Endocrinol Metab.
2021 Aug;36(4):810-822. 10.3803/EnM.2021.988.
Tetrahydrocurcumin Ameliorates Kidney Injury and High Systolic Blood Pressure in High-Fat Diet-Induced Type 2 Diabetic Mice
- Affiliations
-
- 1Department of Internal Medicine, Yonsei University Wonju College of Medicine, Wonju, Korea
- 2Department of Physiology, Faculty of Medicine, Khon Kaen University, Khon Kaen, Thailand
- 3Cardiovascular Research Group, Khon Kaen University, Khon Kaen, Thailand
- 4Institution of Genetic Cohort, Yonsei University Wonju College of Medicine, Wonju, Korea
- 5Astrogen Inc., Daegu, Korea
- 6Department of Internal Medicine and Institute of Tissue Regeneration, BK21 FOUR Project, Soonchunhyang University College of Medicine, Cheonan, Korea
- KMID: 2519669
- DOI: http://doi.org/10.3803/EnM.2021.988
Abstract
- Background
Activation of the intrarenal renin-angiotensin system (RAS) is implicated in the pathogenesis of kidney injury and hypertension. We aimed to investigate the protective effect of tetrahydrocurcumin (THU) on intrarenal RAS expression, kidney injury, and systolic blood pressure (SBP) in high-fat diet (HFD)-induced type 2 diabetic mice.
