Ann Hepatobiliary Pancreat Surg.
2021 Aug;25(3):315-327. 10.14701/ahbps.2021.25.3.315.
Mutations of p53 associated with pancreatic cancer and therapeutic implications
- Affiliations
-
- 1Algoma District Cancer Program, Sault Area Hospital, Sault Ste. Marie, ON, Canada,
- 2Section of Internal Medicine, Division of Clinical Sciences, Northern Ontario School of Medicine, Sudbury, ON, Canada
- KMID: 2519287
- DOI: http://doi.org/10.14701/ahbps.2021.25.3.315
Abstract
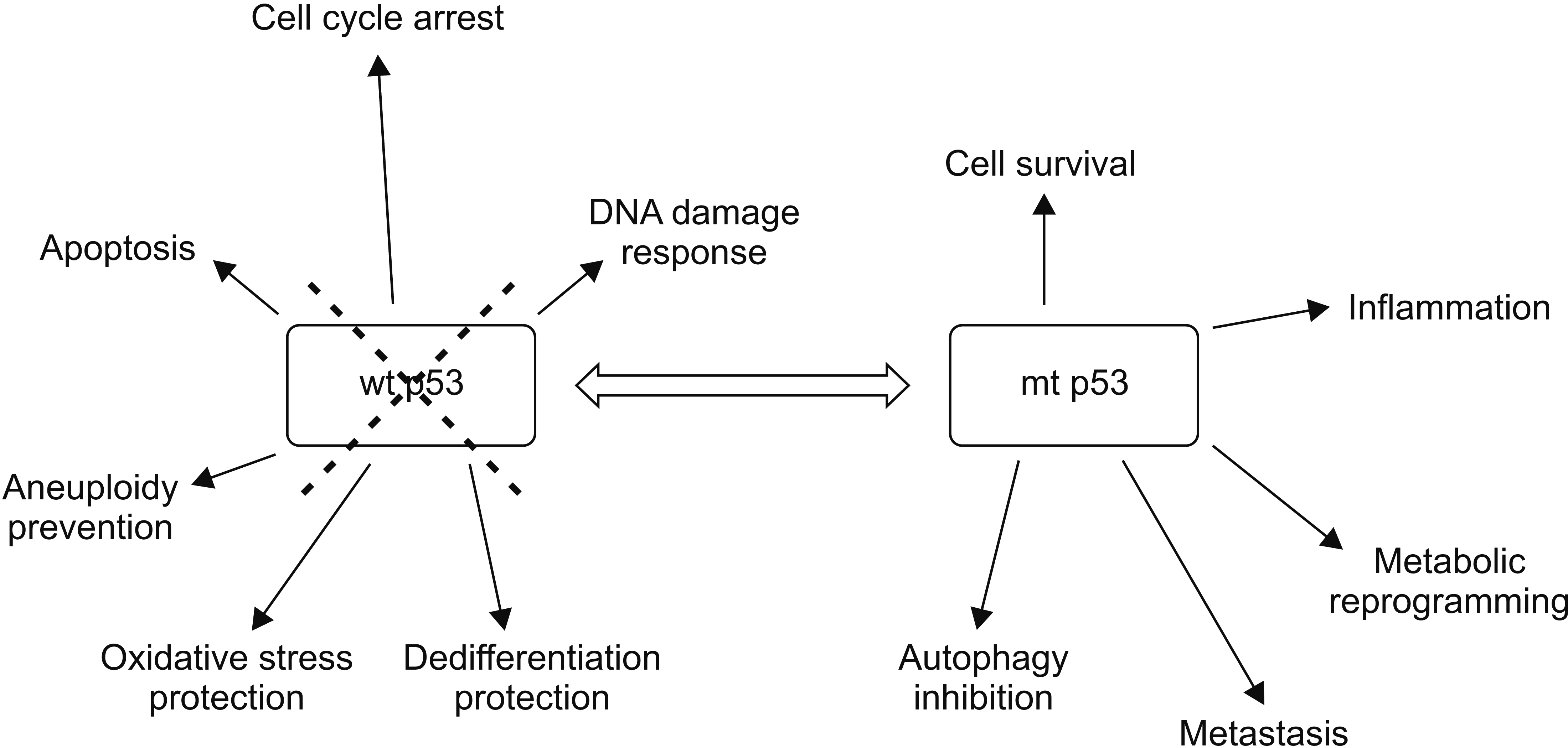
- Pancreatic adenocarcinoma is a malignancy with rising incidence and grim prognosis. Despite improvements in therapeutics for treating metastatic pancreatic cancer, this disease is invariably fatal with survival time less than a few years. New molecular understanding of the pathogenesis of pancreatic adenocarcinoma based on efforts led by The Cancer Genome Atlas and other groups has elucidated the landscape of this disease and started to produce therapeutic results, leading to the first introduction of targeted therapies for subsets of pancreatic cancers bearing specific molecular lesions such as BRCA mutations. These efforts have highlighted that subsets of pancreatic cancers are particularly sensitive to chemotherapy. The most common molecular lesions in pancreatic adenocarcinomas are mutations in an oncogene KRAS and the TP53 gene that encodes for tumor suppressor protein p53. This paper will review the landscape of pancreatic cancers, focusing on mutations of p53, a major tumor suppressor protein, in pancreatic cancers and possible therapeutic repercussions.
Figure
Reference
-
1. Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH, et al. 2016; Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin. 66:271–289. DOI: 10.3322/caac.21349. PMID: 27253694.
Article2. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. 2018; Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 68:394–424. DOI: 10.3322/caac.21492. PMID: 30207593.
