Anesth Pain Med.
2020 Jul;15(3):297-304. 10.17085/apm.20007.
Effects of various methods of dexmedetomidine administration for sedation in elderly patients undergoing spinal anesthesia: a randomized controlled study
- Affiliations
-
- 1Department of Anesthesiology and Pain Medicine, Ewha Womans University Mokdong Hospital, Ewha Womans University College of Medicine, Seoul, Korea
- KMID: 2504891
- DOI: http://doi.org/10.17085/apm.20007
Abstract
- Background
The purpose of this study was to investigate the degree of sedation and the incidence of adverse effects resulting from various methods of administering the initial dose followed by continuous infusion of dexmedetomidine (DEX) for sedation in elderly patients undergoing spinal anesthesia.
Methods
In total, 72 patients aged over 65 years who were to be administered spinal anesthesia were randomly allocated into three groups. The initial doses were injected to the groups as follows: group DD, DEX 0.5 μg/kg for 10 min; group MD, midazolam 0.02 mg/kg; and group D, no initial dose. This was followed immediately by infusing a maintenance dose of DEX 0.5 μg/kg/h to all groups.
Results
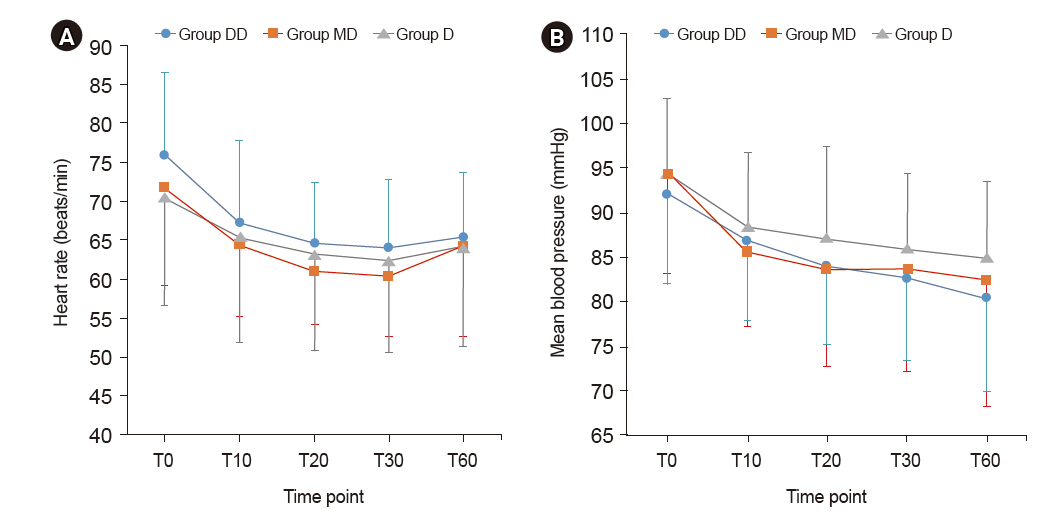
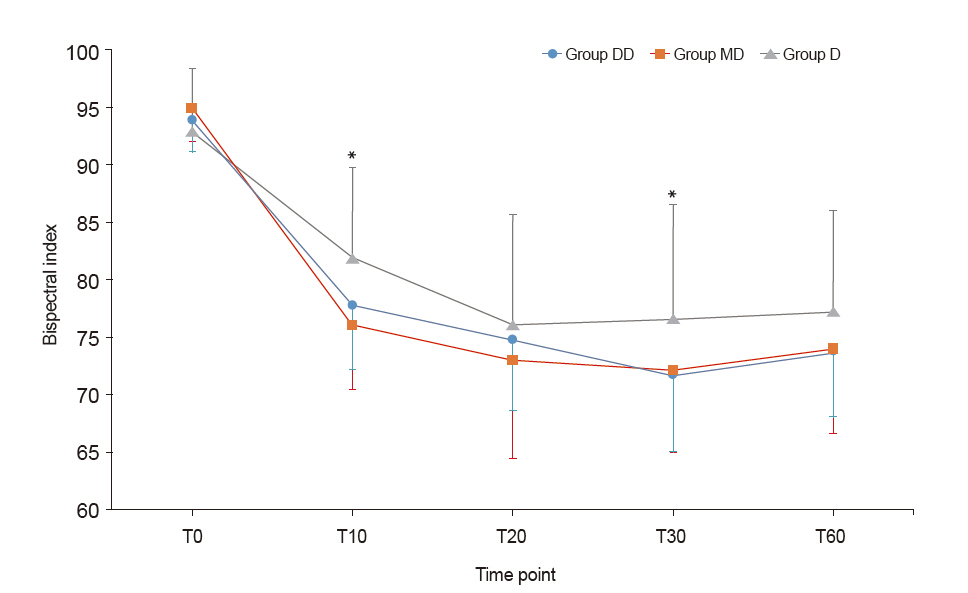
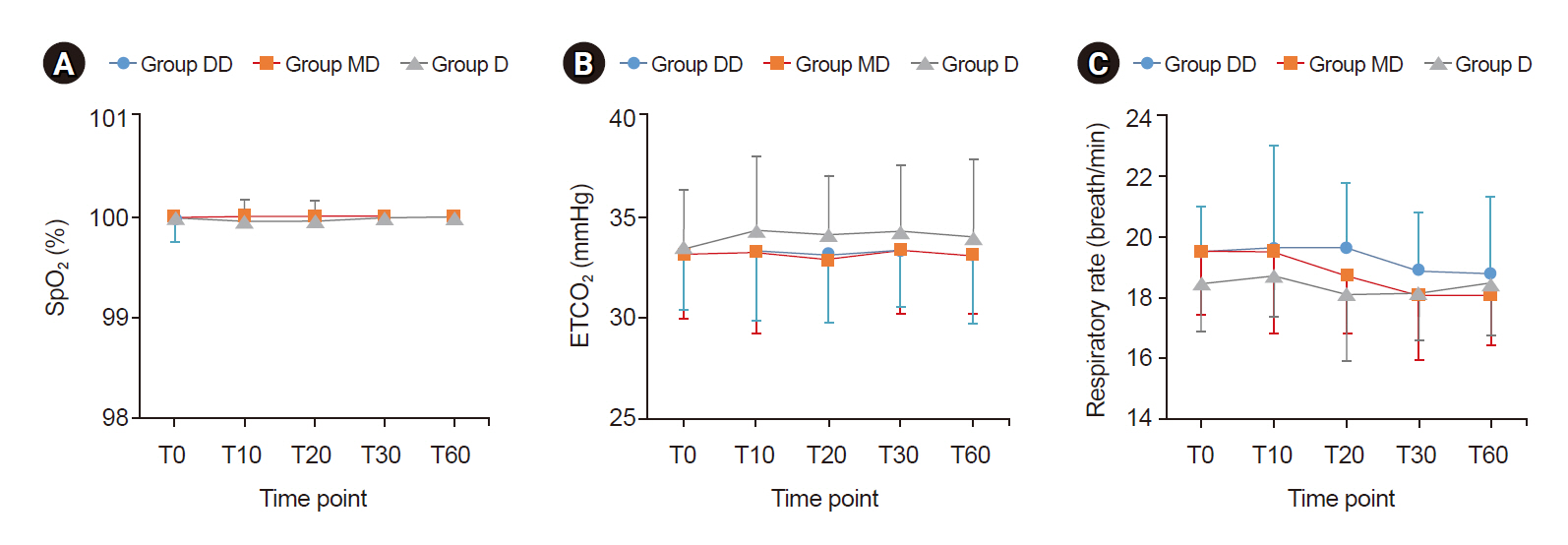
The Bispectral index (BIS) in the D group was significantly higher than in the other two groups. There were no significant differences in the Ramsay sedation scale (RSS) among the groups. The RSS 3 level was reached in 10 min from the start of sedation in MD and DD groups and in 20 min from the start of sedation in D group. Neither bradycardia nor hypotension was observed in any of the groups.
Conclusions
Patients in all three groups reached the RSS 3 sedating-effect level. However, the group that received continuous infusion only without the initial dose showed higher BIS than the other two groups and reached the RSS 3 later. No adverse events were observed in any of the groups.
Keyword
Figure
Cited by 1 articles
-
Spinal anesthesia in elective lumbar spinal surgery
Nicholas S. Hernandez, Benayas Begashaw, Ron I. Riesenburger, James T. Kryzanski, Penny Liu
Anesth Pain Med. 2023;18(4):349-356. doi: 10.17085/apm.23031.
