Lab Med Online.
2020 Jul;10(3):202-206. 10.3343/lmo.2020.10.3.202.
Performance Evaluation of the ABL90 FLEX PLUS Point-of-Care Analyzer in Measuring Creatinine Levels
- Affiliations
-
- 1Department of Laboratory Medicine, National Health Insurance Service Ilsan Hospital, Goyang, Korea
- 2Department of Laboratory Medicine, Yonsei University College of Medicine, Seoul, Korea
- KMID: 2504129
- DOI: http://doi.org/10.3343/lmo.2020.10.3.202
Abstract
- Background
To diagnose acute renal failure, creatinine levels in whole blood are typically assessed using point-of-care testing (POCT) methods. The present study aimed to evaluate the performance of a newly developed POCT blood gas analyzer, the ABL90 FLEX PLUS (Radiometer, Denmark), which can measure creatinine in blood.
Methods
Precision and linearity of the ABL90 FLEX PLUS were evaluated and compared with those of the Beckman Coulter AU5800 (Beckman Coulter, USA), according to the CLSI guidelines for creatinine measurement performance.
Results
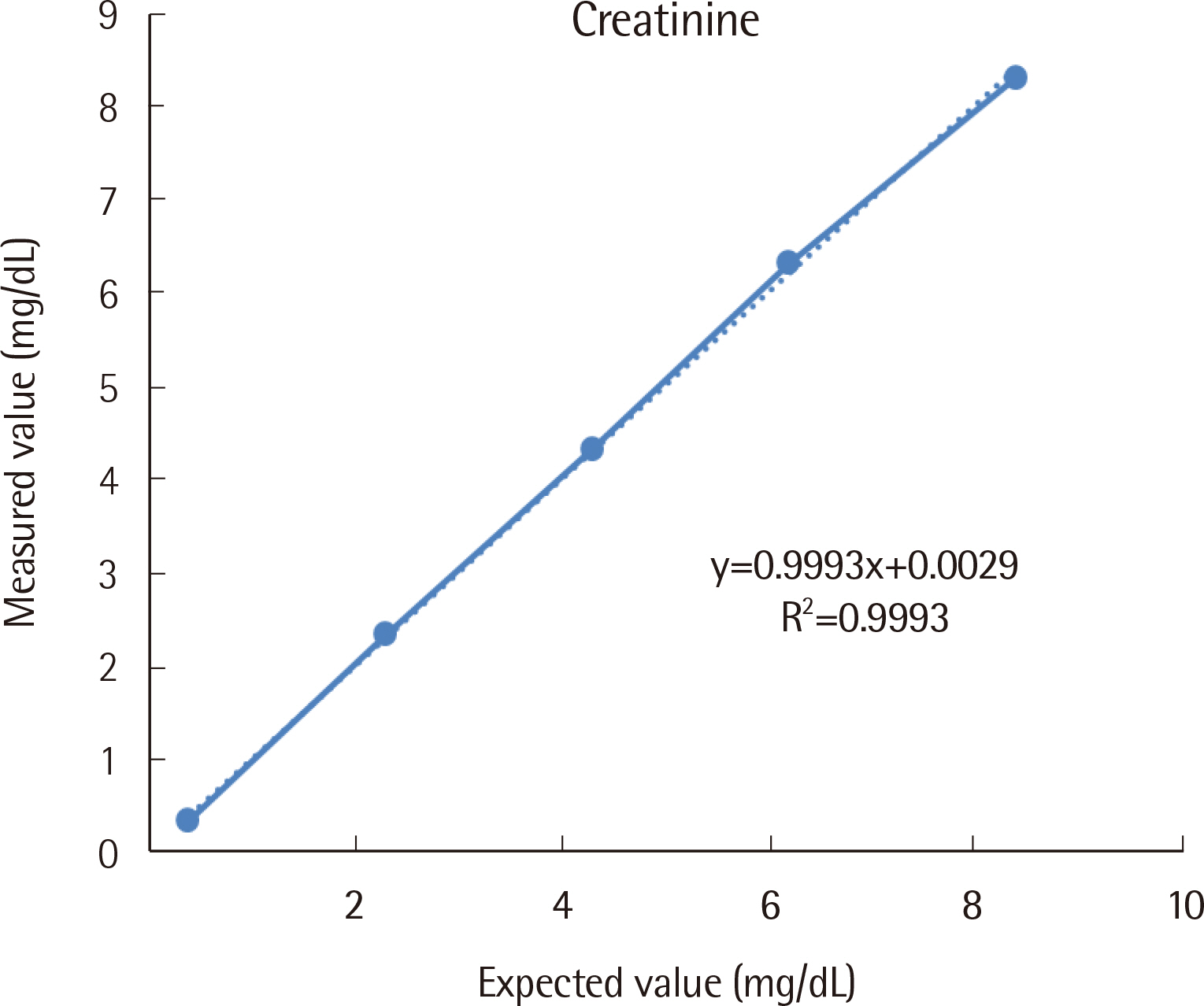
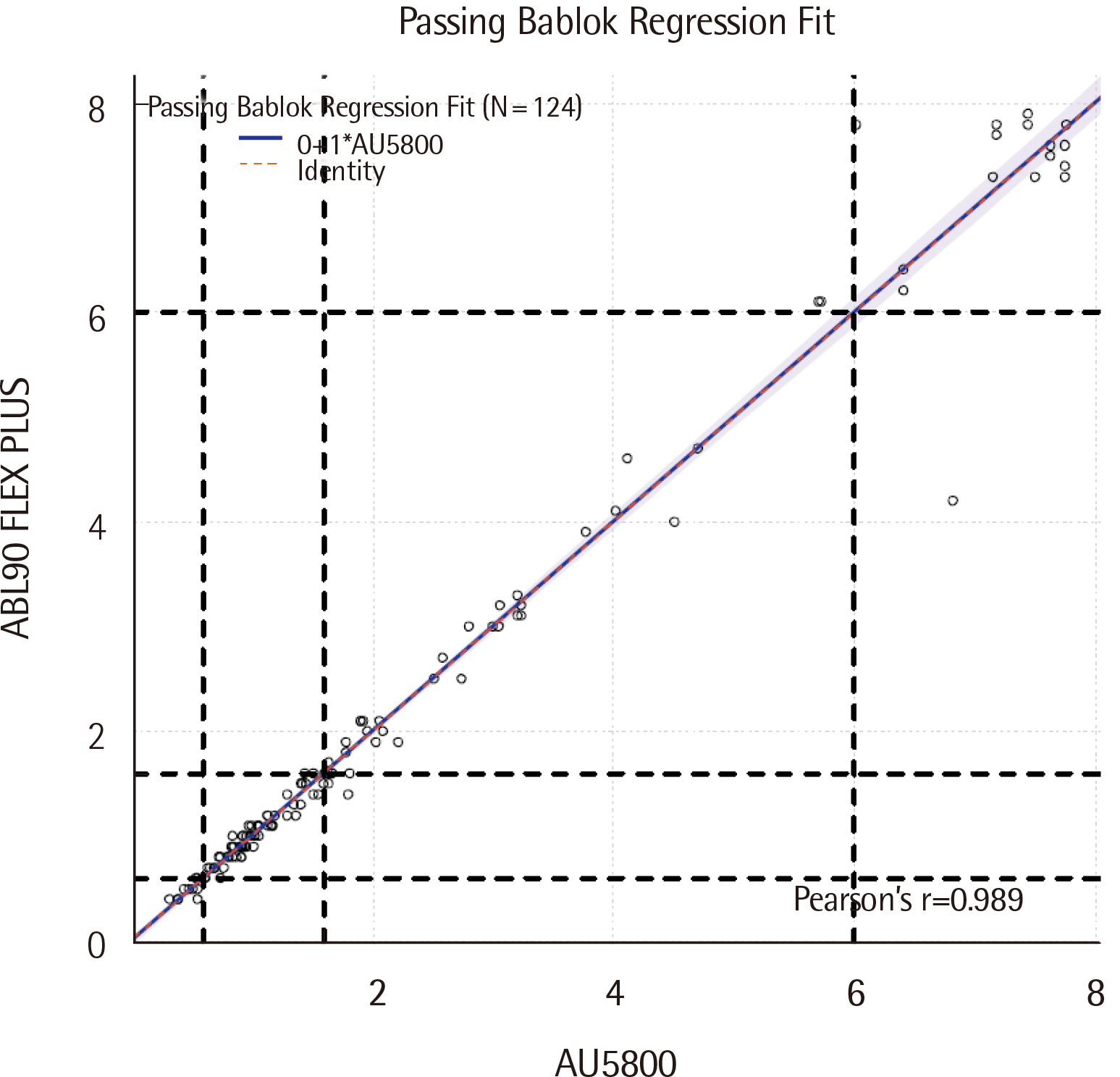
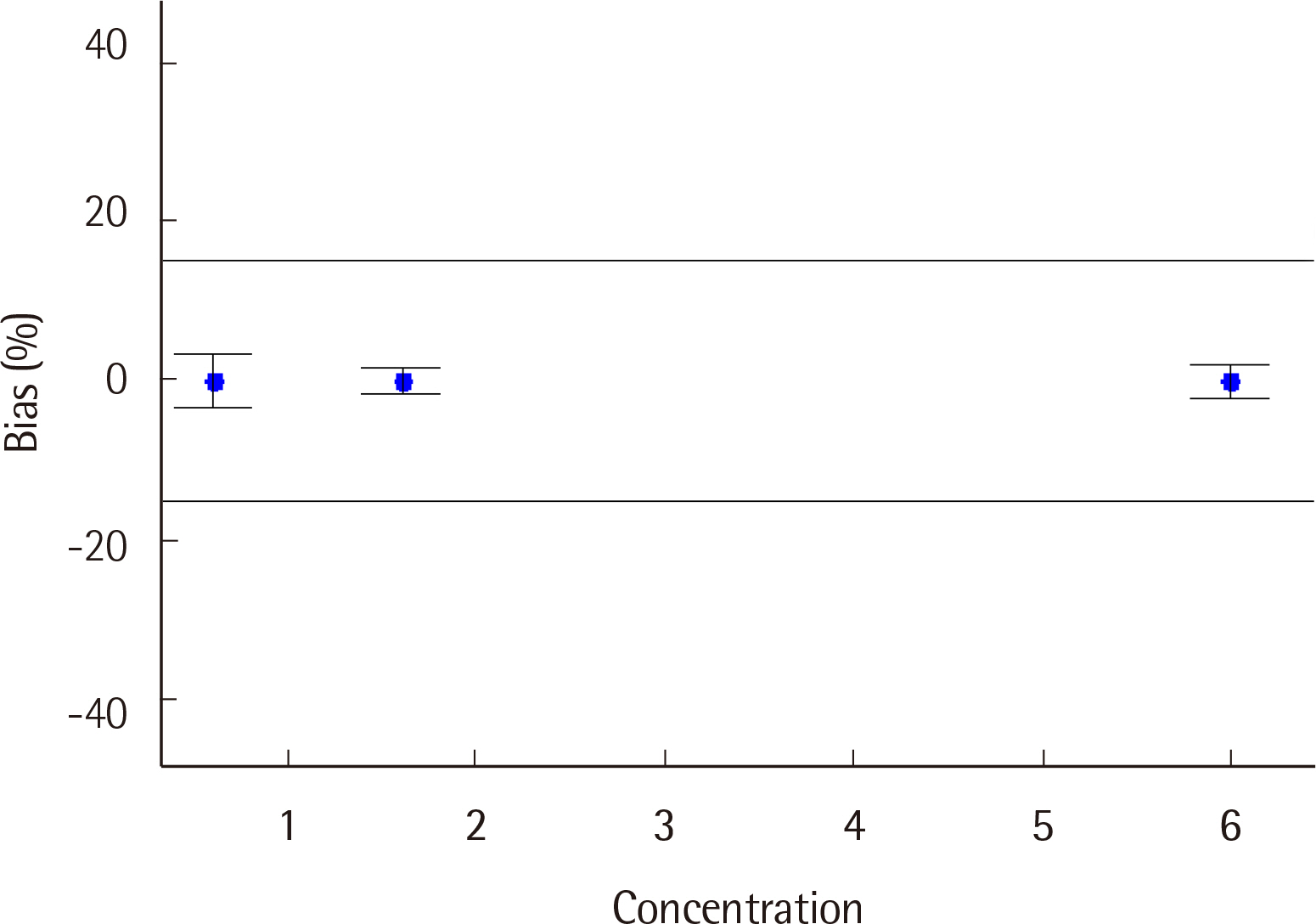
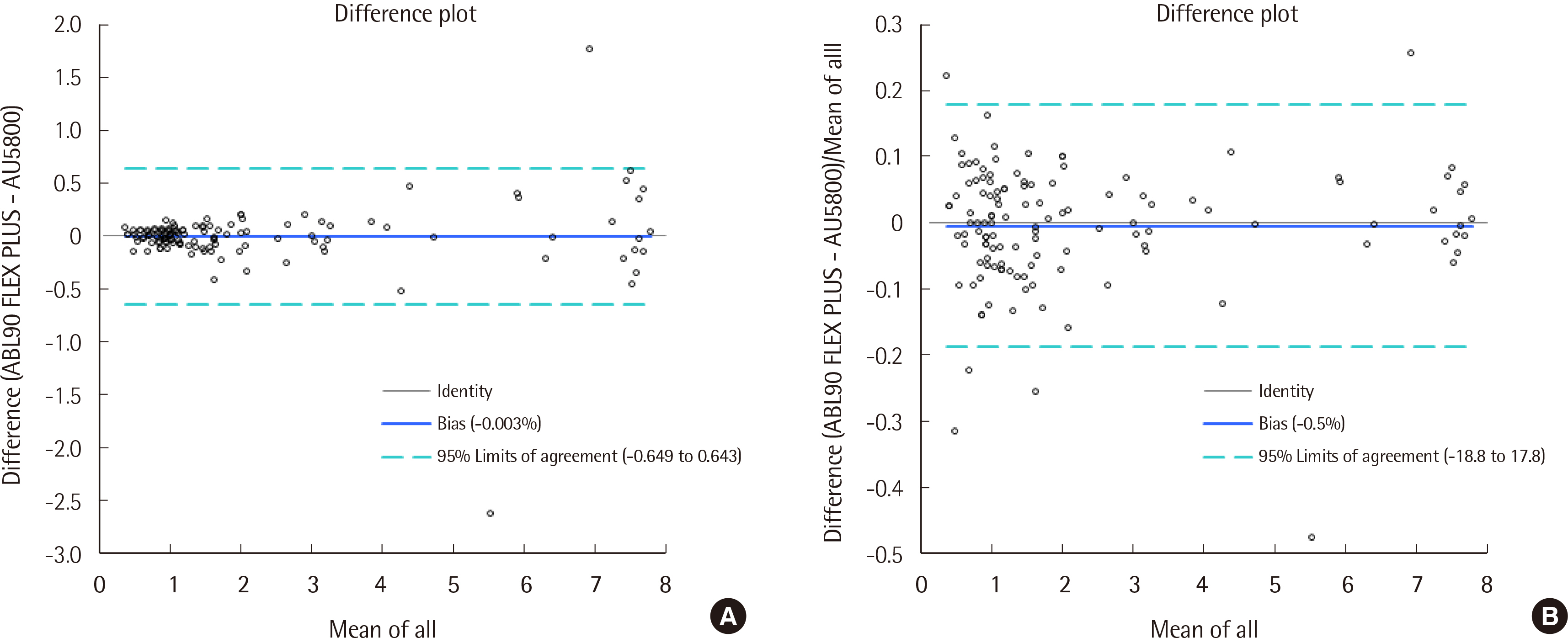
For the ABL90 FLEX PLUS, the total imprecision (%CV) levels of two control materials were measured to be 0.0% and 0.8%, while linearity was evaluated, with the R2 value measured to be 0.9993 (0.4-8.4 mg/dL). Compared to the AU5800, the ABL90 FLEX PLUS correlation coefficient (r) was found to be 0.989. The 95% limits of agreement were determined to be -0.649 and 0.643 mg/dL (-18.8% and 17.8%).
Conclusions
The ABL90 FLEX PLUS exhibited good performance for creatinine test. This indicates that the ABL90 FLEX PLUS can be potentially useful in clinical laboratories.
Figure
Reference
-
1. Levey AS, Perrone RD, Madias NE. 1988; Serum creatinine and renal function. Annu Rev Med. 39:465–90. DOI: 10.1146/annurev.me.39.020188.002341. PMID: 3285786.