Article3. Cancer Genome Atlas Research Network. 2017; Integrated genomic characterization of pancreatic ductal adenocarcinoma. Cancer Cell. 32:185–203.e13. DOI: 10.1016/j.ccell.2017.07.007. PMID: 28810144. PMCID: PMC5964983.4. AACR Project GENIE Consortium. 2017; AACR Project GENIE: powering precision medicine through an international consortium. Cancer Discov. 7:818–831. DOI: 10.1158/2159-8290.CD-17-0151. PMID: 28572459. PMCID: PMC5611790.5. Zehir A, Benayed R, Shah RH, Syed A, Middha S, Kim HR, et al. 2017; Mutational landscape of metastatic cancer revealed from prospective clinical sequencing of 10,000 patients. Nat Med. 23:703–713. DOI: 10.1038/nm.4333. PMID: 28481359. PMCID: PMC5461196.6. Zhang C, Liu J, Xu D, Zhang T, Hu W, Feng Z. 2020; Gain-of-function mutant p53 in cancer progression and therapy. J Mol Cell Biol. 12:674–687. DOI: 10.1093/jmcb/mjaa040. PMID: 32722796. PMCID: PMC7749743.
Article7. Amelio I, Melino G. 2020; Context is everything: extrinsic signalling and gain-of-function p53 mutants. Cell Death Discov. 6:16. DOI: 10.1038/s41420-020-0251-x. PMID: 32218993. PMCID: PMC7090043.
Article8. Voutsadakis IA. 2007; Pathogenesis of colorectal carcinoma and therapeutic implications: the roles of the ubiquitin-proteasome system and Cox-2. J Cell Mol Med. 11:252–285. DOI: 10.1111/j.1582-4934.2007.00032.x. PMID: 17488476. PMCID: PMC3822826.
Article9. Voutsadakis IA. 2008; The ubiquitin-proteasome system in colorectal cancer. Biochim Biophys Acta. 1782:800–808. DOI: 10.1016/j.bbadis.2008.06.007. PMID: 18619533.
Article10. Fukuda A. 2015; Molecular mechanism of intraductal papillary mucinous neoplasm and intraductal papillary mucinous neoplasm-derived pancreatic ductal adenocarcinoma. J Hepatobiliary Pancreat Sci. 22:519–523. DOI: 10.1002/jhbp.246. PMID: 25900667.
Article11. Fryzek JP, Garabrant DH, Schenk M, Kinnard M, Greenson JK, Sarkar FH. 2006; The association between selected risk factors for pancreatic cancer and the expression of p53 and K-ras codon 12 mutations. Int J Gastrointest Cancer. 37:139–145. DOI: 10.1007/s12029-007-9005-8. PMID: 18049799.
Article12. Crous-Bou M, Porta M, López T, Jariod M, Malats N, Alguacil J, et al. 2007; Lifetime history of tobacco consumption and K-ras mutations in exocrine pancreatic cancer. Pancreas. 35:135–141. DOI: 10.1097/mpa.0b013e31805d8fa4. PMID: 17632319.
Article13. Blackford A, Parmigiani G, Kensler TW, Wolfgang C, Jones S, Zhang X, et al. 2009; Genetic mutations associated with cigarette smoking in pancreatic cancer. Cancer Res. 69:3681–3688. DOI: 10.1158/0008-5472.CAN-09-0015. PMID: 19351817. PMCID: PMC2669837.
Article14. Das S, Berlin J, Cardin D. 2018; Harnessing the immune system in pancreatic cancer. Curr Treat Options Oncol. 19:48. DOI: 10.1007/s11864-018-0566-5. PMID: 30128712. PMCID: PMC6524529.
Article15. Gukovsky I, Li N, Todoric J, Gukovskaya A, Karin M. 2013; Inflammation, autophagy, and obesity: common features in the pathogenesis of pancreatitis and pancreatic cancer. Gastroenterology. 144:1199–1209.e4. DOI: 10.1053/j.gastro.2013.02.007. PMID: 23622129. PMCID: PMC3786712.
Article16. Green DR, Galluzzi L, Kroemer G. 2011; Mitochondria and the autophagy-inflammation-cell death axis in organismal aging. Science. 333:1109–1112. DOI: 10.1126/science.1201940. PMID: 21868666. PMCID: PMC3405151.
Article17. Yang A, Rajeshkumar NV, Wang X, Yabuuchi S, Alexander BM, Chu GC, et al. 2014; Autophagy is critical for pancreatic tumor growth and progression in tumors with p53 alterations. Cancer Discov. 4:905–913. DOI: 10.1158/2159-8290.CD-14-0362. PMID: 24875860. PMCID: PMC4125497.
Article18. Diakopoulos KN, Lesina M, Wörmann S, Song L, Aichler M, Schild L, et al. 2015; Impaired autophagy induces chronic atrophic pancreatitis in mice via sex- and nutrition-dependent processes. Gastroenterology. 148:626–638.e17. DOI: 10.1053/j.gastro.2014.12.003. PMID: 25497209.
Article19. Wörmann SM, Song L, Ai J, Diakopoulos KN, Kurkowski MU, Görgülü K, et al. 2016; Loss of P53 function activates JAK2-STAT3 signaling to promote pancreatic tumor growth, stroma modification, and gemcitabine resistance in mice and is associated with patient survival. Gastroenterology. 151:180–193.e12. DOI: 10.1053/j.gastro.2016.03.010. PMID: 27003603.20. Rosenfeldt MT, O'Prey J, Morton JP, Nixon C, MacKay G, Mrowinska A, et al. 2013; p53 status determines the role of autophagy in pancreatic tumour development. Nature. 504:296–300. DOI: 10.1038/nature12865. PMID: 24305049.
Article21. Voutsadakis IA. 2017; Obesity and diabetes as prognostic factors in patients with colorectal cancer. Diabetes Metab Syndr. 11 Suppl 1:S109–S114. DOI: 10.1016/j.dsx.2016.12.018. PMID: 27989518.