Reference
-
1. Jakob SM, Ruokonen E, Grounds RM, Sarapohja T, Garratt C, Pocock SJ, et al. Dexmedetomidine vs midazolam or propofol for sedation during prolonged mechanical ventilation: two randomized controlled trials. JAMA. 2012; 307:1151–60.2. Fraser GL, Devlin JW, Worby CP, Alhazzani W, Barr J, Dasta JF, et al. Benzodiazepine versus nonbenzodiazepine-based sedation for mechanically ventilated, critically ill adults: a systematic review and meta-analysis of randomized trials. Crit Care Med. 2013; 41(9 Suppl 1):S30–8.3. Sebel PS, Lang E, Rampil IJ, White PF, Cork R, Jopling M, et al. A multicenter study of bispectral electroencephalogram analysis for monitoring anesthetic effect. Anesth Analg. 1997; 84:891–9.4. Lakatta EG. Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: Part III: cellular and molecular clues to heart and arterial aging. Circulation. 2003; 107:490–7.5. Priebe HJ. The aged cardiovascular risk patient. Br J Anaesth. 2000; 85:763–78.6. Ebert TJ, Hall JE, Barney JA, Uhrich TD, Colinco MD. The effects of increasing plasma concentrations of dexmedetomidine in humans. Anesthesiology. 2000; 93:382–94.7. Dawson R, von Fintel N, Nairn S. Sedation assessment using the Ramsay scale. Emerg Nurse. 2010; 18:18–20.8. Yoon DK, Ban JS, Lee SG, Lee JH, Kim E, An J. Dexmedetomidine combined with midazolam vs. dexmedetomidine alone for sedation during spinal anesthesia. Korean J Anesthesiol. 2016; 69:446–52.9. Kallio A, Scheinin M, Koulu M, Ponkilainen R, Ruskoaho H, Viinamäki O, et al. Effects of dexmedetomidine, a selective alpha 2-adrenoceptor agonist, on hemodynamic control mechanisms. Clin Pharmacol Ther. 1989; 46:33–42.10. Paris A, Tonner PH. Dexmedetomidine in anaesthesia. Curr Opin Anaesthesiol. 2005; 18:412–8.11. Arain SR, Ebert TJ. The efficacy, side effects, and recovery characteristics of dexmedetomidine versus propofol when used for intraoperative sedation. Anesth Analg. 2002; 95:461–6.12. Riker RR, Shehabi Y, Bokesch PM, Ceraso D, Wisemandle W, Koura F, et al. Dexmedetomidine vs midazolam for sedation of critically ill patients: a randomized trial. JAMA. 2009; 301:489–99.13. Yeom JH, Ahn DW, Kim KH. Comparison of several dosing schedules of intravenous dexmedetomidine in elderly patients under spinal anesthesia. Anesth Pain Med. 2017; 12:320–5.14. Yeom JH, Oh MK, Ahn DW, Park SI. A loading dose of 1 μg/kg and maintenance dose of 0.5 μg/kg/h of dexmedetomidine for sedation under spinal anesthesia may induce excessive sedation and airway obstruction. Anesth Pain Med. 2016; 11:255–9.15. Johson JO, Grecu L, Lawson NW. Autonomic nervous system. In: Clinical anesthesia. 6th ed. Edited by Barash PG, Cullen BF, Stoelting RK, Cahalan MK, Stock MC: Philadelphia, Wolters Kluwer/Lippincott Williams & Wilkins. 2009, pp 326-68.16. Alexander CM, Gross JB. Sedative doses of midazolam depress hypoxic ventilatory responses in humans. Anesth Analg. 1988; 67:377–82.17. Nishiyama T, Hirasaki A, Odaka Y, Iwasaki T, Seto K. Midazolam sedation during spinal anesthesia: optimal dosage. J Jpn Soc Clin Anesth. 1994; 14:257–62.18. Yano H, Iishi H, Tatsuta M, Sakai N, Narahara H, Omori M. Oxygen desaturation during sedation for colonoscopy in elderly patients. Hepatogastroenterology. 1998; 45:2138–41.19. Liu J, Singh H, White PF. Electroencephalographic bispectral index correlates with intraoperative recall and depth of propofol-induced sedation. Anesth Analg. 1997; 84:185–9.20. Bell JK, Laasch HU, Wilbraham L, England RE, Morris JA, Martin DF. Bispectral index monitoring for conscious sedation in intervention: better, safer, faster. Clin Radiol. 2004; 59:1106–13.21. Weinbroum AA, Szold O, Ogorek D, Flaishon R. The midazolam-induced paradox phenomenon is reversible by flumazenil. Epidemiology, patient characteristics and review of the literature. Eur J Anaesthesiol. 2001; 18:789–97.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Fatal pulmonary aspiration during balanced sedation with dexmedetomidine and midazolam: A case report
- The effects of loading dose administration rate of dexmedetomidine on sedation and dexmedetomidine requirement in elderly patients undergoing spinal anesthesia
- Effect of Dexmedetomidine on the Corrected QT and Tp-e Intervals during Spinal Anesthesia
- Dexmedetomidine intravenous sedation using a patient-controlled sedation infusion pump: a case report
- Effective dose of dexmedetomidine to induce adequate sedation in elderly patients under spinal anesthesia