Korean J Gastroenterol.
2021 Jan;77(1):12-21. 10.4166/kjg.2020.119.
Therapeutic Potential of Escherichia coli Nissle 1917 in Clinically Remission-attained Ulcerative Colitis Patients: A Hospital-based Cohort Study
- Affiliations
-
- 1Department of Internal Medicine, Kosin University College of Medicine, Busan, Korea
- KMID: 2510771
- DOI: http://doi.org/10.4166/kjg.2020.119
Abstract
- Background/Aims
Escherichia coli Nissle 1917 (EcN) alone therapy is as effective as mesalamine in inducing and maintaining remission in ulcerative colitis (UC). The efficacy and safety of EcN in combination with standard therapies have not been studied. This study examined the changes in the inflammation markers and symptoms following the additional administration of EcN to patients showing the clinical remission of UC.
Methods
UC patients who received EcN after being in clinical remission for more than 3 months at Kosin University Gospel Hospital between 2013 and 2018 were evaluated through the retrospective medical-record-based review. The partial Mayo score, fecal calprotectin (FC), BMI, hemoglobin, serum cholesterol, serum albumin levels, and the safety profiles were examined at 3rd and 6th months after initiating EcN.
Results
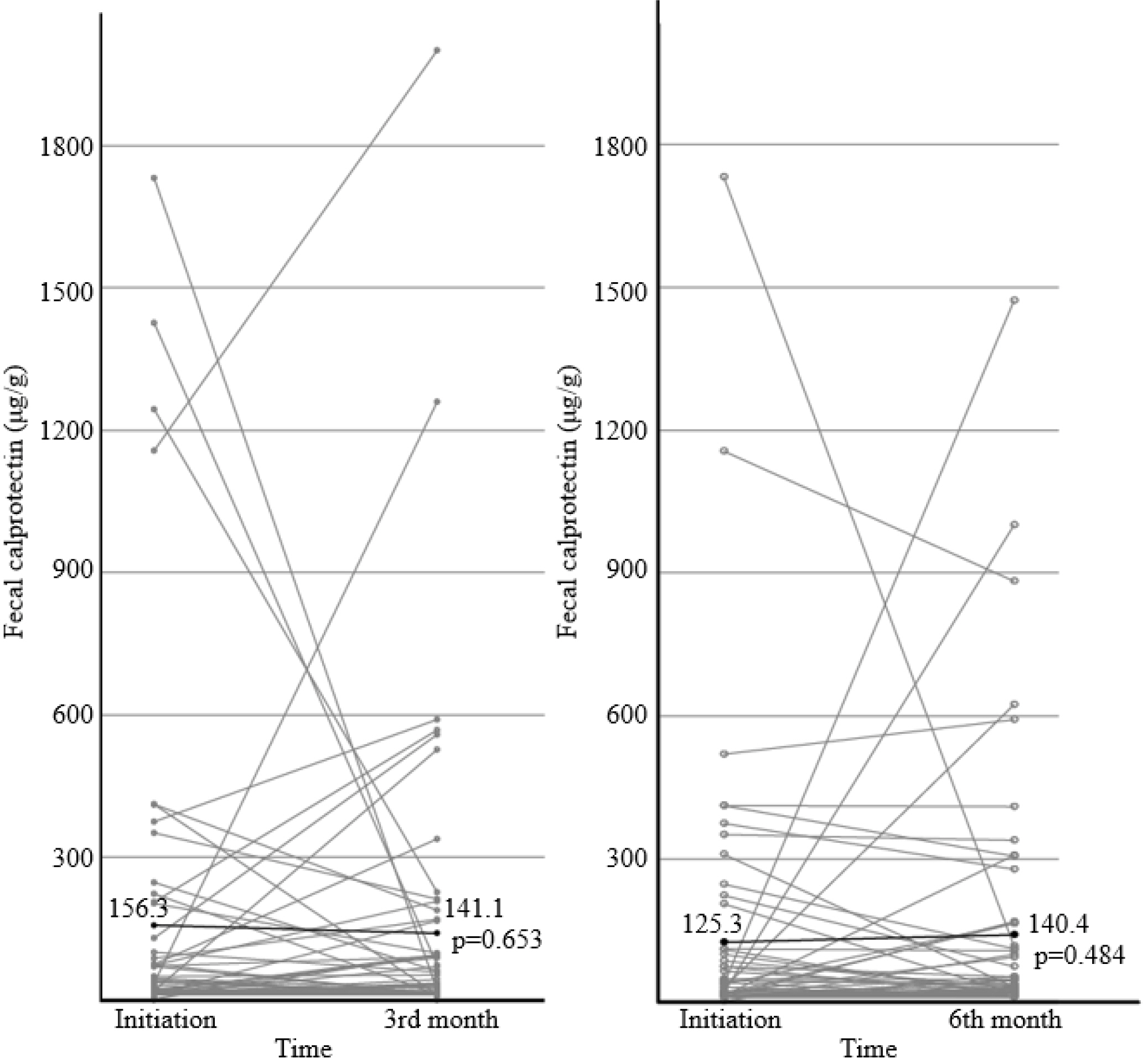
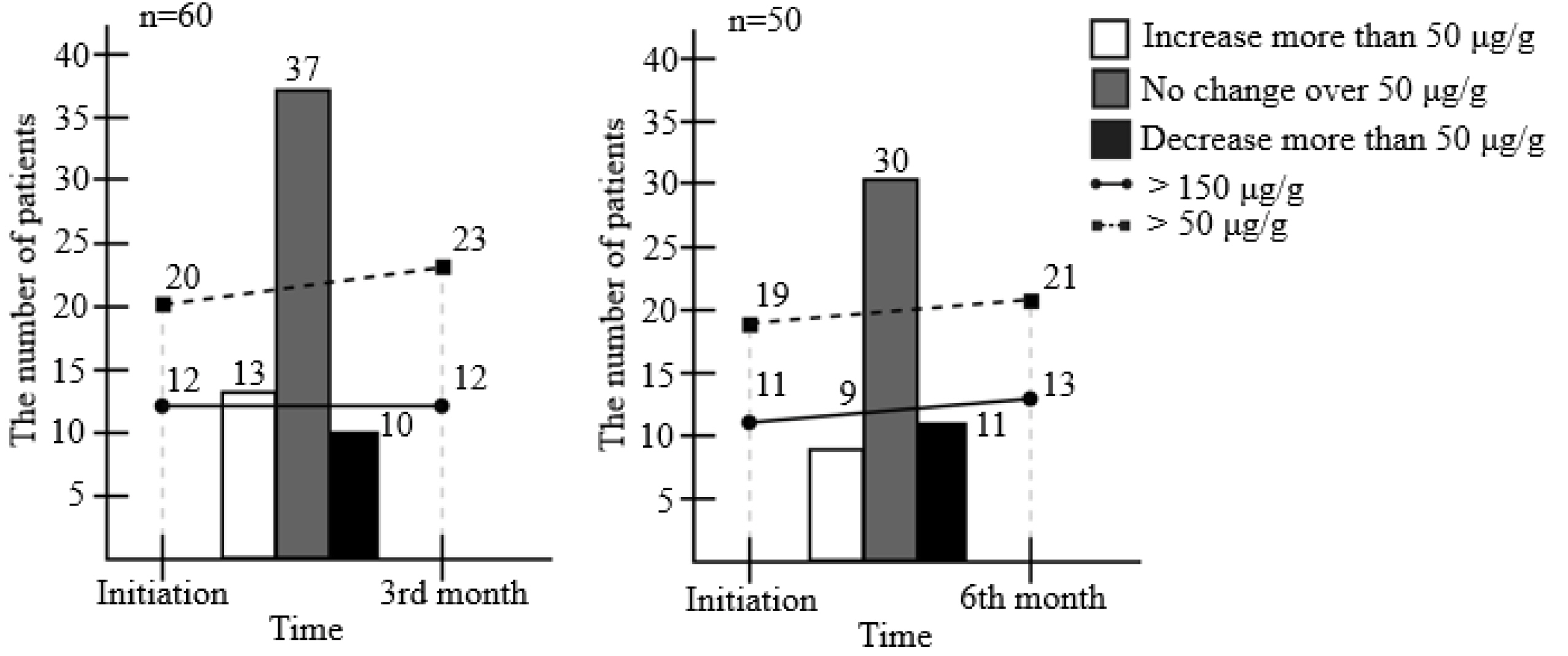
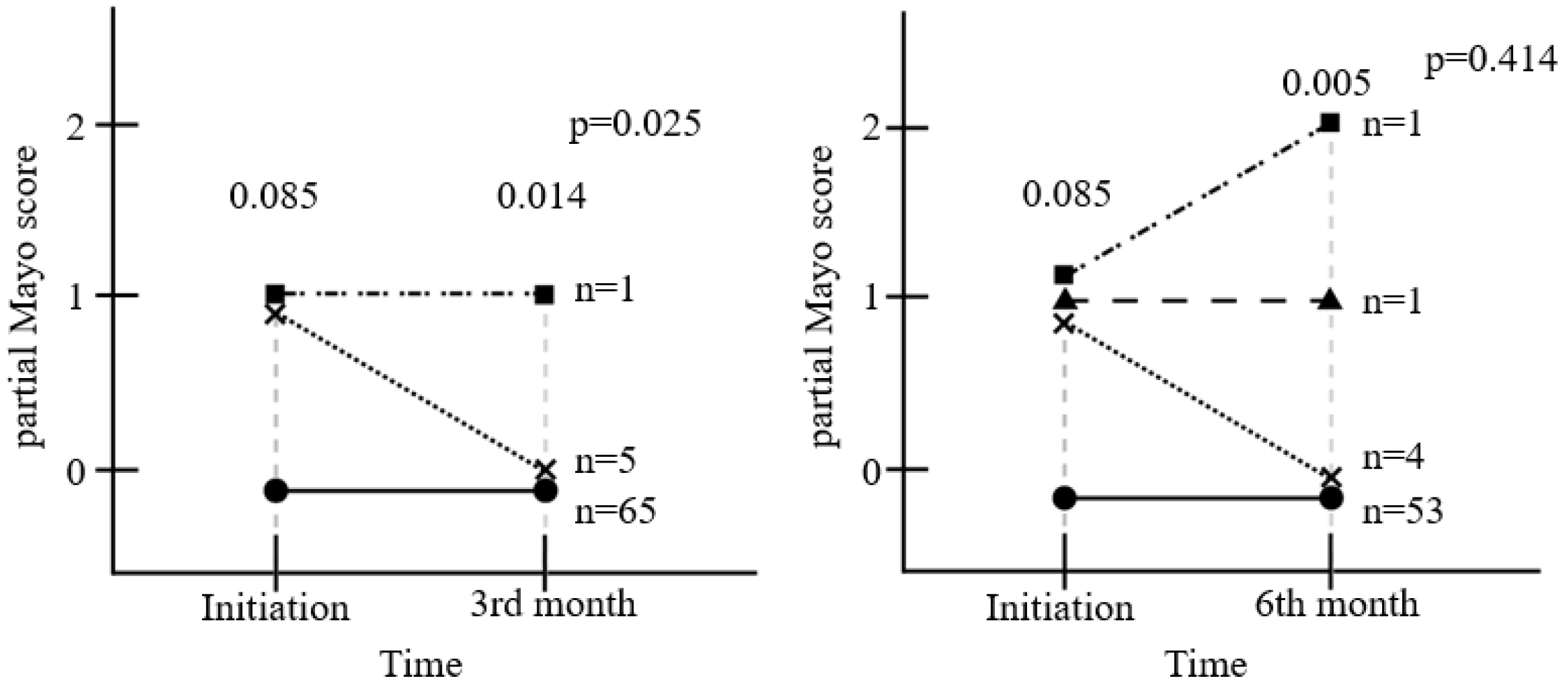