Article22. Zhang AMY, Magrill J, de Winter TJJ, Hu X, Skovsø S, Schaeffer DF, et al. 2019; Endogenous hyperinsulinemia contributes to pancreatic cancer development. Cell Metab. 30:403–404. DOI: 10.1016/j.cmet.2019.07.003. PMID: 31378465.
Article23. Maddipati R, Stanger BZ. 2015; Pancreatic Cancer Metastases Harbor Evidence of Polyclonality. Cancer Discov. 5:1086–1097. DOI: 10.1158/2159-8290.CD-15-0120. PMID: 26209539. PMCID: PMC4657730.
Article24. Muniz VP, Barnes JM, Paliwal S, Zhang X, Tang X, Chen S, et al. 2011; The ARF tumor suppressor inhibits tumor cell colonization independent of p53 in a novel mouse model of pancreatic ductal adenocarcinoma metastasis. Mol Cancer Res. 9:867–877. DOI: 10.1158/1541-7786.MCR-10-0475. PMID: 21636682. PMCID: PMC3140613.
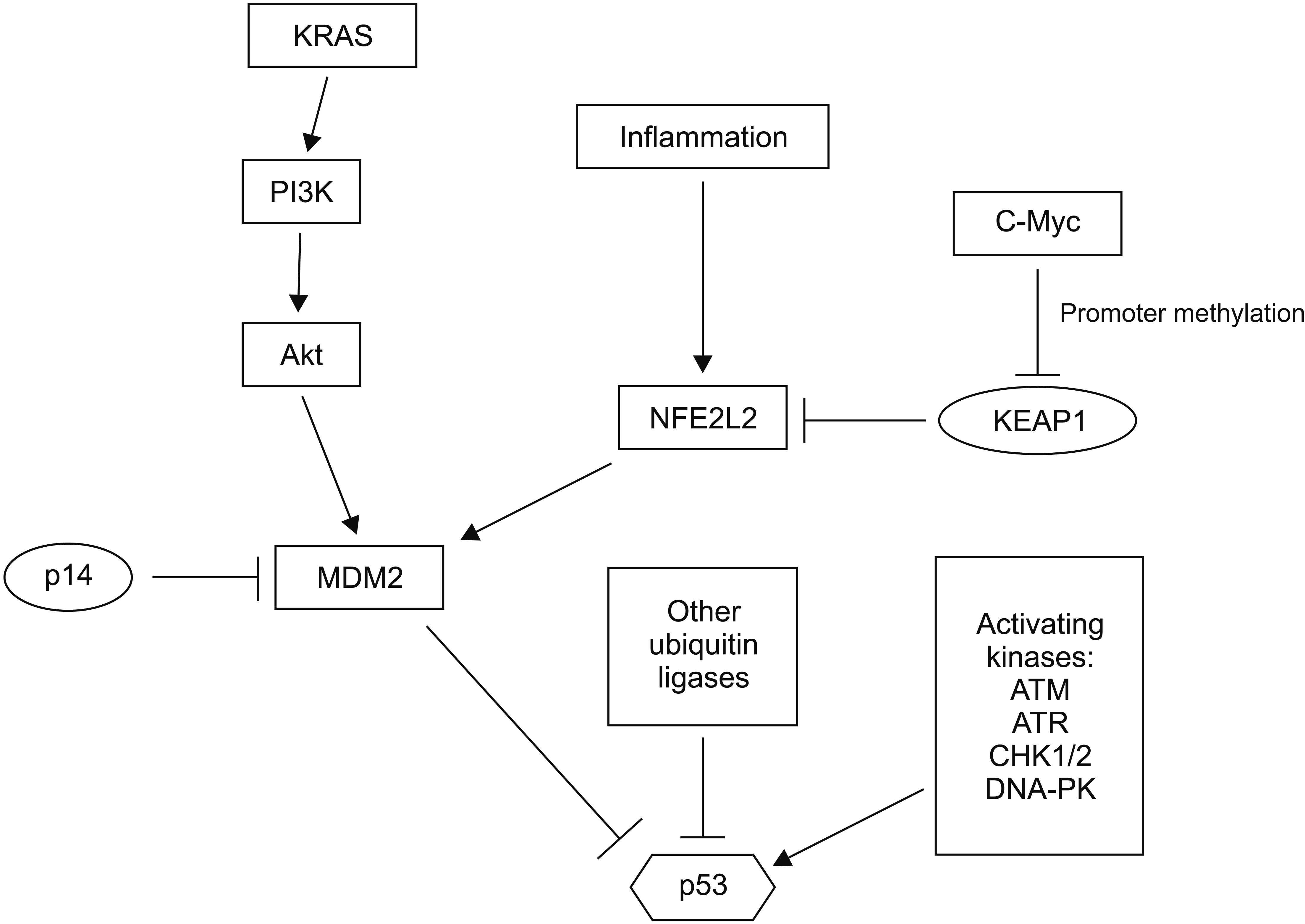
Article25. Hayes AJ, Skouras C, Haugk B, Charnley RM. 2015; Keap1-Nrf2 signalling in pancreatic cancer. Int J Biochem Cell Biol. 65:288–299. DOI: 10.1016/j.biocel.2015.06.017. PMID: 26117456.
Article26. Voutsadakis IA. 2017; Proteasome expression and activity in cancer and cancer stem cells. Tumour Biol. 39:1010428317692248. DOI: 10.1177/1010428317692248. PMID: 28345458.
Article27. Todoric J, Antonucci L, Di Caro G, Li N, Wu X, Lytle NK, et al. 2017; Stress-activated NRF2-MDM2 cascade controls neoplastic progression in pancreas. Cancer Cell. 32:824–839.e8. DOI: 10.1016/j.ccell.2017.10.011. PMID: 29153842. PMCID: PMC5730340.