Methods
Eight-week-old male mice were fed a regular diet (RD) or HFD for 12 weeks, and THU (50 or 100 mg/kg/day) was intragastrically administered with HFD. Physiological and metabolic changes were monitored and the expression of RAS components and markers of kidney injury were assessed.
Results
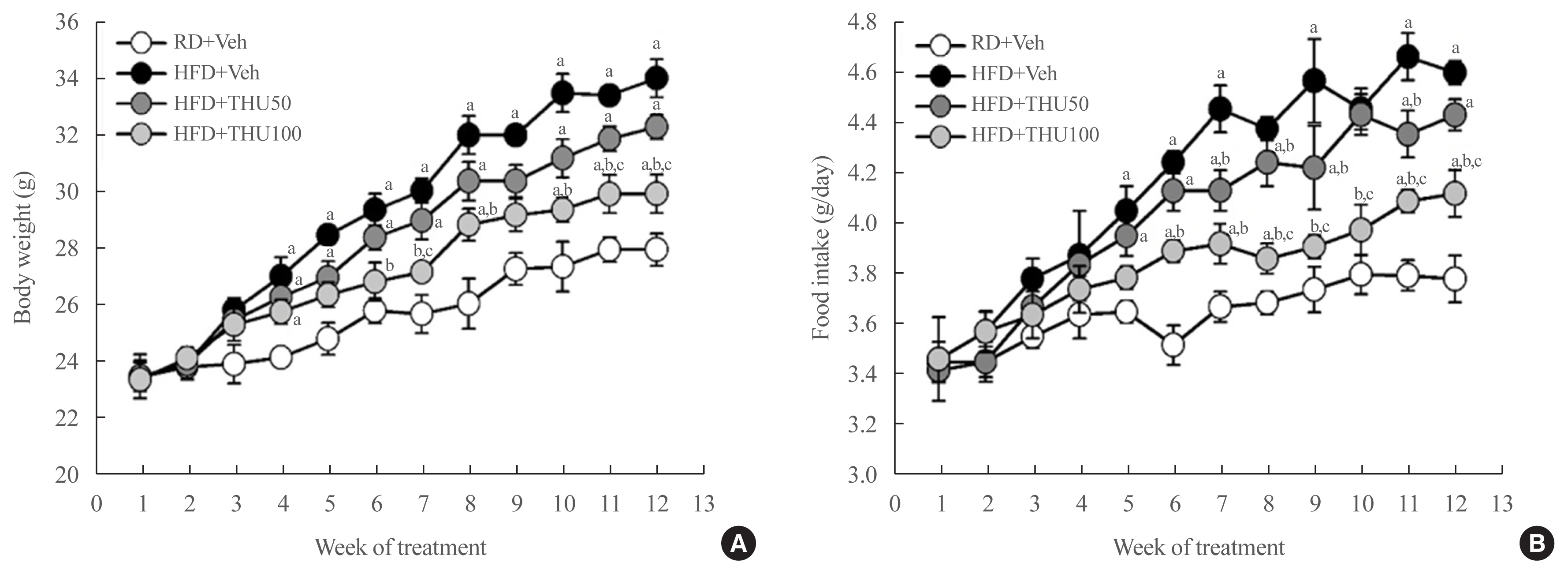
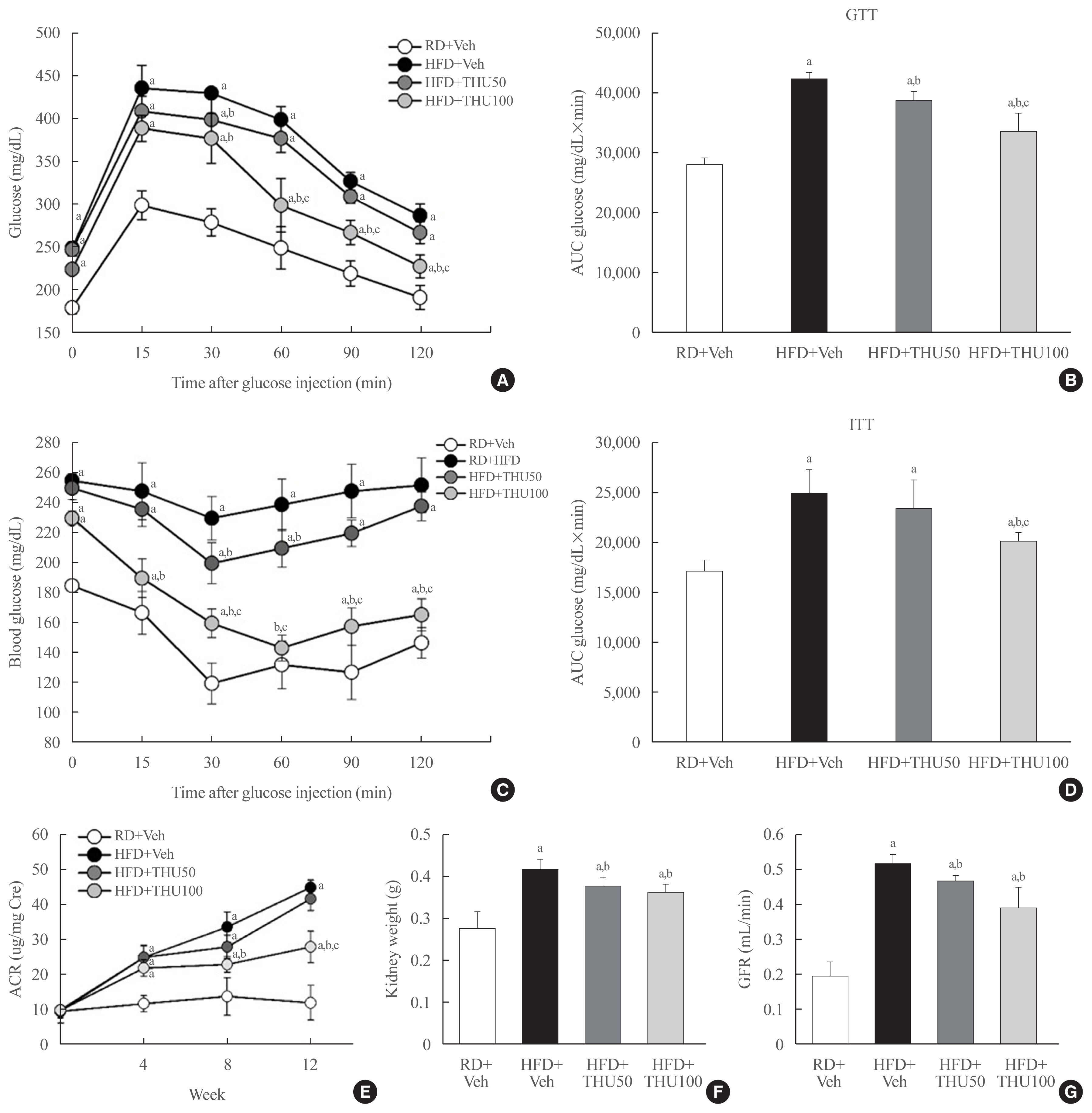
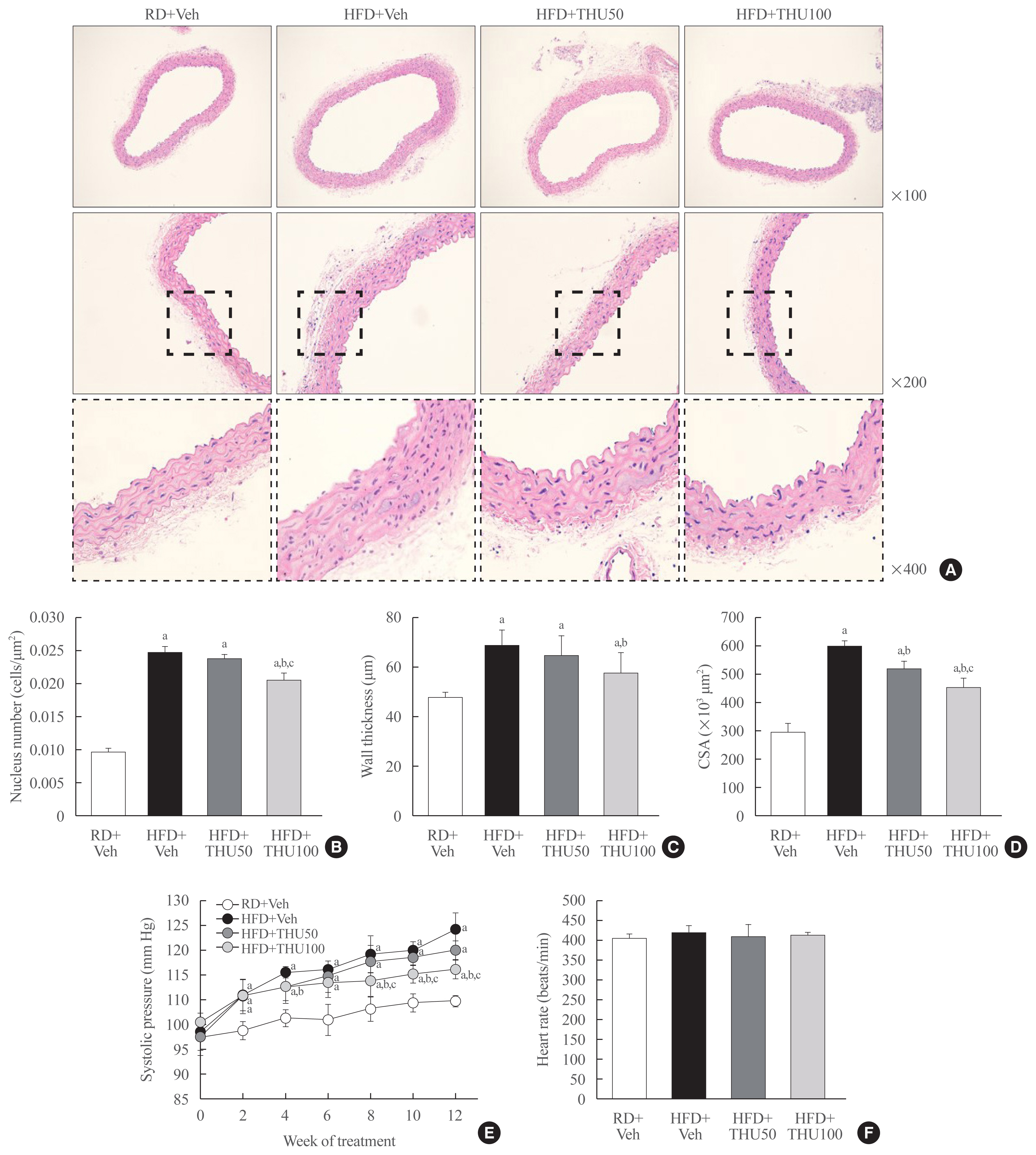
HFD-fed mice exhibited hyperglycemia, insulin resistance, and dyslipidemia compared to those in the RD group (P<0.05). Kidney injury in these mice was indicated by an increase in the ratio of albumin to creatinine, glomerular hypertrophy, and the effacement of podocyte foot processes. Expression of intrarenal angiotensin-converting enzyme, angiotensin II type I receptor, nicotinamide adenine dinucleotide phosphate (NADPH) oxidase-4, and monocyte chemoattractant protein-1 was also markedly increased in HFD-fed mice. HFD-fed mice exhibited elevated SBP that was accompanied by an increase in the wall thickness and vascular cross-sectional area (P<0.05), 12 weeks post-HFD