Article2. Levey AS, Coresh J, Greene T, Marsh J, Stevens LA, Kusek JW, et al. 2007; Expressing the modification of diet in renal disease study equation for estimating glomerular filtration rate with standardized serum creatinine values. Clin Chem. 53:766–72. DOI: 10.1373/clinchem.2006.077180. PMID: 17332152.
Article3. Barrett BJ, Parfrey PS. 2006; Clinical practice. Preventing nephropathy induced by contrast medium. N Engl J Med. 354:379–86. DOI: 10.1056/NEJMcp050801. PMID: 16436769.4. Shephard M, Peake M, Corso O, Shephard A, Mazzachi B, Spaeth B, et al. 2010; Assessment of the Nova StatSensor whole blood point-of-care creatinine analyzer for the measurement of kidney function in screening for chronic kidney disease. Clin Chem Lab Med. 48:1113–9. DOI: 10.1515/CCLM.2010.238. PMID: 20482303.
Article5. Clinical and Laboratory Standards Institute. 2014; Evaluation of precision performance of quantitative measurement methods; Approved guideline-Third edition. CLSI document EP05-A3. Clinical and Laboratory Standards Institute;Wayne, PA:6. Clinical and Laboratory Standards Institute. 2003; Evaluation of the linearity of quantitative measurement procedures: A statistical approach; Approved guideline. CLSI document EP06-A. Clinical and Laboratory Standards Institute;Wayne, PA:7. Clinical and Laboratory Standards Institute. 2013; Measurement procedure comparison and bias estimation using patient samples; Approved guideline-Third edition. CLSI document EP09-A3. Clinical and Laboratory Standards Institute;Wayne, PA:8. Bogaert L, Schiemsky T, Van Hover P, De Schrijver P, Van Hoovels L. 2019; Analytical and diagnostic performance evaluation of five creatinine POCT devices in the identification of patients at risk for post-contrast acute kidney injury (PCAKI). Clin Chem Lab Med. 57:e214–7. DOI: 10.1515/cclm-2018-1105. PMID: 30710476.
Article9. Syal K, Banerjee D, Srinivasan A. 2013; Creatinine estimation and interference. Indian J Clin Biochem. 28:210–1. DOI: 10.1007/s12291-013-0299-y. PMID: 24426213. PMCID: PMC3613509.
Article10. Hunter A, Campbell WR. 1917; The probable accuracy, in whole blood and plasma, of colorimetric determinations of creatinine and creatine. J Biol Chem. 32:195–231.
Article11. Mitchell AM, Jones AE, Tumlin JA, Kline JA. 2010; Incidence of contrast-induced nephropathy after contrast-enhanced computed tomography in the outpatient setting. Clin J Am Soc Nephrol. 5:4–9. DOI: 10.2215/CJN.05200709. PMID: 19965528. PMCID: PMC2801649.
Article12. Ko GJ. 2015; Contrast-Induced Nephropathy. Korean J Med. 88:375–81. DOI: 10.3904/kjm.2015.88.4.375.
Article13. Lee-Lewandrowski E, Chang C, Gregory K, Lewandrowski K. 2012; Evaluation of rapid point-of-care creatinine testing in the radiology service of a large academic medical center: impact on clinical operations and patient disposition. Clin Chim Acta. 413:88–92. DOI: 10.1016/j.cca.2011.05.006. PMID: 21600893.
Article14. Carden AJ, Salcedo ES, Tran NK, Gross E, Mattice J, Shepard J, et al. 2016; Prospective observational study of point-of-care creatinine in trauma. Trauma Surg Acute Care Open. 1:e000014. DOI: 10.1136/tsaco-2016-000014. PMID: 29766058. PMCID: PMC5891719.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Performance Evaluation of the Piccolo xpress Point-of-care Chemistry Analyzer
- Performance Evaluation of the LABGEO PT10 Point-of-care Chemistry Analyzer
- Evaluation of the i-Smart 30 Point-of-Care Analyzer for Use in Clinical Laboratory Settings
- Evaluation of Analytical Performance of an Automated Glycated Hemoglobin Analyzer, HLC-723 G11
- Performance Evaluation of the CAPILLARYS 2 FLEX Piercing Analyzer for HbA1c Determination