Article28. Torrente L, DeNicola GM. 2017; Stressing out PanIN: NRF2 pushes over the edge. Cancer Cell. 32:723–725. DOI: 10.1016/j.ccell.2017.11.014. PMID: 29232549.
Article29. Zhang B, Xu J, Li C, Shi S, Ji S, Xu W, et al. 2016; MBD1 is an epigenetic regulator of KEAP1 in pancreatic cancer. Curr Mol Med. 16:404–411. DOI: 10.2174/1566524016666160316154150. PMID: 26980696.
Article30. Nayak G, Cooper GM. 2012; p53 is a major component of the transcriptional and apoptotic program regulated by PI 3-kinase/Akt/GSK3 signaling. Cell Death Dis. 3:e400. DOI: 10.1038/cddis.2012.138. PMID: 23059819. PMCID: PMC3481126.
Article31. Tang Y, Luo J, Zhang W, Gu W. 2006; Tip60-dependent acetylation of p53 modulates the decision between cell-cycle arrest and apoptosis. Mol Cell. 24:827–839. DOI: 10.1016/j.molcel.2006.11.021. PMID: 17189186.
Article32. Voutsadakis IA. 2012; Ubiquitination and the Ubiquitin-Proteasome System as regulators of transcription and transcription factors in epithelial mesenchymal transition of cancer. Tumour Biol. 33:897–910. DOI: 10.1007/s13277-012-0355-x. PMID: 22399444.33. Inoue S, Hao Z, Elia AJ, Cescon D, Zhou L, Silvester J, et al. 2013; Mule/Huwe1/Arf-BP1 suppresses Ras-driven tumorigenesis by preventing c-Myc/Miz1-mediated down-regulation of p21 and p15. Genes Dev. 27:1101–1114. DOI: 10.1101/gad.214577.113. PMID: 23699408. PMCID: PMC3672645.
Article34. Cassidy KB, Bang S, Kurokawa M, Gerber SA. 2020; Direct regulation of Chk1 protein stability by E3 ubiquitin ligase HUWE1. FEBS J. 287:1985–1999. DOI: 10.1111/febs.15132. PMID: 31713291. PMCID: PMC7226928.
Article35. Voutsadakis IA. 2013; Ubiquitin- and ubiquitin-like proteins-conjugating enzymes (E2s) in breast cancer. Mol Biol Rep. 40:2019–2034. DOI: 10.1007/s11033-012-2261-0. PMID: 23187732.
Article36. Mueller S, Engleitner T, Maresch R, Zukowska M, Lange S, Kaltenbacher T, et al. 2018; Evolutionary routes and KRAS dosage define pancreatic cancer phenotypes. Nature. 554:62–68. DOI: 10.1038/nature25459. PMID: 29364867. PMCID: PMC6097607.
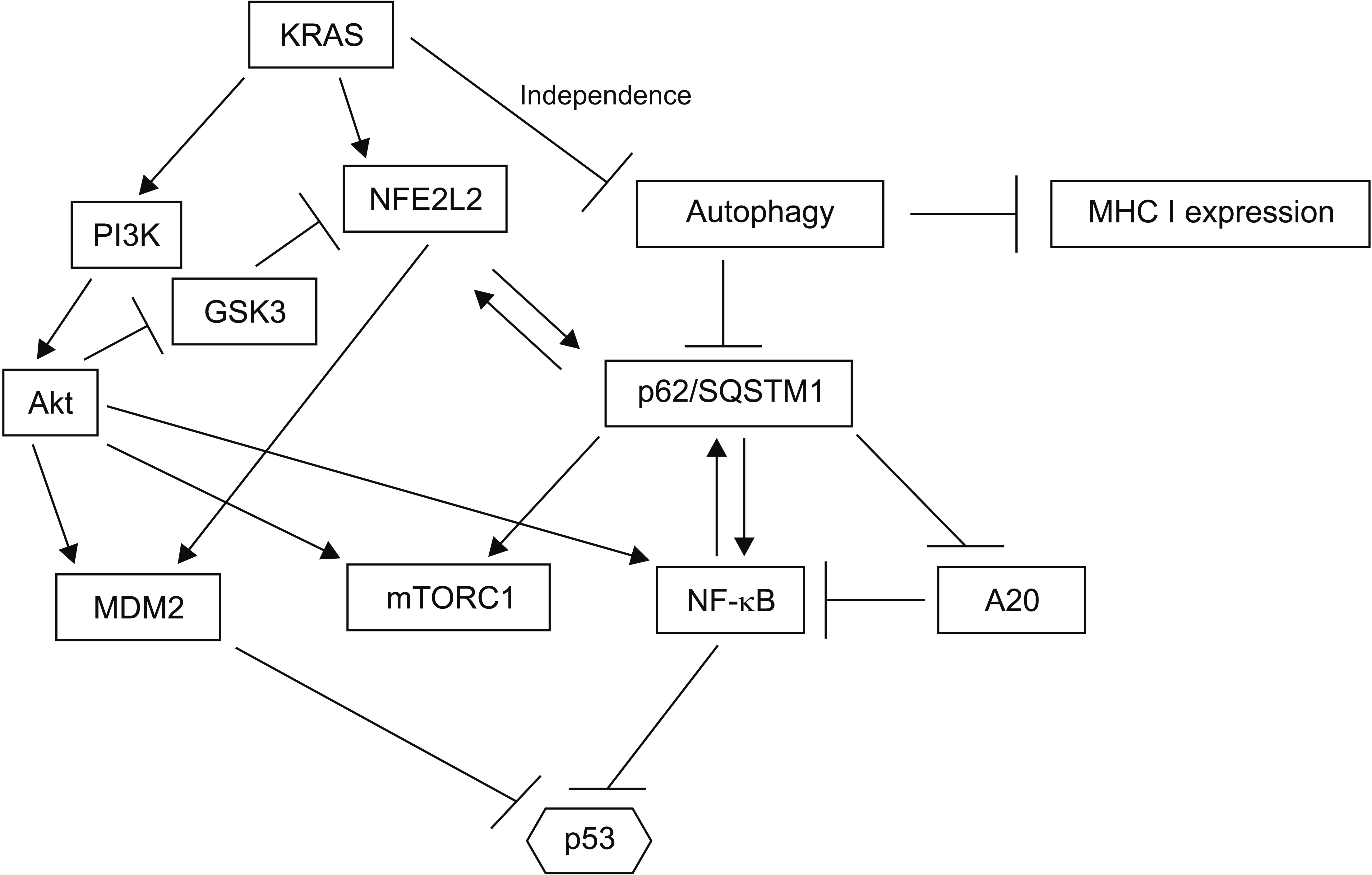
Article37. Alvarez-Meythaler JG, Garcia-Mayea Y, Mir C, Kondoh H, LLeonart ME. 2020; Autophagy takes center stage as a possible cancer hallmark. Front Oncol. 10:586069. DOI: 10.3389/fonc.2020.586069. PMID: 33194736. PMCID: PMC7643020.