consumption. Treatment with THU (100 mg/kg/day) suppressed intrarenal RAS activation, improved insulin sensitivity, and reduced SBP, thus, attenuating kidney injury in these mice.
Conclusion
THU alleviated kidney injury in mice with HFD-induced type 2 diabetes, possibly by blunting the activation of the intrarenal RAS/nicotinamide adenine dinucleotide phosphate oxidase IV (NOX4)/monocyte chemoattractant protein 1 (MCP-1) axis and by lowering the high SBP.
Keyword
Figure
Reference
-
1. Vasavada N, Agarwal R. Role of oxidative stress in diabetic nephropathy. Adv Chronic Kidney Dis. 2005; 12:146–54.
Article2. Rosenbaum M, Leibel RL. The physiology of body weight regulation: relevance to the etiology of obesity in children. Pediatrics. 1998; 101(3 Pt 2):525–39.
Article3. Hall JE, Crook ED, Jones DW, Wofford MR, Dubbert PM. Mechanisms of obesity-associated cardiovascular and renal disease. Am J Med Sci. 2002; 324:127–37.
Article4. Frohlich ED. Clinical management of the obese hypertensive patient. Cardiol Rev. 2002; 10:127–38.
Article5. Ledwozyw A, Michalak J, Stepien A, Kadziolka A. The relationship between plasma triglycerides, cholesterol, total lipids and lipid peroxidation products during human atherosclerosis. Clin Chim Acta. 1986; 155:275–83.
Article6. Kotsis VT, Stabouli SV, Papamichael CM, Zakopoulos NA. Impact of obesity in intima media thickness of carotid arteries. Obesity (Silver Spring). 2006; 14:1708–15.
Article7. Wong LK. Global burden of intracranial atherosclerosis. Int J Stroke. 2006; 1:158–9.
Article8. Kincaid-Smith P. Hypothesis: obesity and the insulin resistance syndrome play a major role in end-stage renal failure attributed to hypertension and labelled ‘hypertensive nephrosclerosis’. J Hypertens. 2004; 22:1051–5.9. Serra A, Romero R, Lopez D, Navarro M, Esteve A, Perez N, et al. Renal injury in the extremely obese patients with normal renal function. Kidney Int. 2008; 73:947–55.
Article10. Gallagher S. The challenges of obesity and skin integrity. Nurs Clin North Am. 2005; 40:325–35.
Article11. Tonneijck L, Muskiet MH, Smits MM, van Bommel EJ, Heerspink HJ, van Raalte DH, et al. Glomerular hyperfiltration in diabetes: mechanisms, clinical significance, and treatment. J Am Soc Nephrol. 2017; 28:1023–39.
