Neurointervention.
2021 Nov;16(3):285-292. 10.5469/neuroint.2021.00290.
Treatment of In-Stent Stenosis Following Flow Diversion of Intracranial Aneurysms with Cilostazol and Clopidogrel
- Affiliations
-
- 1Department of Neurosurgery, MedStar Georgetown University Hospital, Washington, DC, USA
- 2Georgetown University School of Medicine, Washington, DC, USA
- 3Division of Neurosurgery, Walter Reed National Military Medical Center, Bethesda, MD, USA
- 4Department of Neurosurgery, MedStar Washington Hospital Center, Washington, DC, USA
- 5Department of Critical Care Medicine, MedStar Washington Hospital Center, Washington, DC, USA
- 6Department of Radiology, MedStar Washington Hospital Center, Washington, DC, USA
- KMID: 2522044
- DOI: http://doi.org/10.5469/neuroint.2021.00290
Abstract
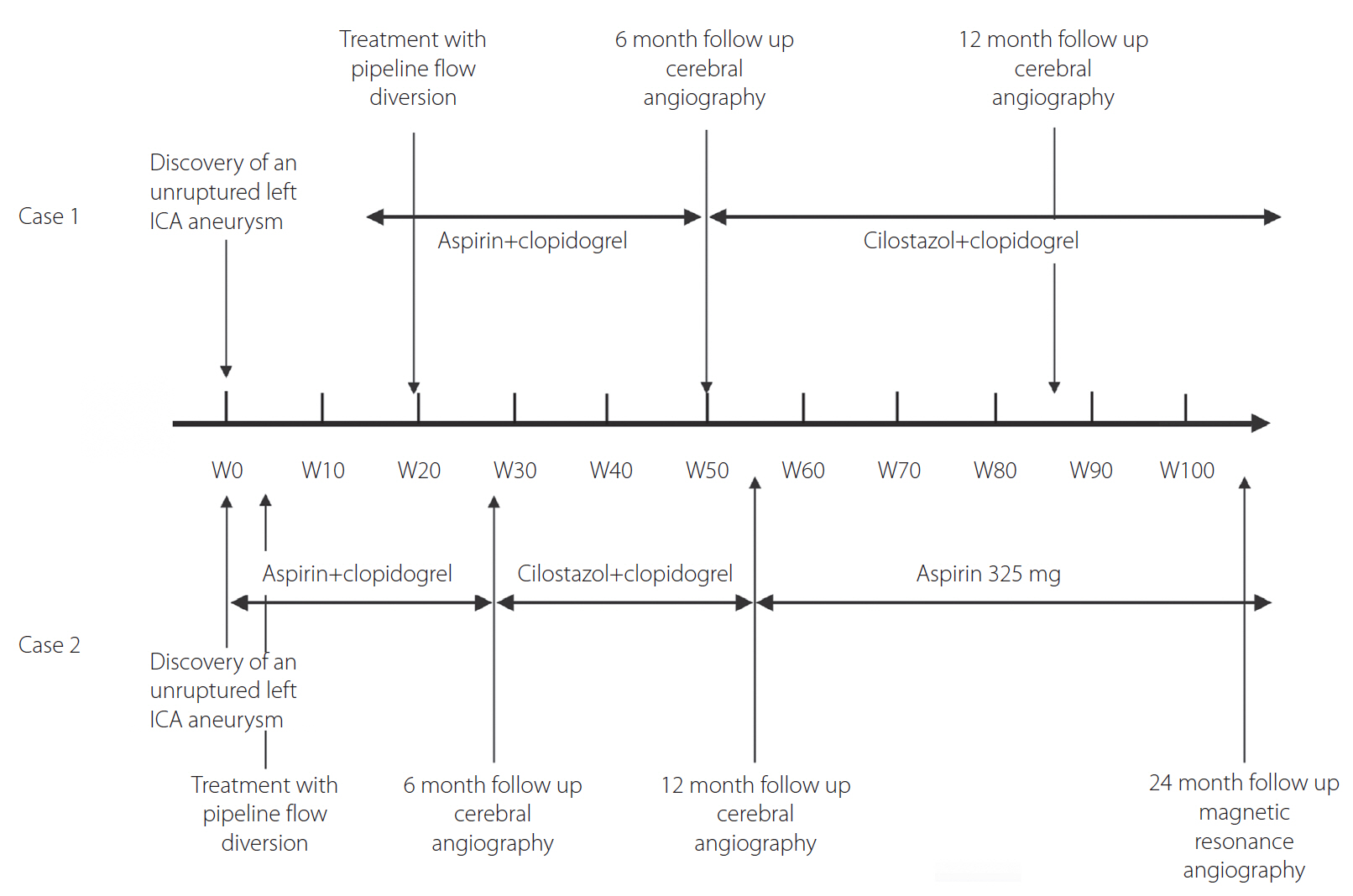
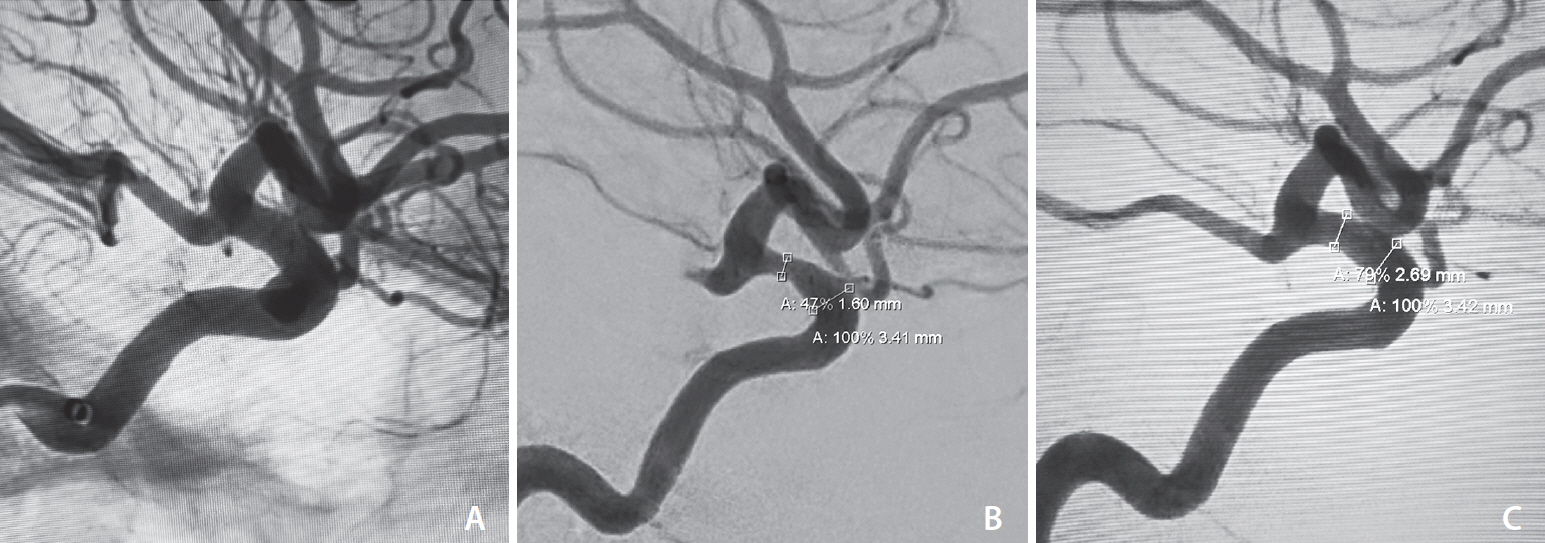
- In-stent stenosis is a feared complication of flow diversion treatment for cerebral aneurysms. We present 2 cases of patients treated with pipeline flow diversion for unruptured cerebral aneurysms. Initial perioperative dual antiplatelet therapy (DAPT) consisted of standard aspirin plus clopidogrel. At 6-month follow-up cerebral angiography, the patients were noted to have developed significant in-stent stenosis (63% and 53%). The patients were treated with cilostazol and clopidogrel for at least 6 months. Subsequent angiography at 1-year post-treatment showed significant improvement of the in-stent stenosis from 63% to 34% and 53% to 21%. The role of cilostazol as treatment of intracranial in-stent stenosis has not been previously described. Cilostazol’s vasodilatory effect and suppression of vascular smooth muscle proliferation provides ideal benefits in this setting. Cilostazol plus clopidogrel may be a safe and effective alternative to standard DAPT for treatment of in-stent stenosis following flow diversion and warrants further consideration and investigation.