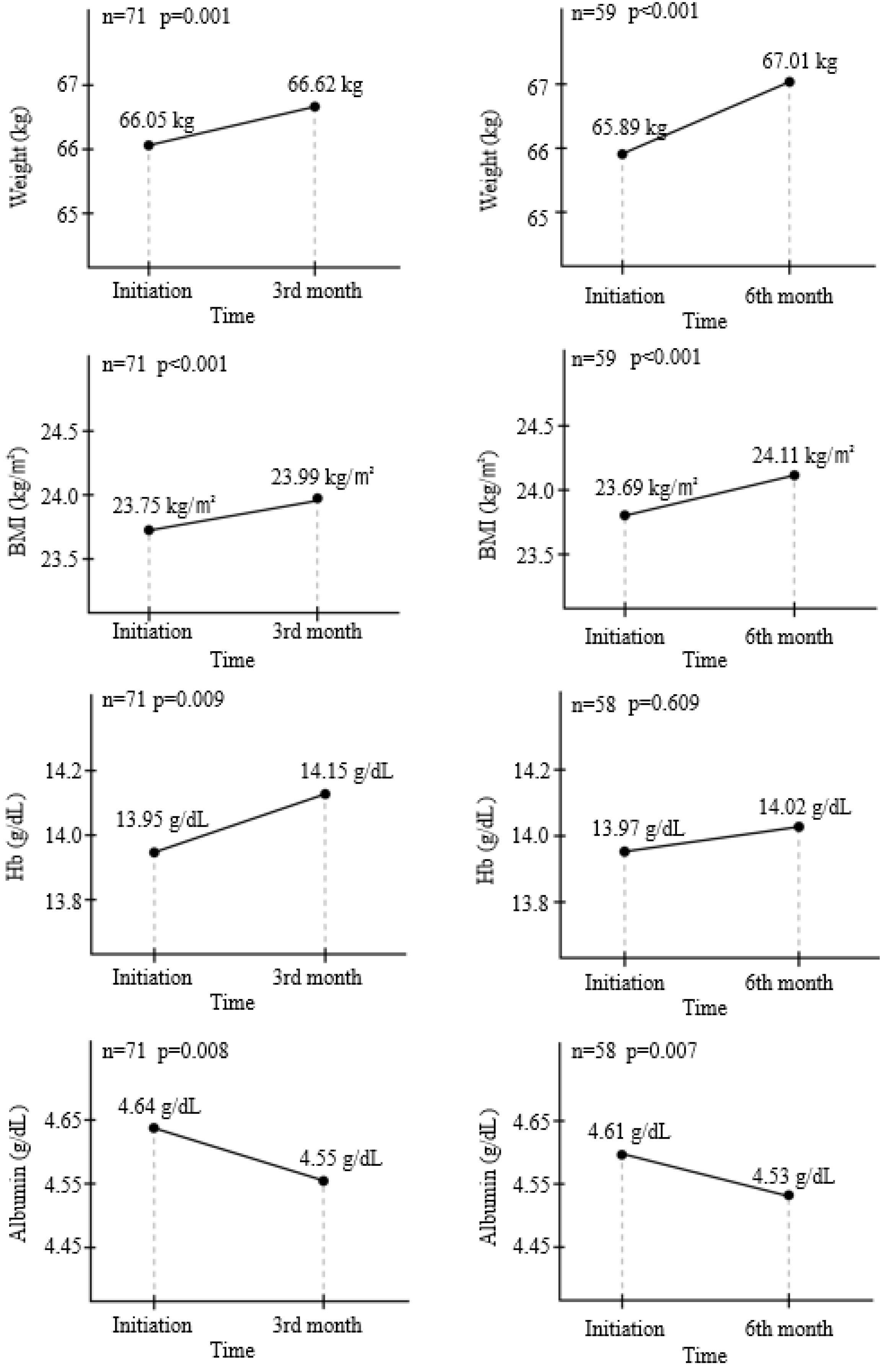
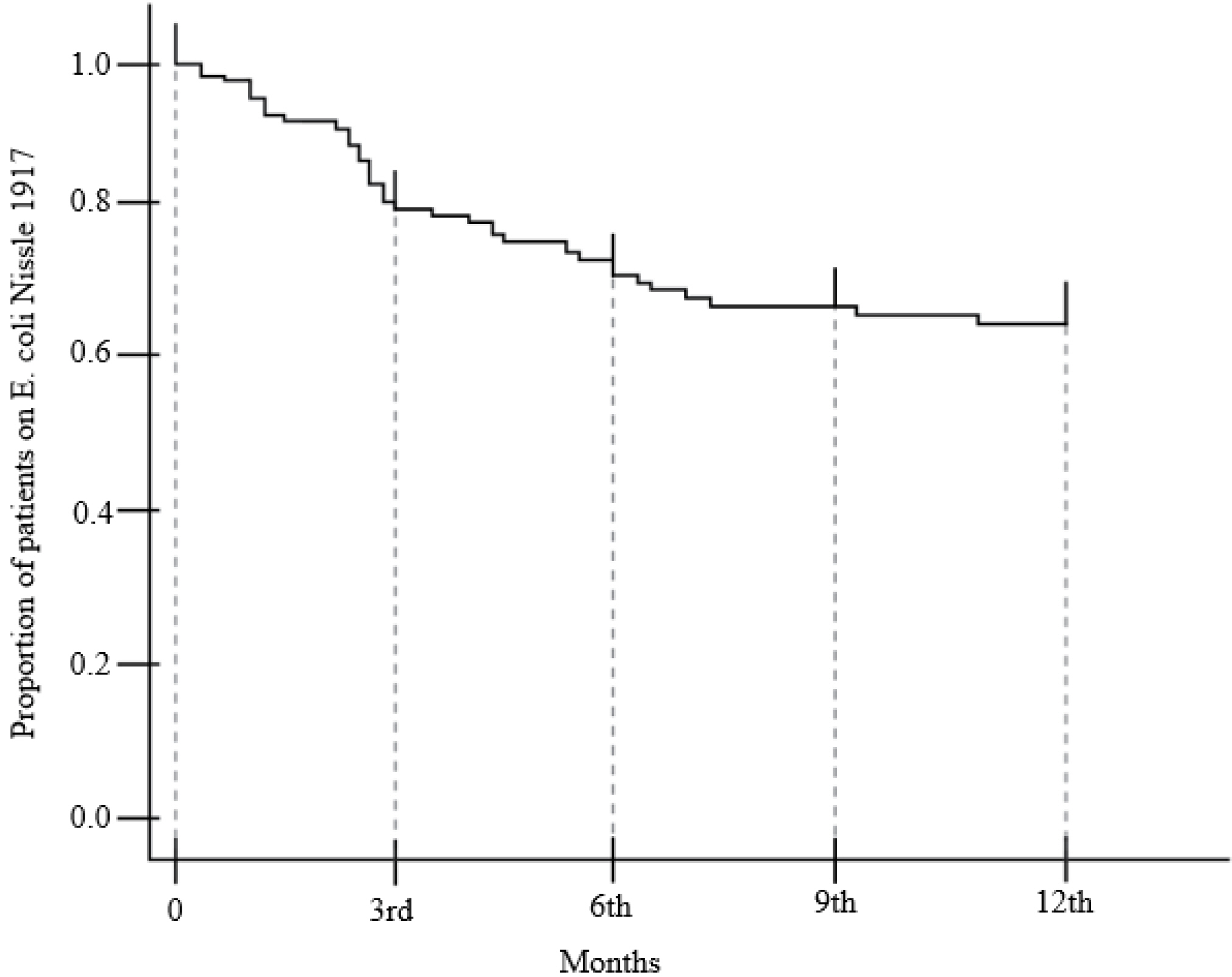
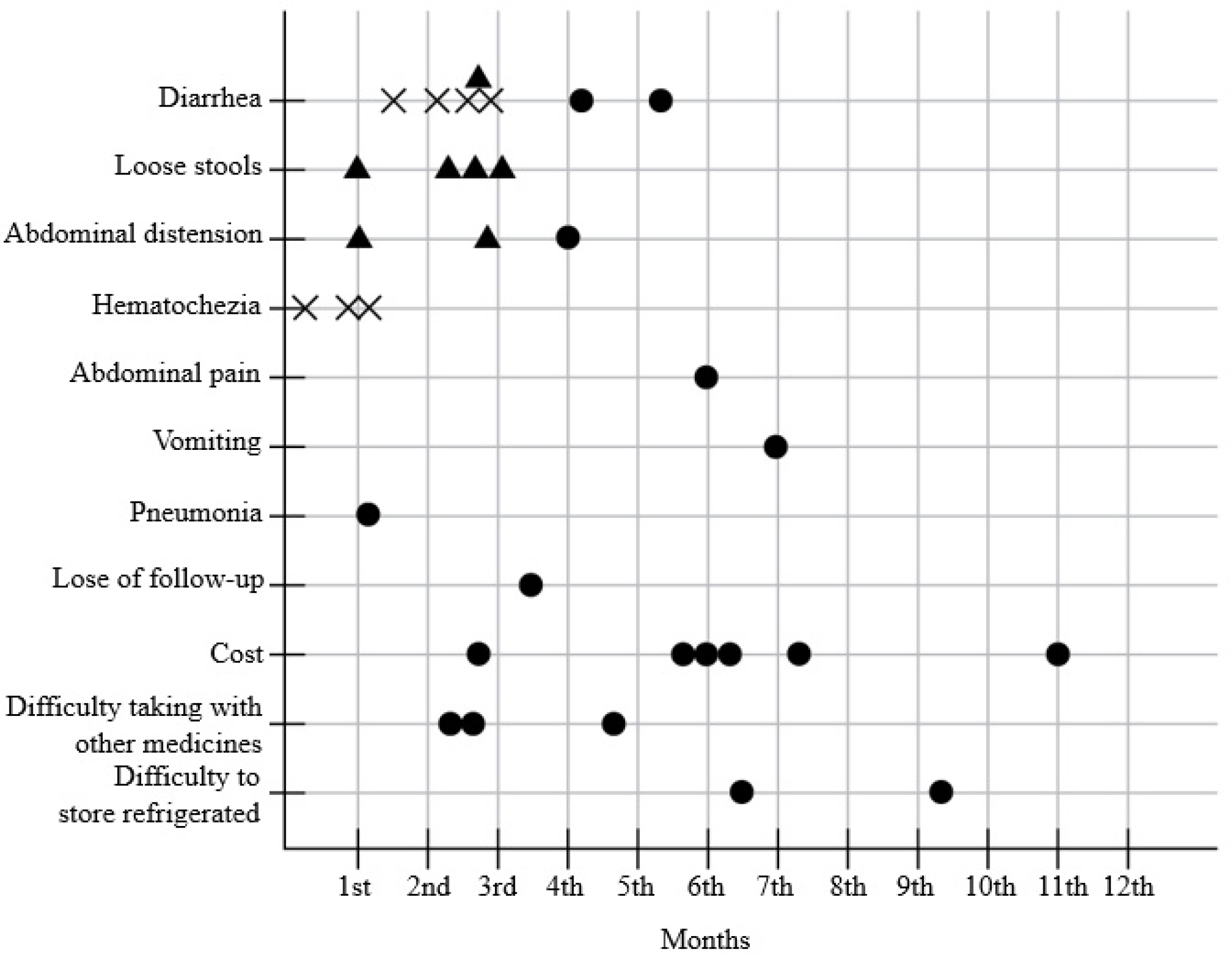
Ninety-four patients were included. After 3 months of treatment, there was no significant change in FC (156.3 μg/g to 141.1 μg/g) (p=0.653). On the other hand, partial Mayo score decreased significantly from 0.085 to 0.014 (p=0.025), and the bodyweight (p=0.001), BMI (p<0.001), hemoglobin (p=0.009), and cholesterol level increased (p=0.148). One patient (1.1%) experienced a serious adverse event with UC flare-up, and 14 patients (14.9%) discontinued EcN due to adverse events; all developed within 3 months.
Conclusions
