J Korean Med Sci.
2021 Jul;36(28):e189. 10.3346/jkms.2021.36.e189.
Association of Microbial Dysbiosis with Gallbladder Diseases Identified by Bile Microbiome Profiling
- Affiliations
-
- 1Department of Internal Medicine, Hanyang University College of Medicine, Seoul, Korea
- 2Department of Pathology, Hanyang University College of Medicine, Seoul, Korea
- 3Department of Computer Science and Engineering, College of Engineering, Hanyang University, Seoul, Korea
- 4Department of Radiology, Hanyang University College of Medicine, Seoul, Korea
- 5Department of Surgery, Hanyang University College of Medicine, Seoul, Korea
- 6Division of Liver Transplantation and Hepatobiliary Surgery, Department of Surgery, University of Ulsan College of Medicine, Asan Medical Center, Seoul, Korea
- 7Department of Laboratory Medicine, Hanyang University College of Medicine, Seoul, Korea
- KMID: 2518525
- DOI: http://doi.org/10.3346/jkms.2021.36.e189
Abstract
- Background
Cholecystitis is an important risk factor for gallbladder cancer, but the bile microbiome and its association with gallbladder disease has not been investigated fully. We aimed to analyze the bile microbiome in normal conditions, chronic cholecystitis, and gallbladder cancer, and to identify candidate bacteria that play an important role in gallbladder carcinogenesis.
Methods
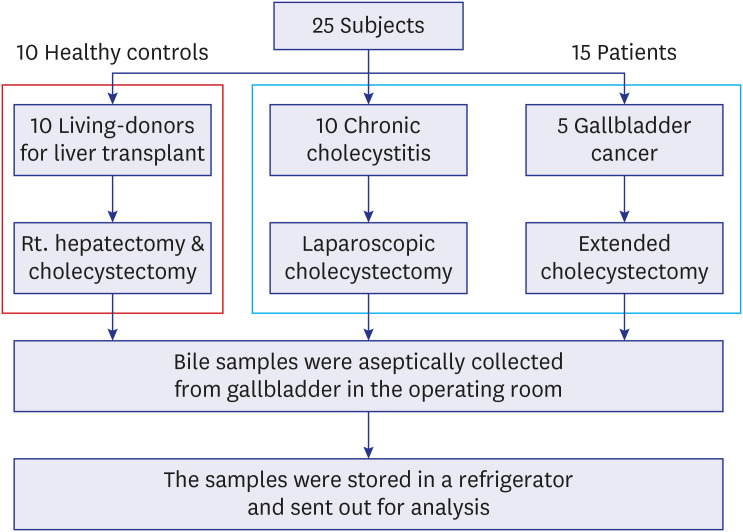
We performed metagenome sequencing on bile samples of 10 healthy individuals, 10 patients with chronic cholecystitis, and 5 patients with gallbladder cancer, and compared the clinical, radiological, and pathological characteristics of the participants.
Results
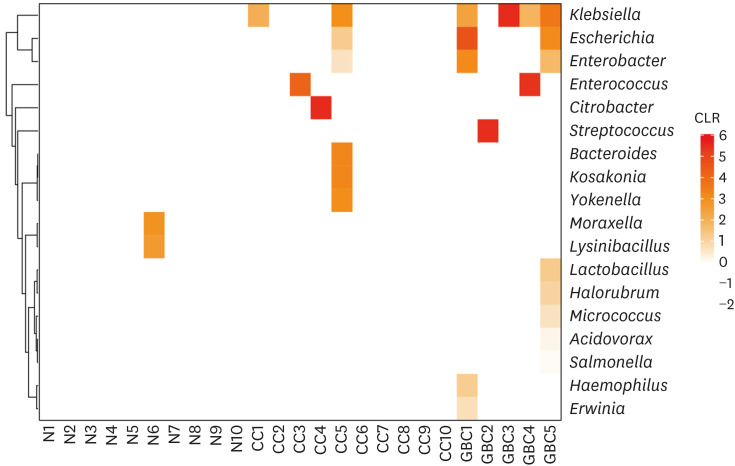
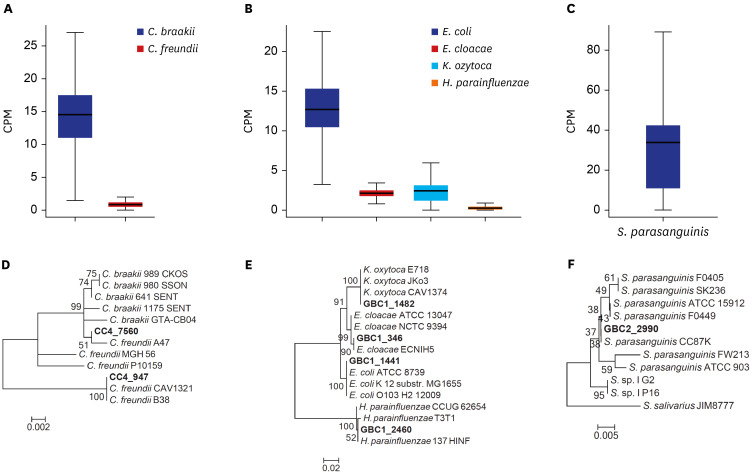
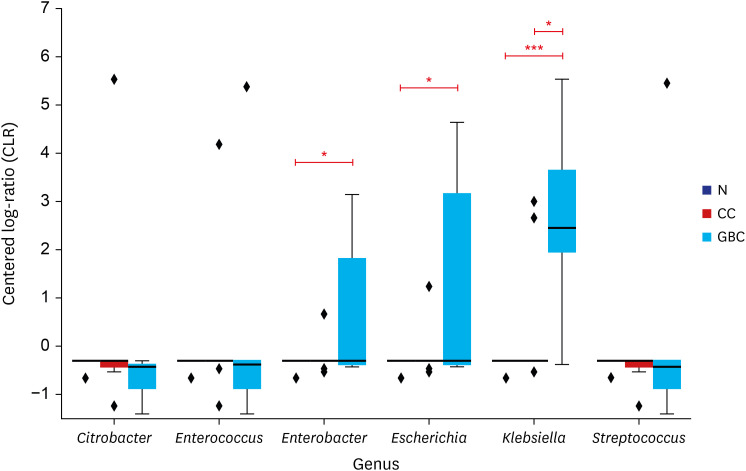
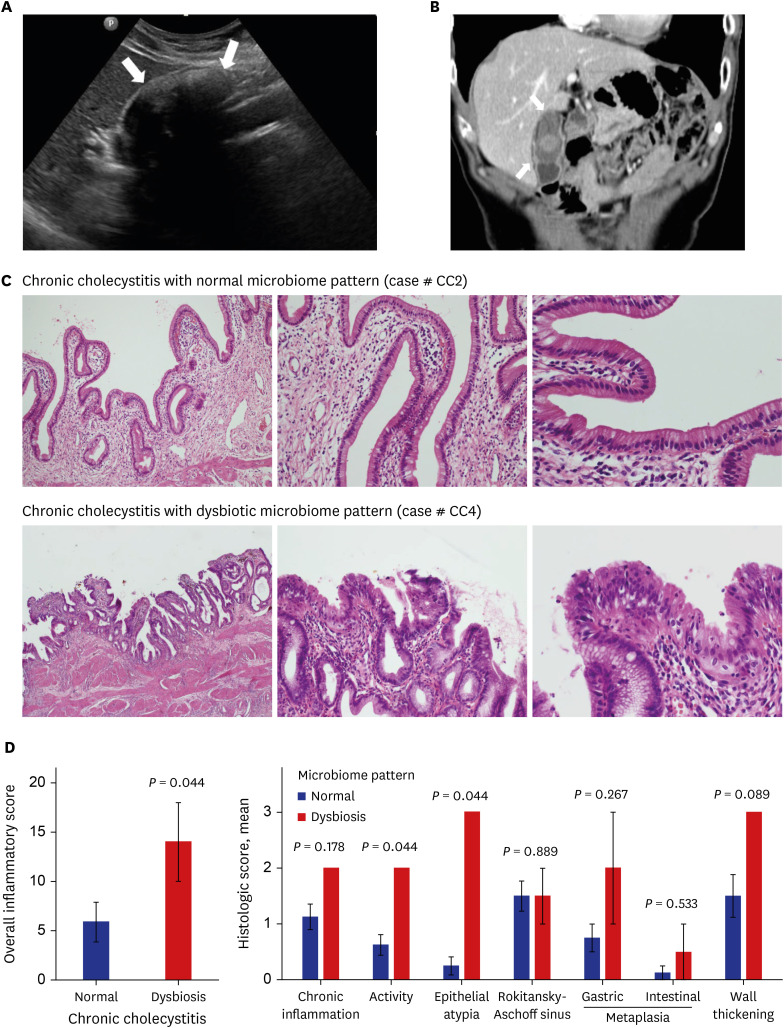
No significant bacterial signal was identified in the normal bile. The predominant dysbiotic bacteria in both chronic cholecystitis and gallbladder cancer were those belonging to the Enterobacteriaceae family. Klebsiella increased significantly in the order of normal, chronic cholecystitis, and gallbladder cancer. Patients with chronic cholecystitis and dysbiotic microbiome patterns had larger gallstones and showed marked epithelial atypia, which are considered as precancerous conditions.