Figure
Reference
-
1. Fiorella D, Lylyk P, Szikora I, Kelly ME, Albuquerque FC, McDougall CG, et al. Curative cerebrovascular reconstruction with the Pipeline embolization device: the emergence of definitive endovascular therapy for intracranial aneurysms. J Neurointerv Surg. 2018; 10(Suppl 1):i9–i18.
Article2. Daou B, Starke RM, Chalouhi N, Barros G, Tjoumakaris S, Rosenwasser RH, et al. P2Y12 reaction units: effect on hemorrhagic and thromboembolic complications in patients with cerebral aneurysms treated with the pipeline embolization device. Neurosurgery. 2016; 78:27–33.3. Daou B, Starke RM, Chalouhi N, Barros G, Tjoumakaris S, Rosenwasser RH, et al. P2Y12 reaction units: effect on hemorrhagic and thromboembolic complications in patients with cerebral aneurysms treated with the pipeline embolization device. Neurosurgery. 2016; 78:27–33.4. Nelson PK, Lylyk P, Szikora I, Wetzel SG, Wanke I, Fiorella D. The pipeline embolization device for the intracranial treatment of aneurysms trial. AJNR Am J Neuroradiol. 2011; 32:34–40.
Article5. Cohen JE, Gomori JM, Moscovici S, Leker RR, Itshayek E. Delayed complications after flow-diverter stenting: reactive in-stent stenosis and creeping stents. J Clin Neurosci. 2014; 21:1116–1122.
Article6. Mühl-Benninghaus R, Haußmann A, Simgen A, Tomori T, Reith W, Yilmaz U. Transient in-stent stenosis: a common finding after flow diverter implantation. J Neurointerv Surg. 2019; 11:196–199.
Article7. John S, Bain MD, Hui FK, Hussain MS, Masaryk TJ, Rasmussen PA, et al. Long-term follow-up of in-stent stenosis after pipeline flow diversion treatment of intracranial aneurysms. Neurosurgery. 2016; 78:862–867.
Article8. Wang T, Zhang CW, Richard SA, Chaohua W, Xie XD. Reactive in-stent stenosis of a pipeline embolization device in a child: a case report. Medicine (Baltimore). 2019; 98:e18092.9. Liu Y, Shakur Y, Yoshitake M, Kambayashi Ji J. Cilostazol (pletal): a dual inhibitor of cyclic nucleotide phosphodiesterase type 3 and adenosine uptake. Cardiovasc Drug Rev. 2001; 19:369–386.
Article10. Wallentin L. P2Y(12) inhibitors: differences in properties and mechanisms of action and potential consequences for clinical use. Eur Heart J. 2009; 30:1964–1977.
Article11. Karan V, Vyas D, Bohra V, Huded V. Ticagrelor use in Indian patients undergoing neuroendovascular procedures: a single center experience. Neurointervention. 2019; 14:125–130.
Article12. Toyoda K, Uchiyama S, Yamaguchi T, Easton JD, Kimura K, Hoshino H, CSPS.com Trial Investigators, et al. Dual antiplatelet therapy using cilostazol for secondary prevention in patients with high-risk ischaemic stroke in Japan: a multicentre, open-label, randomised controlled trial. Lancet Neurol. 2019; 18:539–548.