Additional administration of EcN to clinically remission-attained UC patients may improve the UC symptoms without changing the FC levels. EcN-associated adverse events develop within the early few weeks.
Keyword
Figure
Reference
-
1. Harbord M, Eliakim R, Bettenworth D, et al. 2017; Third European evidence-based consensus on diagnosis and management of ulcerative colitis. Part 2: current management. J Crohns Colitis. 11:769–784. DOI: 10.1093/ecco-jcc/jjx009. PMID: 28513805.
Article2. Panes J, O'Connor M, Peyrin-Biroulet L, Irving P, Petersson J, Colombel JF. 2014; Improving quality of care in inflammatory bowel disease: what changes can be made today? J Crohns Colitis. 8:919–926. DOI: 10.1016/j.crohns.2014.02.022. PMID: 24713174.
Article3. Rubin DT, Ananthakrishnan AN, Siegel CA, Sauer BG, Long MD. 2019; ACG clinical guideline: ulcerative colitis in adults. Am J Gastro- enterol. 114:384–413. DOI: 10.14309/ajg.0000000000000152. PMID: 30840605.
Article4. Choi CH, Moon W, Kim YS, et al. 2017; Second Korean guideline for the management of ulcerative colitis. Korean J Gastroenterol. 69:1–28. DOI: 10.4166/kjg.2017.69.1.1. PMID: 28135789.
Article5. Torres J, Burisch J, Riddle M, Dubinsky M, Colombel JF. 2016; Preclinical disease and preventive strategies in IBD: perspectives, challenges and opportunities. Gut. 65:1061–1069. DOI: 10.1136/gutjnl-2016-311785. PMID: 27196600.
Article6. Morgan XC, Tickle TL, Sokol H, et al. 2012; Dysfunction of the intestinal microbiome in inflammatory bowel disease and treatment. Genome Biol. 13:R79. DOI: 10.1186/gb-2012-13-9-r79. PMID: 23013615. PMCID: PMC3506950.
Article7. Lane ER, Zisman TL, Suskind DL. 2017; The microbiota in inflammatory bowel disease: current and therapeutic insights. J Inflamm Res. 10:63–73. DOI: 10.2147/JIR.S116088. PMID: 28652796. PMCID: PMC5473501.
Article8. Frank DN, Robertson CE, Hamm CM, et al. 2011; Disease phenotype and genotype are associated with shifts in intestinal-associated microbiota in inflammatory bowel diseases. Inflamm Bowel Dis. 17:179–184. DOI: 10.1002/ibd.21339. PMID: 20839241. PMCID: PMC3834564.