Article38. Piffoux M, Eriau E, Cassier PA. 2021; Autophagy as a therapeutic target in pancreatic cancer. Br J Cancer. 124:333–344. DOI: 10.1038/s41416-020-01039-5. PMID: 32929194. PMCID: PMC7852577.
Article39. Yamamoto K, Venida A, Perera RM, Kimmelman AC. 2020; Selective autophagy of MHC-I promotes immune evasion of pancreatic cancer. Autophagy. 16:1524–1525. DOI: 10.1080/15548627.2020.1769973. PMID: 32459143. PMCID: PMC7469632.
Article40. Bryant KL, Stalnecker CA, Zeitouni D, Klomp JE, Peng S, Tikunov AP, et al. 2019; Combination of ERK and autophagy inhibition as a treatment approach for pancreatic cancer. Nat Med. 25:628–640. DOI: 10.1038/s41591-019-0368-8. PMID: 30833752. PMCID: PMC6484853.
Article41. Kinsey CG, Camolotto SA, Boespflug AM, Guillen KP, Foth M, Truong A, et al. 2019; Protective autophagy elicited by RAF→MEK→ERK inhibition suggests a treatment strategy for RAS-driven cancers. Nat Med. 25:620–627. DOI: 10.1038/s41591-019-0367-9. PMID: 30833748. PMCID: PMC6452642.
Article42. Sánchez-Martín P, Saito T, Komatsu M. 2019; p62/SQSTM1: 'Jack of all trades' in health and cancer. FEBS J. 286:8–23. DOI: 10.1111/febs.14712. PMID: 30499183. PMCID: PMC7379270.43. Yang S, Qiang L, Sample A, Shah P, He YY. 2017; NF-κB signaling activation induced by chloroquine requires autophagosome, p62 protein, and c-Jun N-terminal Kinase (JNK) signaling and promotes tumor cell resistance. J Biol Chem. 292:3379–3388. DOI: 10.1074/jbc.M116.756536. PMID: 28082672. PMCID: PMC5336170.
Article44. Kanayama M, Inoue M, Danzaki K, Hammer G, He YW, Shinohara ML. 2015; Autophagy enhances NFkB activity in specific tissue macrophages by sequestering A20 to boost antifungal immunity. Nat Commun. 6:5779. DOI: 10.1038/ncomms6779.
Article45. Cross DA, Alessi DR, Cohen P, Andjelkovich M, Hemmings BA. 1995; Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature. 378:785–789. DOI: 10.1038/378785a0. PMID: 8524413.
Article46. Voutsadakis IA. 2012; The ubiquitin-proteasome system and signal transduction pathways regulating epithelial mesenchymal transition of cancer. J Biomed Sci. 19:67. DOI: 10.1186/1423-0127-19-67. PMID: 22827778. PMCID: PMC3418218.
Article47. Pinho AV, Rooman I, Real FX. 2011; p53-dependent regulation of growth, epithelial-mesenchymal transition and stemness in normal pancreatic epithelial cells. Cell Cycle. 10:1312–1321. DOI: 10.4161/cc.10.8.15363. PMID: 21490434.
Article48. Chang CJ, Chao CH, Xia W, Yang JY, Xiong Y, Li CW, et al. 2011; p53 regulates epithelial-mesenchymal transition and stem cell properties through modulating miRNAs. Nat Cell Biol. 13:317–323. DOI: 10.1038/ncb2173. PMID: 21336307. PMCID: PMC3075845.
Article49. Voutsadakis IA. 2014; The chemosensitivity of testicular germ cell tumors. Cell Oncol (Dordr). 37:79–94. DOI: 10.1007/s13402-014-0168-6. PMID: 24692098.
Article50. Ji Q, Hao X, Zhang M, Tang W, Yang M, Li L, et al. 2009; MicroRNA miR-34 inhibits human pancreatic cancer tumor-initiating cells. PLoS One. 4:e6816. DOI: 10.1371/journal.pone.0006816. PMID: 19714243. PMCID: PMC2729376.
Article51. Jain AK, Allton K, Iacovino M, Mahen E, Milczarek RJ, Zwaka TP, et al. 2012; p53 regulates cell cycle and microRNAs to promote differentiation of human embryonic stem cells. PLoS Biol. 10:e1001268. DOI: 10.1371/journal.pbio.1001268. PMID: 22389628. PMCID: PMC3289600.
