Anesth Pain Med.
2018 Jul;13(3):264-270. 10.17085/apm.2018.13.3.264.
The effects of loading dose administration rate of dexmedetomidine on sedation and dexmedetomidine requirement in elderly patients undergoing spinal anesthesia
- Affiliations
-
- 1Department of Anesthesiology and Pain Medicine, Kosin University College of Medicine, Busan, Korea. uamyfriends@hanmail.net
- KMID: 2436033
- DOI: http://doi.org/10.17085/apm.2018.13.3.264
Abstract
- BACKGROUND
This study evaluated the effect of decrease in loading dose administration rate of dexmedetomidine (DMT) on sedation and DMT requirement in elderly patients.
METHODS
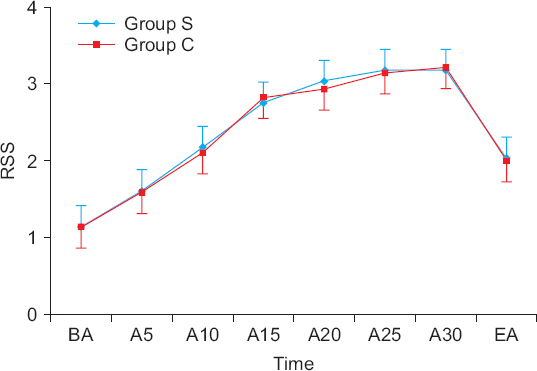
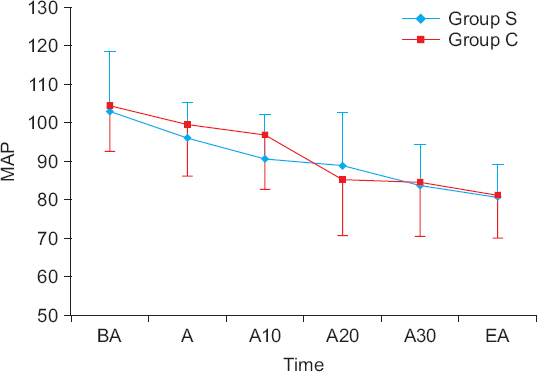
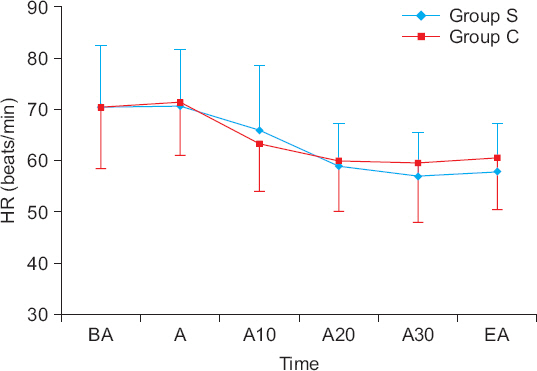
Fifty-eight patients over 65 years old with ASA I-II who were planned to receive DMT sedation during spinal anesthesia were randomly assigned to two groups. Group S (n = 29) received a 0.5 µg/kg DMT loading dose over 20 minutes, while group C (n = 29) received the DMT loading dose over 10 minutes. Then, both groups received a continuous infusion of 0.4 µg/kg/h. The sedative status was recorded before and at 5, 10, 15, 20, 25, and 30 minutes after administration of DMT and at the end of the anesthesia according to the Ramsay sedation scale (RSS). Also, the time to reach RSS-3 (patients asleep, responsive to commands) and the dose of DMT until reaching RSS-3 were recorded.
RESULTS
The time to reach RSS-3 was similar between the two groups (group S = 16.0 ± 4.3 minutes vs. group C = 15.5 ± 4.2 minutes, P = 0.673). However, the DMT required to reach RSS-3 in group S was significantly lower than that in group C (23.3 ± 7.1 vs. 32.5 ± 6.0 µg, P < 0.001). There was no difference in RSS between the two groups from the administration of DMT to the end of the anesthesia (P = 0.927).
CONCLUSIONS
Decreasing the administration rate of the DMT loading dose did not delay the onset of RSS-3 sedation and reduced the DMT requirement in elderly patients.