Article9. Matthes H, Krummenerl T, Giensch M, Wolff C, Schulze J. 2010; Clinical trial: probiotic treatment of acute distal ulcerative colitis with rectally administered Escherichia coli Nissle 1917 (EcN). BMC Complement Altern Med. 10:13. DOI: 10.1186/1472-6882-10-13. PMID: 20398311. PMCID: PMC2861635.
Article10. Kruis W, Schutz E, Fric P, Fixa B, Judmaier G, Stolte M. 1997; Double-blind comparison of an oral Escherichia coli preparation and mesalazine in maintaining remission of ulcerative colitis. Aliment Pharmacol Ther. 11:853–858. DOI: 10.1046/j.1365-2036.1997.00225.x. PMID: 9354192.11. Losurdo G, Iannone A, Contaldo A, Ierardi E, Di Leo A, Principi M. 2015; Escherichia coli Nissle 1917 in Ulcerative Colitis Treatment: Systematic Review and Meta-analysis. J Gastrointestin Liver Dis. 24:499–505. DOI: 10.15403/jgld.2014.1121.244.ecn. PMID: 26697577.12. Kruis W, Fric P, Pokrotnieks J, et al. 2004; Maintaining remission of ulcerative colitis with the probiotic Escherichia coli Nissle 1917 is as effective as with standard mesalazine. Gut. 53:1617–1623. DOI: 10.1136/gut.2003.037747. PMID: 15479682. PMCID: PMC1774300.13. Rembacken BJ, Snelling AM, Hawkey PM, Chalmers DM, Axon ATR. 1999; Non-pathogenic Escherichia coli versus mesalazine for the treatment of ulcerative colitis: a randomised trial. The Lancet. 354:635–639. DOI: 10.1016/S0140-6736(98)06343-0. PMID: 10466665.14. Hajela N, Ramakrishna BS, Nair GB, Abraham P, Gopalan S, Ganguly NK. 2015; Gut microbiome, gut function, and probiotics: Implications for health. Indian J Gastroenterol. 34:93–107. DOI: 10.1007/s12664-015-0547-6. PMID: 25917520.
Article15. Scaldaferri F, Gerardi V, Mangiola F, et al. 2016; Role and mechanisms of action of Escherichia coli Nissle 1917 in the maintenance of remission in ulcerative colitis patients: An update. World J Gastroenterol. 22:5505–5511. DOI: 10.3748/wjg.v22.i24.5505. PMID: 27350728. PMCID: PMC4917610.16. Sheehan D, Moran C, Shanahan F. 2015; The microbiota in inflammatory bowel disease. J Gastroenterol. 50:495–507. DOI: 10.1007/s00535-015-1064-1. PMID: 25808229.
Article17. Ganji-Arjenaki M, Rafieian-Kopaei M. 2018; Probiotics are a good choice in remission of inflammatory bowel diseases: A meta analysis and systematic review. J Cell Physiol. 233:2091–2103. DOI: 10.1002/jcp.25911. PMID: 28294322.
Article18. Lewis JD, Chuai S, Nessel L, Lichtenstein GR, Aberra FN, Ellenberg JH. 2008; Use of the noninvasive components of the Mayo score to assess clinical response in ulcerative colitis. Inflamm Bowel Dis. 14:1660–1666. DOI: 10.1002/ibd.20520. PMID: 18623174. PMCID: PMC2597552.
Article19. Mao R, Xiao YL, Gao X, et al. 2012; Fecal calprotectin in predicting relapse of inflammatory bowel diseases: a meta-analysis of prospective studies. Inflamm Bowel Dis. 18:1894–1899. DOI: 10.1002/ibd.22861. PMID: 22238138.
Article20. Maaser C, Sturm A, Vavricka SR, et al. 2019; ECCO-ESGAR Guideline for Diagnostic Assessment in IBD Part 1: Initial diagnosis, monitoring of known IBD, detection of complications. J Crohns Colitis. 13:144–164. DOI: 10.1093/ecco-jcc/jjy113. PMID: 30137275.
Article21. Schoepfer AM, Beglinger C, Straumann A, Trummler M, Renzulli P, Seibold F. 2009; Ulcerative colitis: correlation of the Rachmilewitz endoscopic activity index with fecal calprotectin, clinical activity, C-reactive protein, and blood leukocytes. Inflamm Bowel Dis. 15:1851–1858. DOI: 10.1002/ibd.20986. PMID: 19462421.22. Gao K, Pi Y, Peng Y, Mu CL, Zhu WY. 2018; Time-course responses of ileal and fecal microbiota and metabolite profiles to antibiotics in cannulated pigs. Appl Microbiol Biotechnol. 102:2289–2299. DOI: 10.1007/s00253-018-8774-2. PMID: 29362824.