Article52. Saleh AD, Savage JE, Cao L, Soule BP, Ly D, DeGraff W, et al. 2011; Cellular stress induced alterations in microRNA let-7a and let-7b expression are dependent on p53. PLoS One. 6:e24429. DOI: 10.1371/journal.pone.0024429. PMID: 22022355. PMCID: PMC3191136.
Article53. Liu Y, Wang D, Zhou M, Chen H, Wang H, Min J, et al. 2021; The KRAS/Lin28B axis maintains stemness of pancreatic cancer cells via the let-7i/TET3 pathway. Mol Oncol. 15:262–278. DOI: 10.1002/1878-0261.12836. PMID: 33107691. PMCID: PMC7782082.
Article54. Ghosh B, Leach SD. 2011; p53: guardian of pancreatic epithelial identity. Cell Cycle. 10:1717. DOI: 10.4161/cc.10.11.15686. PMID: 21597331. PMCID: PMC3233485.
Article55. Wang W, Friedland SC, Guo B, O'Dell MR, Alexander WB, Whitney-Miller CL, et al. 2019; ARID1A, a SWI/SNF subunit, is critical to acinar cell homeostasis and regeneration and is a barrier to transformation and epithelial-mesenchymal transition in the pancreas. Gut. 68:1245–1258. DOI: 10.1136/gutjnl-2017-315541. PMID: 30228219. PMCID: PMC6551318.
Article56. Lo HG, Jin RU, Sibbel G, Liu D, Karki A, Joens MS, et al. 2017; A single transcription factor is sufficient to induce and maintain secretory cell architecture. Genes Dev. 31:154–171. DOI: 10.1101/gad.285684.116. PMID: 28174210. PMCID: PMC5322730.
Article57. Roy N, Takeuchi KK, Ruggeri JM, Bailey P, Chang D, Li J, et al. 2016; PDX1 dynamically regulates pancreatic ductal adenocarcinoma initiation and maintenance. Genes Dev. 30:2669–2683. DOI: 10.1101/gad.291021.116. PMID: 28087712. PMCID: PMC5238727.
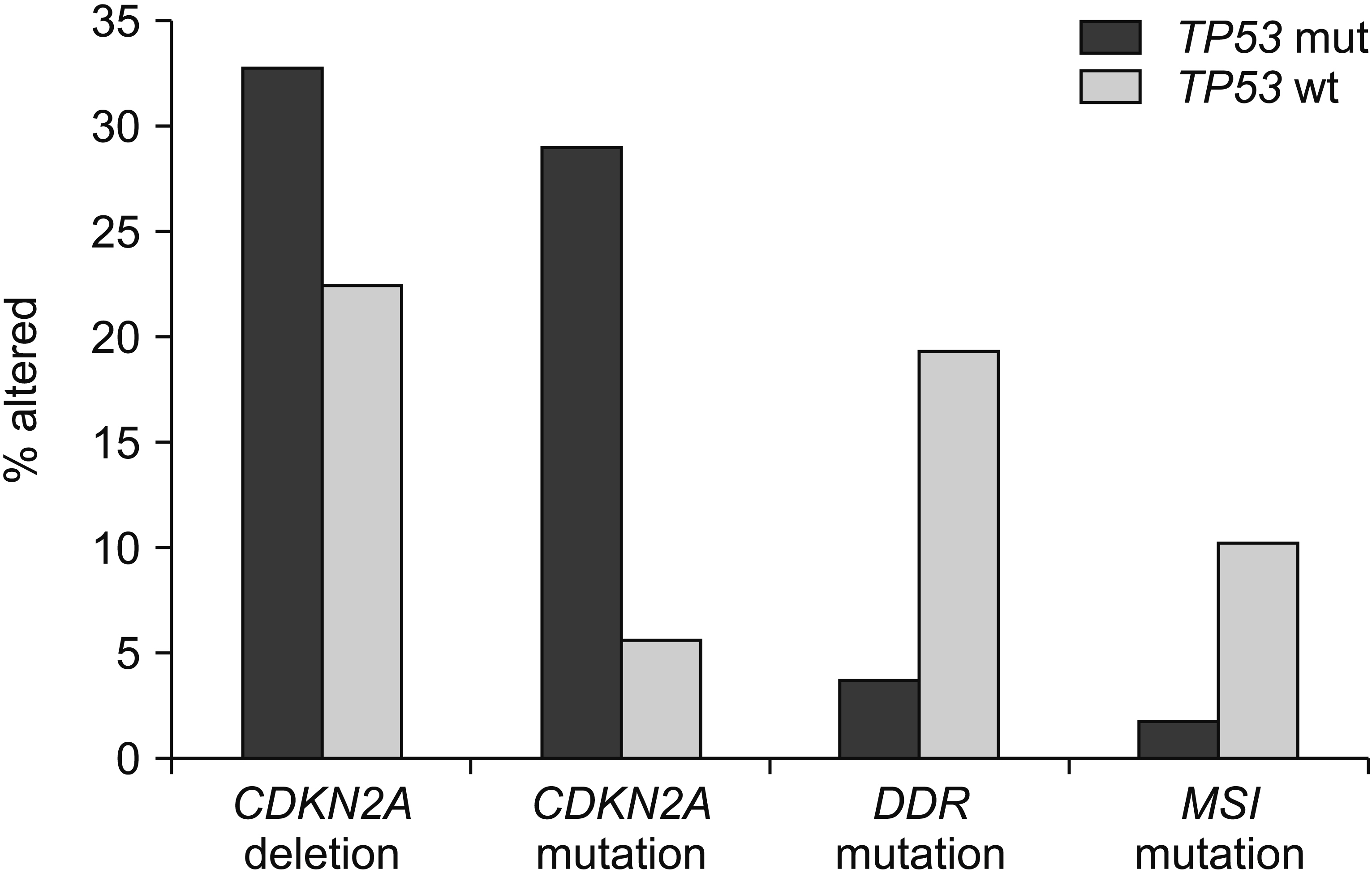
Article58. Gout J, Perkhofer L, Morawe M, Arnold F, Ihle M, Biber S, et al. 2021; Synergistic targeting and resistance to PARP inhibition in DNA damage repair-deficient pancreatic cancer. Gut. 70:743–760. DOI: 10.1136/gutjnl-2019-319970. PMID: 32873698. PMCID: PMC7948173.