Conclusion
We investigated the bile microbiome in normal, chronic cholecystitis, and gallbladder cancer. We suggest possible roles of Enterobacteriaceae, including Klebsiella, in gallbladder carcinogenesis. Our findings reveal a possible link between a dysbiotic bile microbiome and the development of chronic calculous cholecystitis and gallbladder cancer.
Figure
Reference
-
1. Ferlay JE, Lam F, Colombet M, Mery L, Piñeros M, Znaor A, et al. Global cancer observatory: cancer today. Updated 2020. Accessed March 20, 2020. https://gco.iarc.fr/today.2. Yamaguchi K, Chijiiwa K, Saiki S, Nishihara K, Takashima M, Kawakami K, et al. Retrospective analysis of 70 operations for gallbladder carcinoma. Br J Surg. 1997; 84(2):200–204. PMID: 9052434.
Article3. Boutros C, Gary M, Baldwin K, Somasundar P. Gallbladder cancer: past, present and an uncertain future. Surg Oncol. 2012; 21(4):e183–e191. PMID: 23025910.
Article4. Nath G, Gulati AK, Shukla VK. Role of bacteria in carcinogenesis, with special reference to carcinoma of the gallbladder. World J Gastroenterol. 2010; 16(43):5395–5404. PMID: 21086555.
Article5. Nath G, Singh H, Shukla VK. Chronic typhoid carriage and carcinoma of the gallbladder. Eur J Cancer Prev. 1997; 6(6):557–559. PMID: 9496458.
Article6. de Martel C, Plummer M, Parsonnet J, van Doorn LJ, Franceschi S. Helicobacter species in cancers of the gallbladder and extrahepatic biliary tract. Br J Cancer. 2009; 100(1):194–199. PMID: 19034278.
Article7. Scanu T, Spaapen RM, Bakker JM, Pratap CB, Wu LE, Hofland I, et al. Salmonella manipulation of host signaling pathways provokes cellular transformation associated with gallbladder carcinoma. Cell Host Microbe. 2015; 17(6):763–774. PMID: 26028364.
Article8. Gonzalez-Escobedo G, La Perle KM, Gunn JS. Histopathological analysis of Salmonella chronic carriage in the mouse hepatopancreatobiliary system. PLoS One. 2013; 8(12):e84058. PMID: 24349565.
Article9. Abreu MT, Peek RM Jr. Gastrointestinal malignancy and the microbiome. Gastroenterology. 2014; 146(6):1534–1546.e3. PMID: 24406471.
Article10. Lykkegaard Nielsen M, Asnaes S, Justesen T. Susceptibility of the liver and biliary tract to anaerobic infection in extrahepatic biliary tract obstruction. III. Possible synergistic effect between anaerobic and aerobic bacteria. An experimental study in rabbits. Scand J Gastroenterol. 1976; 11(3):263–272. PMID: 775613.11. Kim B, Park JS, Bae J, Hwang N. Bile microbiota in patients with pigment common bile duct stones. J Korean Med Sci. 2021; 36(15):e94. PMID: 33876584.
Article12. Begley M, Gahan CG, Hill C. The interaction between bacteria and bile. FEMS Microbiol Rev. 2005; 29(4):625–651. PMID: 16102595.
Article13. Molinero N, Ruiz L, Milani C, Gutiérrez-Díaz I, Sánchez B, Mangifesta M, et al. The human gallbladder microbiome is related to the physiological state and the biliary metabolic profile. Microbiome. 2019; 7(1):100. PMID: 31272480.
Article14. Furlan A, Ferris JV, Hosseinzadeh K, Borhani AA. Gallbladder carcinoma update: multimodality imaging evaluation, staging, and treatment options. AJR Am J Roentgenol. 2008; 191(5):1440–1447. PMID: 18941083.
Article15. Kim SJ, Lee JM, Lee JY, Kim SH, Han JK, Choi BI, et al. Analysis of enhancement pattern of flat gallbladder wall thickening on MDCT to differentiate gallbladder cancer from cholecystitis. AJR Am J Roentgenol. 2008; 191(3):765–771. PMID: 18716107.
Article16. Yun EJ, Cho SG, Park S, Park SW, Kim WH, Kim HJ, et al. Gallbladder carcinoma and chronic cholecystitis: differentiation with two-phase spiral CT. Abdom Imaging. 2004; 29(1):102–108. PMID: 15160762.