Article23. Geerling BJ, Badart-Smook A, Stockbrugger RW, Brummer RJ. 2000; Comprehensive nutritional status in recently diagnosed patients with inflammatory bowel disease compared with population controls. Eur J Clin Nutr. 54:514–521. DOI: 10.1038/sj.ejcn.1601049. PMID: 10878655.
Article24. Mijac DD, Jankovic GL, Jorga J, Krstic MN. 2010; Nutritional status in patients with active inflammatory bowel disease: prevalence of malnutrition and methods for routine nutritional assessment. Eur J Intern Med. 21:315–319. DOI: 10.1016/j.ejim.2010.04.012. PMID: 20603043.25. Chaudhari AS, Raghuvanshi R, Kumar GN. 2017; Genetically engineered Escherichia coli Nissle 1917 synbiotic counters fructose-induced metabolic syndrome and iron deficiency. Appl Microbiol Biotechnol. 101:4713–4723. DOI: 10.1007/s00253-017-8207-7. PMID: 28283693.26. Ukena SN, Singh A, Dringenberg U, et al. 2007; Probiotic Escherichia coli Nissle 1917 inhibits leaky gut by enhancing mucosal integrity. PLoS One. 2:e1308. DOI: 10.1371/journal.pone.0001308. PMID: 18074031. PMCID: PMC2110898.27. Farraye FA, Melmed GY, Lichtenstein GR, Kane SV. 2017; ACG Clinical Guideline: Preventive Care in Inflammatory Bowel Disease. Am J Gastroenterol. 112:241–258. DOI: 10.1038/ajg.2016.537. PMID: 28071656.
Article28. Valentini L, Schulzke JD. 2011; Mundane, yet challenging: the assessment of malnutrition in inflammatory bowel disease. Eur J Intern Med. 22:13–15. DOI: 10.1016/j.ejim.2010.07.021. PMID: 21238886.
Article29. Di Paolo MC, Paoluzi OA, Pica R, et al. 2001; Sulphasalazine and 5-aminosalicylic acid in long-term treatment of ulcerative colitis: report on tolerance and side-effects. Dig Liver Dis. 33:563–569. DOI: 10.1016/S1590-8658(01)80108-0. PMID: 11816545.
Article30. Fiorentini MT, Fracchia M, Galatola G, Barlotta A, de la Pierre M. 1990; Acute pancreatitis during oral 5-aminosalicylic acid therapy. Dig Dis Sci. 35:1180–1182. DOI: 10.1007/BF01537594. PMID: 2390934.
Article31. de Jong DJ, Goullet M, Naber TH. 2004; Side effects of azathioprine in patients with Crohn's disease. Eur J Gastroenterol Hepatol. 16:207–212. DOI: 10.1097/00042737-200402000-00014. PMID: 15075996.
Article32. Sartor RB. 2006; Mechanisms of disease: pathogenesis of Crohn's disease and ulcerative colitis. Nat Clin Pract Gastroenterol Hepatol. 3:390–407. DOI: 10.1038/ncpgasthep0528. PMID: 16819502.
Article33. Valentini L, Schaper L, Buning C, et al. 2008; Malnutrition and impaired muscle strength in patients with Crohn's disease and ulcerative colitis in remission. Nutrition. 24:694–702. DOI: 10.1016/j.nut.2008.03.018. PMID: 18499398.
Article34. Silverberg MS, Satsangi J, Ahmad T, et al. 2005; Toward an integrated clinical, molecular and serological classification of inflammatory bowel disease: report of a Working Party of the 2005 Montreal World Congress of Gastroenterology. Can J Gastroenterol . 19 Suppl A:5A–36A. DOI: 10.1155/2005/269076. PMID: 16151544.
Article35. Ford AC, Bernstein CN, Khan KJ, et al. 2011; Glucocorticosteroid therapy in inflammatory bowel disease: systematic review and meta-analysis. Am J Gastroenterol. 106:590–599. DOI: 10.1038/ajg.2011.70. PMID: 21407179.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Additive effect of probiotics (Mutaflor) on 5-aminosalicylic acid therapy in patients with ulcerative colitis
- A Case of Escherichia coli O157 Hemorrhagic Colitis
- Remission of Ulcerative Colitis after Appendectomy: A Case Report
- Anti-inflammatory properties of Escherichia coli Nissle 1917 in a murine colitis model
- A Case of Hemolytic Uremic Syndrome Caused by Escherichia coli O8: Case Report