Article12. Thallas-Bonke V, Jandeleit-Dahm KA, Cooper ME. Nox-4 and progressive kidney disease. Curr Opin Nephrol Hypertens. 2015; 24:74–80.
Article13. Hall JE, do Carmo JM, da Silva AA, Wang Z, Hall ME. Obesity, kidney dysfunction and hypertension: mechanistic links. Nat Rev Nephrol. 2019; 15:367–85.
Article14. Peterson JC, Adler S, Burkart JM, Greene T, Hebert LA, Hunsicker LG, et al. Blood pressure control, proteinuria, and the progression of renal disease. The modification of diet in renal disease study. Ann Intern Med. 1995; 123:754–62.
Article15. Viazzi F, Leoncini G, Pontremoli R. Antihypertensive treatment and renal protection: the role of drugs inhibiting the renin-angiotensin-aldosterone system. High Blood Press Cardiovasc Prev. 2013; 20:273–82.
Article16. Mogensen CE, Neldam S, Tikkanen I, Oren S, Viskoper R, Watts RW, et al. Randomised controlled trial of dual blockade of renin-angiotensin system in patients with hypertension, microalbuminuria, and non-insulin dependent diabetes: the candesartan and lisinopril microalbuminuria (CALM) study. BMJ. 2000; 321:1440–4.
Article17. Go YM, Jones DP. Redox compartmentalization in eukaryotic cells. Biochim Biophys Acta. 2008; 1780:1273–90.
Article18. Cooper ME, Mundel P, Boner G. Role of nephrin in renal disease including diabetic nephropathy. Semin Nephrol. 2002; 22:393–8.
Article19. Sarafidis PA, Khosla N, Bakris GL. Antihypertensive therapy in the presence of proteinuria. Am J Kidney Dis. 2007; 49:12–26.
Article20. Murugan P, Pari L. Antioxidant effect of tetrahydrocurcumin in streptozotocin-nicotinamide induced diabetic rats. Life Sci. 2006; 79:1720–8.
Article21. Nakmareong S, Kukongviriyapan U, Pakdeechote P, Donpunha W, Kukongviriyapan V, Kongyingyoes B, et al. Antioxidant and vascular protective effects of curcumin and tetrahydrocurcumin in rats with L-NAME-induced hypertension. Naunyn Schmiedebergs Arch Pharmacol. 2011; 383:519–29.
Article22. Zhang XX, Kong J, Yun K. Prevalence of diabetic nephropathy among patients with type 2 diabetes mellitus in China: a meta-analysis of observational studies. J Diabetes Res. 2020; 2020:2315607.
Article23. Zhang ZB, Luo DD, Xie JH, Xian YF, Lai ZQ, Liu YH, et al. Curcumin’s metabolites, tetrahydrocurcumin and octahydrocurcumin, possess superior anti-inflammatory effects in vivo through suppression of TAK1-NF-κB pathway. Front Pharmacol. 2018; 9:1181.
Article24. Noshahr ZS, Salmani H, Khajavi Rad A, Sahebkar A. Animal models of diabetes-associated renal injury. J Diabetes Res. 2020; 2020:9416419.
Article25. Masters SL, Latz E, O’Neill LA. The inflammasome in atherosclerosis and type 2 diabetes. Sci Transl Med. 2011; 3:81ps17.
Article26. Boini KM, Xia M, Abais JM, Li G, Pitzer AL, Gehr TW, et al. Activation of inflammasomes in podocyte injury of mice on the high fat diet: effects of ASC gene deletion and silencing. Biochim Biophys Acta. 2014; 1843:836–45.
Article27. Hunley TE, Ma LJ, Kon V. Scope and mechanisms of obesity-related renal disease. Curr Opin Nephrol Hypertens. 2010; 19:227–34.
Article28. Kalantar-Zadeh K, Kopple JD. Obesity paradox in patients on maintenance dialysis. Contrib Nephrol. 2006; 151:57–69.
Article29. Kamiyama M, Urushihara M, Morikawa T, Konishi Y, Imanishi M, Nishiyama A, et al. Oxidative stress/angiotensinogen/renin-angiotensin system axis in patients with diabetic nephropathy. Int J Mol Sci. 2013; 14:23045–62.