Article17. Barcia JJ. Histologic analysis of chronic inflammatory patterns in the gallbladder: diagnostic criteria for reporting cholecystitis. Ann Diagn Pathol. 2003; 7(3):147–153. PMID: 12808565.
Article18. Mago\xc4\x8d T, Salzberg SL. FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics. 2011; 27(21):2957–2963. PMID: 21903629.
Article19. Fu L, Niu B, Zhu Z, Wu S, Li W. CD-HIT: accelerated for clustering the next-generation sequencing data. Bioinformatics. 2012; 28(23):3150–3152. PMID: 23060610.
Article20. Wang Q, Garrity GM, Tiedje JM, Cole JR. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol. 2007; 73(16):5261–5267. PMID: 17586664.
Article21. Salter SJ, Cox MJ, Turek EM, Calus ST, Cookson WO, Moffatt MF, et al. Reagent and laboratory contamination can critically impact sequence-based microbiome analyses. BMC Biol. 2014; 12(1):87. PMID: 25387460.
Article22. Karstens L, Asquith M, Davin S, Fair D, Gregory WT, Wolfe AJ, et al. Controlling for contaminants in low-biomass 16S rRNA gene sequencing experiments. mSystems. 2019; 4(4):e00290-19.
Article23. Li D, Liu CM, Luo R, Sadakane K, Lam TW. MEGAHIT: an ultra-fast single-node solution for large and complex metagenomics assembly via succinct de Bruijn graph. Bioinformatics. 2015; 31(10):1674–1676. PMID: 25609793.
Article24. Rho M, Tang H, Ye Y. FragGeneScan: predicting genes in short and error-prone reads. Nucleic Acids Res. 2010; 38(20):e191. PMID: 20805240.
Article25. Langmead B, Salzberg SL. Fast gapped-read alignment with Bowtie 2. Nat Methods. 2012; 9(4):357–359. PMID: 22388286.
Article26. Mistry J, Finn RD, Eddy SR, Bateman A, Punta M. Challenges in homology search: HMMER3 and convergent evolution of coiled-coil regions. Nucleic Acids Res. 2013; 41(12):e121. PMID: 23598997.
Article27. Edgar RC. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004; 32(5):1792–1797. PMID: 15034147.
Article28. Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol. 2013; 30(12):2725–2729. PMID: 24132122.
Article29. Jiménez E, Sánchez B, Farina A, Margolles A, Rodríguez JM. Characterization of the bile and gall bladder microbiota of healthy pigs. MicrobiologyOpen. 2014; 3(6):937–949. PMID: 25336405.
Article30. Octavia S, Lan R. The family Enterobacteriaceae. In : Rosenberg E, DeLong EF, Lory S, Stackebrandt E, Thompson F, editors. The Prokaryotes: Gammaproteobacteria. Berlin, Heidelberg, Germany: Springer Berlin Heidelberg;2014. p. 225–286.31. Brower-Sinning R, Zhong D, Good M, Firek B, Baker R, Sodhi CP, et al. Mucosa-associated bacterial diversity in necrotizing enterocolitis. PLoS One. 2014; 9(9):e105046. PMID: 25203729.
Article32. Samonis G, Karageorgopoulos DE, Kofteridis DP, Matthaiou DK, Sidiropoulou V, Maraki S, et al. Citrobacter infections in a general hospital: characteristics and outcomes. Eur J Clin Microbiol Infect Dis. 2009; 28(1):61–68. PMID: 18682995.
Article33. Drelichman V, Band JD. Bacteremias due to Citrobacter diversus and Citrobacter freundii. Incidence, risk factors, and clinical outcome. Arch Intern Med. 1985; 145(10):1808–1810. PMID: 3899035.
Article34. Apostolov E, Al-Soud WA, Nilsson I, Kornilovska I, Usenko V, Lyzogubov V, et al. Helicobacter pylori and other Helicobacter species in gallbladder and liver of patients with chronic cholecystitis detected by immunological and molecular methods. Scand J Gastroenterol. 2005; 40(1):96–102. PMID: 15841721.35. Fox JG, Dewhirst FE, Shen Z, Feng Y, Taylor NS, Paster BJ, et al. Hepatic Helicobacter species identified in bile and gallbladder tissue from Chileans with chronic cholecystitis. Gastroenterology. 1998; 114(4):755–763. PMID: 9516396.
Article36. Dutta U, Garg PK, Kumar R, Tandon RK. Typhoid carriers among patients with gallstones are at increased risk for carcinoma of the gallbladder. Am J Gastroenterol. 2000; 95(3):784–787. PMID: 10710075.