Article59. Ischenko I, Zhi J, Moll UM, Nemajerova A, Petrenko O. 2013; Direct reprogramming by oncogenic Ras and Myc. Proc Natl Acad Sci U S A. 110:3937–3942. DOI: 10.1073/pnas.1219592110. PMID: 23431158. PMCID: PMC3593888.
Article60. Lancho O, Herranz D. 2018; The MYC enhancer-ome: long-range transcriptional regulation of MYC in cancer. Trends Cancer. 4:810–822. DOI: 10.1016/j.trecan.2018.10.003. PMID: 30470303. PMCID: PMC6260942.
Article61. Voutsadakis IA. 2021; Amplification of 8p11. 23 in cancers and the role of amplicon genes. Life Sci. 264:118729. DOI: 10.1016/j.lfs.2020.118729. PMID: 33166592.62. Masetti M, Acquaviva G, Visani M, Tallini G, Fornelli A, Ragazzi M, et al. 2018; Long-term survivors of pancreatic adenocarcinoma show low rates of genetic alterations in KRAS, TP53 and SMAD4. Cancer Biomark. 21:323–334. DOI: 10.3233/CBM-170464. PMID: 29103024.
Article63. Stravodimou A, Mazzoccoli G, Voutsadakis IA. 2012; Peroxisome proliferator-activated receptor gamma and regulations by the ubiquitin-proteasome system in pancreatic cancer. PPAR Res. 2012:367450. DOI: 10.1155/2012/367450. PMID: 23049538. PMCID: PMC3459232.
Article64. Khandekar MJ, Banks AS, Laznik-Bogoslavski D, White JP, Choi JH, Kazak L, et al. 2018; Noncanonical agonist PPARγ ligands modulate the response to DNA damage and sensitize cancer cells to cytotoxic chemotherapy. Proc Natl Acad Sci U S A. 115:561–566. DOI: 10.1073/pnas.1717776115. PMID: 29295932. PMCID: PMC5776997.
Article65. Deng X, Li Y, Gu S, Chen Y, Yu B, Su J, et al. 2020; p53 affects PGC1α stability through AKT/GSK-3β to enhance cisplatin sensitivity in non-small cell lung cancer. Front Oncol. 10:1252. DOI: 10.3389/fonc.2020.01252. PMID: 32974127. PMCID: PMC7471661.
Article66. Maniati E, Bossard M, Cook N, Candido JB, Emami-Shahri N, Nedospasov SA, et al. 2011; Crosstalk between the canonical NF-κB and Notch signaling pathways inhibits Pparγ expression and promotes pancreatic cancer progression in mice. J Clin Invest. 121:4685–4699. DOI: 10.1172/JCI45797. PMID: 22056382. PMCID: PMC3225987.
Article67. Fiorini C, Cordani M, Padroni C, Blandino G, Di Agostino S, Donadelli M. 2015; Mutant p53 stimulates chemoresistance of pancreatic adenocarcinoma cells to gemcitabine. Biochim Biophys Acta. 1853:89–100. DOI: 10.1016/j.bbamcr.2014.10.003. PMID: 25311384.
Article68. Wiegering A, Matthes N, Mühling B, Koospal M, Quenzer A, Peter S, et al. 2017; Reactivating p53 and inducing tumor apoptosis (RITA) enhances the response of RITA-sensitive colorectal cancer cells to chemotherapeutic agents 5-fluorouracil and oxaliplatin. Neoplasia. 19:301–309. DOI: 10.1016/j.neo.2017.01.007. PMID: 28284059. PMCID: PMC5345961.
Article69. Hirano K, Okumura T, Shimada Y, Watanabe T, Yamaguchi T, Nagata T, et al. 2015; Establishment and characterization of two novel human pancreatic carcinoma cell lines. Anticancer Res. 35:3821–3828. PMID: 26124327.70. Sinn M, Sinn BV, Treue D, Keilholz U, Damm F, Schmuck R, et al. 2020; TP53 mutations predict sensitivity to adjuvant gemcitabine in patients with pancreatic ductal adenocarcinoma: next-generation sequencing results from the CONKO-001 trial. Clin Cancer Res. 26:3732–3739. DOI: 10.1158/1078-0432.CCR-19-3034. PMID: 32234756.
Article71. Conradt L, Henrich A, Wirth M, Reichert M, Lesina M, Algül H, et al. 2013; Mdm2 inhibitors synergize with topoisomerase II inhibitors to induce p53-independent pancreatic cancer cell death. Int J Cancer. 132:2248–2257. DOI: 10.1002/ijc.27916. PMID: 23115126.
Article72. Alt JR, Bouska A, Fernandez MR, Cerny RL, Xiao H, Eischen CM. 2005; Mdm2 binds to Nbs1 at sites of DNA damage and regulates double strand break repair. J Biol Chem. 280:18771–18781. DOI: 10.1074/jbc.M413387200. PMID: 15734743.
Article73. Saison-Ridinger M, DelGiorno KE, Zhang T, Kraus A, French R, Jaquish D, et al. 2017; Reprogramming pancreatic stellate cells via p53 activation: a putative target for pancreatic cancer therapy. PLoS One. 12:e0189051. DOI: 10.1371/journal.pone.0189051. PMID: 29211796. PMCID: PMC5718507.