Article30. Chen CM, Juan SH, Chou HC. Hyperglycemia activates the renin-angiotensin system and induces epithelial-mesenchymal transition in streptozotocin-induced diabetic kidneys. J Renin Angiotensin Aldosterone Syst. 2018; 19:14703203188–03009.
Article31. Ustundag B, Canatan H, Cinkilinc N, Halifeoglu I, Bahcecioglu IH. Angiotensin converting enzyme (ACE) activity levels in insulin-independent diabetes mellitus and effect of ACE levels on diabetic patients with nephropathy. Cell Biochem Funct. 2000; 18:23–8.
Article32. Micakovic T, Papagiannarou S, Clark E, Kuzay Y, Abramovic K, Peters J, et al. The angiotensin II type 2 receptors protect renal tubule mitochondria in early stages of diabetes mellitus. Kidney Int. 2018; 94:937–50.
Article33. Lee YW, Lee WH, Kim PH. Role of NADPH oxidase in interleukin-4-induced monocyte chemoattractant protein-1 expression in vascular endothelium. Inflamm Res. 2010; 59:755–65.
Article34. Wu JC, Lai CS, Badmaev V, Nagabhushanam K, Ho CT, Pan MH. Tetrahydrocurcumin, a major metabolite of curcumin, induced autophagic cell death through coordinative modulation of PI3K/Akt-mTOR and MAPK signaling pathways in human leukemia HL-60 cells. Mol Nutr Food Res. 2011; 55:1646–54.
Article35. Sangartit W, Kukongviriyapan U, Donpunha W, Pakdeechote P, Kukongviriyapan V, Surawattanawan P, et al. Tetrahydrocurcumin protects against cadmium-induced hypertension, raised arterial stiffness and vascular remodeling in mice. PLoS One. 2014; 9:e114908.
Article36. Yaribeygi H, Sathyapalan T, Atkin SL, Sahebkar A. Molecular mechanisms linking oxidative stress and diabetes mellitus. Oxid Med Cell Longev. 2020; 2020:8609213.
Article37. Chen JW, Kong ZL, Tsai ML, Lo CY, Ho CT, Lai CS. Tetrahydrocurcumin ameliorates free fatty acid-induced hepatic steatosis and improves insulin resistance in HepG2 cells. J Food Drug Anal. 2018; 26:1075–85.
Article38. Pari L, Murugan P. Effect of tetrahydrocurcumin on blood glucose, plasma insulin and hepatic key enzymes in streptozotocin induced diabetic rats. J Basic Clin Physiol Pharmacol. 2005; 16:257–74.
Article39. Jauregui A, Mintz DH, Mundel P, Fornoni A. Role of altered insulin signaling pathways in the pathogenesis of podocyte malfunction and microalbuminuria. Curr Opin Nephrol Hypertens. 2009; 18:539–45.
Article40. Reiser J, Mundel P. Dual effects of RAS blockade on blood pressure and podocyte function. Curr Hypertens Rep. 2007; 9:403–8.
Article41. Yuan Y, Xu X, Zhao C, Zhao M, Wang H, Zhang B, et al. The roles of oxidative stress, endoplasmic reticulum stress, and autophagy in aldosterone/mineralocorticoid receptor-induced podocyte injury. Lab Invest. 2015; 95:1374–86.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Blood Sugar Control and Low-Carbohydrate High-Fat Diet
- Effects of a Low-carbohydrate, High-fat Diet
- Effect of Fermented Yacon (Smallanthus Sonchifolius) Leaves Tea on Blood Glucose Levels and Glucose Metabolism in High-Fat Diet and Streptozotocin-Induced Type 2 Diabetic Mice
- Partial Deletion of Perk Improved High-Fat Diet-Induced Glucose Intolerance in Mice
- Short-term resistance training improves lipid metabolism and inflammation in diet-induced obese mice