure
Reference
-
1. Lee HK. Mitochondrial dysfunction and insulin resistance: the contribution of dioxin-like substances. Diabetes Metab J. 2011; 35:207–15.
Article2. Ray PD, Huang BW, Tsuji Y. Reactive oxygen species (ROS) homeostasis and redox regulation in cellular signaling. Cell Signal. 2012; 24:981–90.
Article3. Lee HK, Cho YM, Kwak SH, Lim S, Park KS, Shim EB. Mitochondrial dysfunction and metabolic syndrome-looking for environmental factors. Biochim Biophys Acta. 2010; 1800:282–9.
Article4. Lowell BB, Shulman GI. Mitochondrial dysfunction and type 2 diabetes. Science. 2005; 307:384–7.
Article5. Bournat JC, Brown CW. Mitochondrial dysfunction in obesity. Curr Opin Endocrinol Diabetes Obes. 2010; 17:446–52.
Article6. Bhargava P, Schnellmann RG. Mitochondrial energetics in the kidney. Nat Rev Nephrol. 2017; 13:629–46.
Article7. Che R, Yuan Y, Huang S, Zhang A. Mitochondrial dysfunction in the pathophysiology of renal diseases. Am J Physiol Renal Physiol. 2014; 306:F367–78.
Article8. Granata S, Dalla Gassa A, Bellin G, Lupo A, Zaza G. Transcriptomics: a step behind the comprehension of the polygenic influence on oxidative stress, immune deregulation, and mitochondrial dysfunction in chronic kidney disease. Biomed Res Int. 2016; 2016:9290857.
Article9. Sharma K, Karl B, Mathew AV, Gangoiti JA, Wassel CL, Saito R, et al. Metabolomics reveals signature of mitochondrial dysfunction in diabetic kidney disease. J Am Soc Nephrol. 2013; 24:1901–12.
Article10. Gamboa JL, Billings FT 4th, Bojanowski MT, Gilliam LA, Yu C, Roshanravan B, et al. Mitochondrial dysfunction and oxidative stress in patients with chronic kidney disease. Physiol Rep. 2016; 4:e12780.
Article11. Granata S, Zaza G, Simone S, Villani G, Latorre D, Pontrelli P, et al. Mitochondrial dysregulation and oxidative stress in patients with chronic kidney disease. BMC Genomics. 2009; 10:388.
Article12. Zaza G, Granata S, Masola V, Rugiu C, Fantin F, Gesualdo L, et al. Downregulation of nuclear-encoded genes of oxidative metabolism in dialyzed chronic kidney disease patients. PLoS One. 2013; 8:e77847.
Article13. Eirin A, Saad A, Tang H, Herrmann SM, Woollard JR, Lerman A, et al. Urinary mitochondrial DNA copy number identifies chronic renal injury in hypertensive patients. Hypertension. 2016; 68:401–10.
Article14. Mutsaers HA, Wilmer MJ, Reijnders D, Jansen J, van den Broek PH, Forkink M, et al. Uremic toxins inhibit renal metabolic capacity through interference with glucuronidation and mitochondrial respiration. Biochim Biophys Acta. 2013; 1832:142–50.
Article15. Nadal A, Quesada I, Tuduri E, Nogueiras R, Alonso-Magdalena P. Endocrine-disrupting chemicals and the regulation of energy balance. Nat Rev Endocrinol. 2017; 13:536–46.
Article16. Kim JT, Lee HK. Metabolic syndrome and the environmental pollutants from mitochondrial perspectives. Rev Endocr Metab Disord. 2014; 15:253–62.
Article17. Park WH, Kang S, Lee HK, Salihovic S, Bavel BV, Lind PM, et al. Relationships between serum-induced AhR bioactivity or mitochondrial inhibition and circulating polychlorinated biphenyls (PCBs). Sci Rep. 2017; 7:9383.
Article18. Lee HK, Park WH, Kang YC, Kang S, Im S, Park S, et al. Serum biomarkers from cell-based assays for AhRL and MIS strongly predicted the future development of diabetes in a large community-based prospective study in Korea. Sci Rep. 2020; 10:6339.
Article19. Kim JT, Kim SS, Jun DW, Hwang YH, Park WH, Pak YK, et al. Serum arylhydrocarbon receptor transactivating activity is elevated in type 2 diabetic patients with diabetic nephropathy. J Diabetes Investig. 2013; 4:483–91.
Article20. Kim Y, Han BG. KoGES group. Cohort profile: the Korean Genome and Epidemiology Study (KoGES) Consortium. Int J Epidemiol. 2017; 46:e20.
Article21. Park WH, Jun DW, Kim JT, Jeong JH, Park H, Chang YS, et al. Novel cell-based assay reveals associations of circulating serum AhR-ligands with metabolic syndrome and mitochondrial dysfunction. Biofactors. 2013; 39:494–504.
Article22. Vondracek J, Pencikova K, Neca J, Ciganek M, Grycova A, Dvorak Z, et al. Assessment of the aryl hydrocarbon receptor-mediated activities of polycyclic aromatic hydrocarbons in a human cell-based reporter gene assay. Environ Pollut. 2017; 220(Pt A):307–16.
Article23. Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009; 150:604–12.
Article24. National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am J Kidney Dis. 2002; 39(2 Suppl 1):S1–266.25. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985; 28:412–9.
Article26. Kolachalama VB, Shashar M, Alousi F, Shivanna S, Rijal K, Belghasem ME, et al. Uremic solute-aryl hydrocarbon receptor-tissue factor axis associates with thrombosis after vascular injury in humans. J Am Soc Nephrol. 2018; 29:1063–72.
Article27. Shinde R, McGaha TL. The aryl hydrocarbon receptor: connecting immunity to the microenvironment. Trends Immunol. 2018; 39:1005–20.
Article28. Brinkmann V, Ale-Agha N, Haendeler J, Ventura N. The aryl hydrocarbon receptor (AhR) in the aging process: another puzzling role for this highly conserved transcription factor. Front Physiol. 2020; 10:1561.
Article29. Stanifer JW, Stapleton HM, Souma T, Wittmer A, Zhao X, Boulware LE. Perfluorinated chemicals as emerging environmental threats to kidney health: a scoping review. Clin J Am Soc Nephrol. 2018; 13:1479–92.30. Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl. 2013; 3:1–150.31. Avilla MN, Malecki KM, Hahn ME, Wilson RH, Bradfield CA. The Ah receptor: adaptive metabolism, ligand diversity, and the Xenokine Model. Chem Res Toxicol. 2020; 33:860–79.
Article32. Kataria A, Trasande L, Trachtman H. The effects of environmental chemicals on renal function. Nat Rev Nephrol. 2015; 11:610–25.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Existing test data for the Act on Registration & Evaluation, etc. of Chemical Substances
- Analysis of the utilization of existing test data for phase-in substance registration under the Act on the Registration and Evaluation, etc. of Chemical Substances
- Occupational health hazards in a prosthodontic practice: review of risk factors and management strategies
- Developing a short standard questionnaire for assessing work organization hazards: the Healthy Work Survey (HWS)
- Respiratory and Other Hazard Characteristics of Substances in Cleaning Products Used in Healthcare Centres in England and Wales