Article37. Tsuchiya Y, Loza E, Villa-Gomez G, Trujillo CC, Baez S, Asai T, et al. Metagenomics of microbial communities in gallbladder bile from patients with gallbladder cancer or cholelithiasis. Asian Pac J Cancer Prev. 2018; 19(4):961–967. PMID: 29693356.38. Kumar S, Kumar S, Kumar S. Infection as a risk factor for gallbladder cancer. J Surg Oncol. 2006; 93(8):633–639. PMID: 16724347.
Article39. O’Brien B, Dyer B, DeLuca J, Richmond BK. Streptococcus bovis bacteremia as the initial presentation of carcinoma of the gallbladder. W V Med J. 2014; 110(2):32–33.40. Alves JR, Silva RC, Guerra SC, Freitas TT, Souza DL, Amico EC. Microbiological analysis of bile in patients with benign and malignant biliopancreatic diseases and its consequences. Arq Gastroenterol. 2016; 53(3):156–162. PMID: 27438420.
Article41. Bottone EJ, Zhang DY. Haemophilus parainfluenzae biliary tract infection: rationale for an ascending route of infection from the gastrointestinal tract. J Clin Microbiol. 1995; 33(11):3042–3043. PMID: 8576372.
Article42. Alvarez M, Potel C, Rey L, Rodriguez-Sousa T, Otero I. Maximiliano Alvarez, Carmen Potel. Biliary tract infection caused by Haemophilus parainfluenzae. Scand J Infect Dis. 1999; 31(2):212–213. PMID: 10447339.43. Hazrah P, Oahn KT, Tewari M, Pandey AK, Kumar K, Mohapatra TM, et al. The frequency of live bacteria in gallstones. HPB (Oxford). 2004; 6(1):28–32. PMID: 18333042.
Article44. Csendes A, Becerra M, Burdiles P, Demian I, Bancalari K, Csendes P. Bacteriological studies of bile from the gallbladder in patients with carcinoma of the gallbladder, cholelithiasis, common bile duct stones and no gallstones disease. Eur J Surg. 1994; 160(6-7):363–367. PMID: 7948355.45. Melzer M, Toner R, Lacey S, Bettany E, Rait G. Biliary tract infection and bacteraemia: presentation, structural abnormalities, causative organisms and clinical outcomes. Postgrad Med J. 2007; 83(986):773–776. PMID: 18057178.
Article46. Lin RD, Hsueh PR, Chang SC, Chen YC, Hsieh WC, Luh KT. Bacteremia due to Klebsiella oxytoca: clinical features of patients and antimicrobial susceptibilities of the isolates. Clin Infect Dis. 1997; 24(6):1217–1222. PMID: 9195086.47. Katyal A, Bala K, Bansal A, Chaudhary U. Clinico-microbiological analysis of bactibilia isolates in patients of cholecystectomy. Int J Res Med Sci. 2017; 5(9):6.
Article48. Pötgens SA, Brossel H, Sboarina M, Catry E, Cani PD, Neyrinck AM, et al. Klebsiella oxytoca expands in cancer cachexia and acts as a gut pathobiont contributing to intestinal dysfunction. Sci Rep. 2018; 8(1):12321. PMID: 30120320.
Article49. Roa I, Araya JC, Villaseca M, Roa J, de Aretxabala X, Ibacache G. Gallbladder cancer in a high risk area: morphological features and spread patterns. Hepatogastroenterology. 1999; 46(27):1540–1546. PMID: 10430291.50. Diehl AK. Gallstone size and the risk of gallbladder cancer. JAMA. 1983; 250(17):2323–2326. PMID: 6632129.
Article51. Shaffer EA. Gallbladder cancer: the basics. Gastroenterol Hepatol (N Y). 2008; 4(10):737–741. PMID: 21960896.52. Hundal R, Shaffer EA. Gallbladder cancer: epidemiology and outcome. Clin Epidemiol. 2014; 6:99–109. PMID: 24634588.53. Zen Y, Aishima S, Ajioka Y, Haratake J, Kage M, Kondo F, et al. Proposal of histological criteria for intraepithelial atypical/proliferative biliary epithelial lesions of the bile duct in hepatolithiasis with respect to cholangiocarcinoma: preliminary report based on interobserver agreement. Pathol Int. 2005; 55(4):180–188. PMID: 15826244.
Article54. Lee KB. Histologic and molecular pathogenesis of gallbladder cancer. Korean J Pancreas Biliary Tract. 2018; 23(1):1–6.
Article