Article13. Stone GW, Brodie BR, Griffin JJ, Morice MC, Costantini C, St Goar FG, et al. Prospective, multicenter study of the safety and feasibility of primary stenting in acute myocardial infarction: in-hospital and 30-day results of the PAMI stent pilot trial. Primary Angioplasty in Myocardial Infarction Stent Pilot Trial Investigators. J Am Coll Cardiol. 1998; 31:23–30.14. Tamhane U, Meier P, Chetcuti S, Chen KY, Rha SW, Grossman MP, et al. Efficacy of cilostazol in reducing restenosis in patients undergoing contemporary stent based PCI: a meta-analysis of randomised controlled trials. EuroIntervention. 2009; 5:384–393.
Article15. Douglas JS Jr, Holmes DR Jr, Kereiakes DJ, Grines CL, Block E, Ghazzal ZM, Cilostazol for Restenosis Trial (CREST) Investigators, et al. Coronary stent restenosis in patients treated with cilostazol. Circulation. 2005; 112:2826–2832.
Article16. Takayama K, Taoka T, Nakagawa H, Myouchin K, Wada T, Sakamoto M, et al. Effect of cilostazol in preventing restenosis after carotid artery stenting using the carotid wallstent: a multicenter retrospective study. AJNR Am J Neuroradiol. 2012; 33:2167–2170.
Article17. Takigawa T, Matsumaru Y, Hayakawa M, Nemoto S, Matsumura A. Cilostazol reduces restenosis after carotid artery stenting. J Vasc Surg. 2010; 51:51–56.
Article18. Galyfos G, Geropapas G, Sigala F, Aggeli K, Sianou A, Filis K. Meta-analysis of studies evaluating the effect of cilostazol on major outcomes after carotid stenting. J Endovasc Ther. 2016; 23:186–195.
Article19. Nakagawa I, Wada T, Park HS, Nishimura F, Yamada S, Nakagawa H, et al. Platelet inhibition by adjunctive cilostazol suppresses the frequency of cerebral ischemic lesions after carotid artery stenting in patients with carotid artery stenosis. J Vasc Surg. 2014; 59:761–767.
Article20. Hassan AE, Zacharatos H, Grigoryan M, Tekle WG, Khan A, Siddiq F, et al. Open-label phase I clinical study to assess the safety and efficacy of cilostazol in patients undergoing internal carotid artery stent placement. Interv Neurol. 2017; 6:42–48.
Article21. Aguilar Pérez M, Bhogal P, Henkes E, Ganslandt O, Bäzner H, Henkes H. In-stent stenosis after p64 flow diverter treatment. Clin Neuroradiol. 2018; 28:563–568.
Article22. Schwartz RS, Chronos NA, Virmani R. Preclinical restenosis models and drug-eluting stents: still important, still much to learn. J Am Coll Cardiol. 2004; 44:1373–1385.23. Monteiro A, Lopes DK, Aghaebrahim A, Hanel R. Optical coherence tomography for elucidation of flow-diversion phenomena: the concept of endothelized mural thrombus behind reversible in-stent stenosis in flow-diverters. [published online ahead of print Mar 22, 2021]. Interv Neuroradiol. 2021.
Article24. Wu CK, Lin JW, Wu LC, Chang CH. Risk of heart failure hospitalization associated with cilostazol in diabetes: a nationwide case-crossover study. Front Pharmacol. 2019; 9:1467.
Article25. Zhang Z, Foster JK, Kolm P, Jurkovitz CT, Parker KM, Murrah NV, et al. Reduced 6-month resource use and costs associated with cilostazol in patients after successful coronary stent implantation: results from the Cilostazol for RESTenosis (CREST) trial. Am Heart J. 2006; 152:770–776.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Flow diverter stenting for intracranial aneurysms in the pediatric population: Two case reports and literature review
- Endovascular treatment of intracranial aneurysms: Past and present
- Physiologic Flow Diversion Coiling Technique for Wide-Necked Aneurysms with an Asymmetric Bidirectional Flow at the Aneurysm Neck
- In-Stent Stenosis of Stent Assisted Endovascular Treatment on Intracranial Complex Aneurysms
- Clinical and Angiographic Outcomes of Endovascular Treatment for Acute Intracranial Vertebral Artery Dissecting Aneurysms Using Double-Overlapping Stents : Low-Profile Visualized Intraluminal Support within Enterprise Stents