Keyword
MeSH Terms
Figure
Reference
-
1. Hall JE, Uhrich TD, Barney JA, Arain SR, Ebert TJ. Sedative, amnestic, and analgesic properties of small-dose dexmedetomidine infusions. Anesth Analg. 2000; 90:699–705. DOI: 10.1097/00000539-200003000-00035. PMID: 10702460.2. Gertler R, Brown HC, Mitchell DH, Silvius EN. Dexmedetomidine: a novel sedative-analgesic agent. Proc (Bayl Univ Med Cent). 2001; 14:13–21. DOI: 10.1080/08998280.2001.11927725.3. Gurbet A, Basagan-Mogol E, Turker G, Ugun F, Kaya FN, Ozcan B. Intraoperative infusion of dexmedetomidine reduces perioperative analgesic requirements. Can J Anaesth. 2006; 53:646–52. DOI: 10.1007/BF03021622. PMID: 16803911.4. Carollo DS, Nossaman BD, Ramadhyani U. Dexmedetomidine: a review of clinical applications. Curr Opin Anaesthesiol. 2008; 21:457–61. DOI: 10.1097/ACO.0b013e328305e3ef. PMID: 18660652.5. Fan TW, Ti LK, Islam I. Comparison of dexmedetomidine and midazolam for conscious sedation in dental surgery monitored by bispectral index. Br J Oral Maxillofac Surg. 2013; 51:428–33. DOI: 10.1016/j.bjoms.2012.08.013. PMID: 23058230.6. Paris A, Tonner PH. Dexmedetomidine in anaesthesia. Curr Opin Anaesthesiol. 2005; 18:412–8. DOI: 10.1097/01.aco.0000174958.05383.d5. PMID: 16534267.7. Giovannitti JA Jr, Thoms SM, Crawford JJ. Alpha-2 adrenergic receptor agonists: a review of current clinical applications. Anesth Prog. 2015; 62:31–9. DOI: 10.2344/0003-3006-62.1.31. PMID: 25849473. PMCID: PMC4389556.8. Ebert TJ, Hall JE, Barney JA, Uhrich TD, Colinco MD. The effects of increasing plasma concentrations of dexmedetomidine in humans. Anesthesiology. 2000; 93:382–94. DOI: 10.1097/00000542-200008000-00016. PMID: 10910487.9. Tanskanen PE, Kyttä JV, Randell TT, Aantaa RE. Dexmedetomidine as an anaesthetic adjuvant in patients undergoing intracranial tumour surgery: a double-blind, randomized and placebo-controlled study. Br J Anaesth. 2006; 97:658–65. DOI: 10.1093/bja/ael220. PMID: 16914460.10. Ice CJ, Personett HA, Frazee EN, Dierkhising RA, Kashyap R, Oeckler RA. Risk factors for dexmedetomidine-associated hemodynamic instability in noncardiac intensive care unit patients. Anesth Analg. 2016; 122:462–9. DOI: 10.1213/ANE.0000000000001125. PMID: 26702868.11. Park SH, Shin YD, Yu HJ, Bae JH, Yim KH. Comparison of two dosing schedules of intravenous dexmedetomidine in elderly patients during spinal anesthesia. Korean J Anesthesiol. 2014; 66:371–6. DOI: 10.4097/kjae.2014.66.5.371. PMID: 24910729. PMCID: PMC4041956.12. Ko KH, Jun IJ, Lee S, Lim Y, Yoo B, Kim KM. Effective dose of dexmedetomidine to induce adequate sedation in elderly patients under spinal anesthesia. Korean J Anesthesiol. 2015; 68:575–80. DOI: 10.4097/kjae.2015.68.6.575. PMID: 26634081. PMCID: PMC4667143.13. Ramsay MA, Savege TM, Simpson BR, Goodwin R. Controlled sedation with alphaxalone-alphadolone. Br Med J. 1974; 2:656–9. DOI: 10.1136/bmj.2.5920.656. PMID: 4835444. PMCID: PMC1613102.14. Iirola T, Ihmsen H, Laitio R, Kentala E, Aantaa R, Kurvinen JP, et al. Population pharmacokinetics of dexmedetomidine during long-term sedation in intensive care patients. Br J Anaesth. 2012; 108:460–8. DOI: 10.1093/bja/aer441. PMID: 22277665.15. Angst MS, Ramaswamy B, Davies MF, Maze M. Comparative analgesic and mental effects of increasing plasma concentrations of dexmedetomidine and alfentanil in humans. Anesthesiology. 2004; 101:744–52. DOI: 10.1097/00000542-200409000-00024. PMID: 15329600.16. Goodwin H, Lewin JJ, Mirski MA. 'Cooperative sedation': optimizing comfort while maximizing systemic and neurological function. Crit Care. 2012; 16:217. DOI: 10.1186/cc11231. PMID: 22429840. PMCID: PMC3681362.17. Ostermann ME, Keenan SP, Seiferling RA, Sibbald WJ. Sedation in the intensive care unit: a systematic review. JAMA. 2000; 283:1451–9. DOI: 10.1001/jama.283.11.1451. PMID: 10732935.18. Hong JY, Kim WO, Yoon Y, Choi Y, Kim SH, Kil HK. Effects of intravenous dexmedetomidine on low-dose bupivacaine spinal anaesthesia in elderly patients. Acta Anaesthesiol Scand. 2012; 56:382–7. DOI: 10.1111/j.1399-6576.2011.02614.x. PMID: 22220945.19. Belleville JP, Ward DS, Bloor BC, Maze M. Effects of intravenous dexmedetomidine in humans. I. Sedation, ventilation, and metabolic rate. Anesthesiology. 1992; 77:1125–33. DOI: 10.1097/00000542-199212000-00013. PMID: 1361310.20. Kronenberg RS, Drage CW. Attenuation of the ventilatory and heart rate responses to hypoxia and hypercapnia with aging in normal men. J Clin Invest. 1973; 52:1812–9. DOI: 10.1172/JCI107363. PMID: 4719663. PMCID: PMC302461.21. Bloor BC, Ward DS, Belleville JP, Maze M. Effects of intravenous dexmedetomidine in humans. II. Hemodynamic changes. Anesthesiology. 1992; 77:1134–42. DOI: 10.1097/00000542-199212000-00014. PMID: 1361311.22. Gerlach AT, Blais DM, Jones GM, Burcham PK, Stawicki SP, Cook CH, et al. Predictors of dexmedetomidine-associated hypotension in critically ill patients. Int J Crit Illn Inj Sci. 2016; 6:109–14. DOI: 10.4103/2229-5151.190656. PMID: 27722111. PMCID: PMC5051052.23. Höhener D, Blumenthal S, Borgeat A. Sedation and regional anaesthesia in the adult patient. Br J Anaesth. 2008; 100:8–16. DOI: 10.1093/bja/aem342. PMID: 18070783.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Effective dose of dexmedetomidine to induce adequate sedation in elderly patients under spinal anesthesia
- Fatal pulmonary aspiration during balanced sedation with dexmedetomidine and midazolam: A case report
- Dexmedetomidine combined with midazolam vs. dexmedetomidine alone for sedation during spinal anesthesia
- Dexmedetomidine for sedation of patients undergoing elective surgery under regional anesthesia
- Optimal dose of dexmedetomidine for sedation during spinal anesthesia