Article74. Lou K, Steri V, Ge AY, Hwang YC, Yogodzinski CH, Shkedi AR, et al. 2019; KRASG12C inhibition produces a driver-limited state revealing collateral dependencies. Sci Signal. 12:eaaw9450. DOI: 10.1126/scisignal.aaw9450. PMID: 31138768. PMCID: PMC6871662.
Article75. Lankes K, Hassan Z, Doffo MJ, Schneeweis C, Lier S, Öllinger R, et al. 2020; Targeting the ubiquitin-proteasome system in a pancreatic cancer subtype with hyperactive MYC. Mol Oncol. 14:3048–3064. DOI: 10.1002/1878-0261.12835. PMID: 33099868. PMCID: PMC7718946.
Article76. Alberts SR, Foster NR, Morton RF, Kugler J, Schaefer P, Wiesenfeld M, et al. 2005; PS-341 and gemcitabine in patients with metastatic pancreatic adenocarcinoma: a North Central Cancer Treatment Group (NCCTG) randomized phase II study. Ann Oncol. 16:1654–1661. DOI: 10.1093/annonc/mdi324. PMID: 16085692.
Article77. Xu J, Patel NH, Gewirtz DA. 2020; Triangular relationship between p53, autophagy, and chemotherapy resistance. Int J Mol Sci. 21:8991. DOI: 10.3390/ijms21238991. PMID: 33256191. PMCID: PMC7730978.
Article78. Wolpin BM, Rubinson DA, Wang X, Chan JA, Cleary JM, Enzinger PC, et al. 2014; Phase II and pharmacodynamic study of autophagy inhibition using hydroxychloroquine in patients with metastatic pancreatic adenocarcinoma. Oncologist. 19:637–638. DOI: 10.1634/theoncologist.2014-0086. PMID: 24821822. PMCID: PMC4041680.
Article79. Karasic TB, O'Hara MH, Loaiza-Bonilla A, Reiss KA, Teitelbaum UR, Borazanci E, et al. 2019; Effect of gemcitabine and nab-paclitaxel with or without hydroxychloroquine on patients with advanced pancreatic cancer: a phase 2 randomized clinical trial. JAMA Oncol. 5:993–998. DOI: 10.1001/jamaoncol.2019.0684. PMID: 31120501. PMCID: PMC6547080.80. Chude CI, Amaravadi RK. 2017; Targeting autophagy in cancer: update on clinical trials and novel inhibitors. Int J Mol Sci. 18:1279. DOI: 10.3390/ijms18061279. PMID: 28621712. PMCID: PMC5486101.
Article81. Balaburski GM, Hontz RD, Murphy ME. 2010; p53 and ARF: unexpected players in autophagy. Trends Cell Biol. 20:363–369. DOI: 10.1016/j.tcb.2010.02.007. PMID: 20303758. PMCID: PMC2891045.
Article82. Matsushima S, Okita N, Oku M, Nagai W, Kobayashi M, Higami Y. 2011; An Mdm2 antagonist, Nutlin-3a, induces p53-dependent and proteasome-mediated poly(ADP-ribose) polymerase1 degradation in mouse fibroblasts. Biochem Biophys Res Commun. 407:557–561. DOI: 10.1016/j.bbrc.2011.03.061. PMID: 21419099.
Article83. Kobayashi M, Ishizaki Y, Owaki M, Matsumoto Y, Kakiyama Y, Hoshino S, et al. 2020; Nutlin-3a suppresses poly (ADP-ribose) polymerase 1 by mechanisms different from conventional PARP1 suppressors in a human breast cancer cell line. Oncotarget. 11:1653–1665. DOI: 10.18632/oncotarget.27581. PMID: 32405340. PMCID: PMC7210013.
Article84. Zanjirband M, Curtin N, Edmondson RJ, Lunec J. 2017; Combination treatment with rucaparib (Rubraca) and MDM2 inhibitors, Nutlin-3 and RG7388, has synergistic and dose reduction potential in ovarian cancer. Oncotarget. 8:69779–69796. DOI: 10.18632/oncotarget.19266. PMID: 29050241. PMCID: PMC5642516.
Article85. Vena F, Jia R, Esfandiari A, Garcia-Gomez JJ, Rodriguez-Justo M, Ma J, et al. 2018; MEK inhibition leads to BRCA2 downregulation and sensitization to DNA damaging agents in pancreas and ovarian cancer models. Oncotarget. 9:11592–11603. DOI: 10.18632/oncotarget.24294. PMID: 29545922. PMCID: PMC5837749.
Article86. Deer EL, González-Hernández J, Coursen JD, Shea JE, Ngatia J, Scaife CL, et al. 2010; Phenotype and genotype of pancreatic cancer cell lines. Pancreas. 39:425–435. DOI: 10.1097/MPA.0b013e3181c15963. PMID: 20418756. PMCID: PMC2860631.
Article87. Bhutani MS, Cazacu IM, Roy-Chowdhuri S, Maitra A, Pishvaian MJ. 2020; Upfront molecular profiling of pancreatic cancer patients - an idea whose time has come. Pancreatology. 20:391–393. DOI: 10.1016/j.pan.2020.01.017. PMID: 32192904.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical Significance of p53 Protein Expression and K-ras Point Mutation in Pancreatic Cancer and Chronic Pancreatitis
- The Clinical Significance of Transforming Growth Factor (TGF) beta1, TGF beta Receptor beta II, p53 Protein and K-ras Point Mutation in Pancreatic Cancer
- p53 Gene Mutations in Bladder Cancer (I): Comparative Analysis by Immunohistochemistry and DNA Sequencing
- Mutation of p53 and p16 Tumor Suppressor Genes in Cancer and Precancerous Lesions of the Gallbladder
- p53 Gene Mutations in Transitional Cell Carcinoma of the Renal Pelvis