Nutr Res Pract.
2020 Jun;14(3):175-187. 10.4162/nrp.2020.14.3.175.
Skate cartilage extracts containing chondroitin sulfate ameliorates hyperlipidemia-induced inflammation and oxidative stress in high cholesterol diet-fed LDL receptor knockout mice in comparison with shark chondroitin sulfate
- Affiliations
-
- 1Department of Food Science and Nutrition & Kimchi Research Institute, Pusan National University, Busan 46241, Korea
- 2Busan Innovation Institute of Industry, Science & Technology Planning (BISTEP), Busan 48058, Korea
- 3Anti-Aging Research Center, Dong-eui University, Busan 47227, Korea
- 4Department of of Biochemistry, Dong-eui University College of Korean Medicine, Busan 47227, Korea
- 5Department of of Food Science and Nutrition, Tongmyong University, Busan 48520, Korea
- KMID: 2502479
- DOI: http://doi.org/10.4162/nrp.2020.14.3.175
Abstract
- BACKGROUND/OBJECTIVES
In this study, we investigated the beneficial effects of skate cartilage extracts containing chondroitin sulfate (SCS) on hyperlipidemia-induced inflammation and oxidative stress in high cholesterol diet (HCD)-fed mice in comparison with the effects of shark cartilage-derived chondroitin sulfate (CS).
MATERIALS/METHODS
Low-density lipoprotein receptor knockout (LDLR-KO) mice were fed HCD with an oral administration of CS (50 and 100 mg/kg BW/day), SCS (100 and 200 mg/kg BW/day), or water, respectively, for ten weeks.
RESULTS
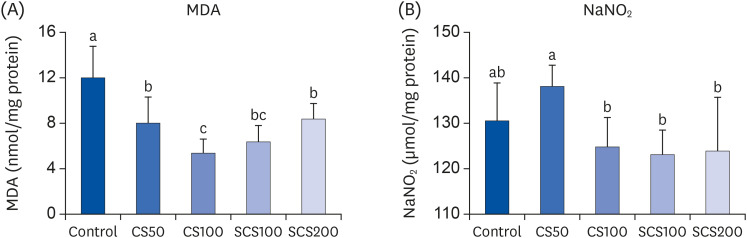
The administration of CS or SCS reduced the levels of serum triglyceride (TG), total cholesterol (TC), and LDL cholesterol and elevated the levels of high-density lipoprotein cholesterol, compared with those of the control group (P < 0.05). Furthermore, CS or SCS significantly attenuated inflammation by reducing the serum levels of interleukin (IL)-1β and hepatic protein expression levels of nuclear factor kappa B, inducible nitric oxide synthase, cyclooxygenase-2, and IL-1beta (P < 0.05). In particular, the serum level of tumor necrosis factor-alpha was reduced only in the 100 mg/kg BW/day of SCS-fed group, whereas the IL-6 level was reduced in the 100 and 200 mg/kg BW/day of SCS-fed groups (P < 0.05). In addition, lipid peroxidation and nitric oxide production were attenuated in the livers of the CS and SCS groups mediated by the upregulation of hepatic proteins of antioxidant enzymes, such as superoxide dismutase, catalase, and glutathione peroxidase (P < 0.05).
CONCLUSIONS
These results suggest that the biological effects of SCS, similar to those of CS, are attributed to improved lipid profiles as well as suppressed inflammation and oxidative stress induced by the intake of HCD.
Figure
Reference
-
1. Tannock LR. Advances in the management of hyperlipidemia-induced atherosclerosis. Expert Rev Cardiovasc Ther. 2008; 6:369–383. PMID: 18327996.
Article2. Nelson RH. Hyperlipidemia as a risk factor for cardiovascular disease. Prim Care. 2013; 40:195–211. PMID: 23402469.
Article3. Tomizawa M, Kawanabe Y, Shinozaki F, Sato S, Motoyoshi Y, Sugiyama T, Yamamoto S, Sueishi M. Triglyceride is strongly associated with nonalcoholic fatty liver disease among markers of hyperlipidemia and diabetes. Biomed Rep. 2014; 2:633–636. PMID: 25054002.
Article4. Wouters K, van Gorp PJ, Bieghs V, Gijbels MJ, Duimel H, Lütjohann D, Kerksiek A, van Kruchten R, Maeda N, Staels B, van Bilsen M, Shiri-Sverdlov R, Hofker MH. Dietary cholesterol, rather than liver steatosis, leads to hepatic inflammation in hyperlipidemic mouse models of nonalcoholic steatohepatitis. Hepatology. 2008; 48:474–486. PMID: 18666236.
Article5. Onody A, Csonka C, Giricz Z, Ferdinandy P. Hyperlipidemia induced by a cholesterol-rich diet leads to enhanced peroxynitrite formation in rat hearts. Cardiovasc Res. 2003; 58:663–670. PMID: 12798440.6. Czakó L, Szabolcs A, Vajda A, Csáti S, Venglovecz V, Rakonczay Z Jr, Hegyi P, Tiszlavicz L, Csont T, Pósa A, Berkó A, Varga C, Varga Ilona S, Boros I, Lonovics J. Hyperlipidemia induced by a cholesterolrich diet aggravates necrotizing pancreatitis in rats. Eur J Pharmacol. 2007; 572:74–81. PMID: 17628538.
Article7. Puskás LG, Nagy ZB, Giricz Z, Ónody A, Csonka C, Kitajka K, Hackler L Jr, Zvara A, Ferdinandy P. Cholesterol diet-induced hyperlipidemia influences gene expression pattern of rat hearts: a DNA microarray study. FEBS Lett. 2004; 562:99–104. PMID: 15044008.
Article8. Lee LC, Wei L, Huang WC, Hsu YJ, Chen YM, Huang CC. Hypolipidemic effect of tomato juice in hamsters in high cholesterol diet-induced hyperlipidemia. Nutrients. 2015; 7:10525–10537. PMID: 26694461.
Article9. Bondia-Pons I, Ryan L, Martinez JA. Oxidative stress and inflammation interactions in human obesity. J Physiol Biochem. 2012; 68:701–711. PMID: 22351038.
Article10. Harmon RC, Tiniakos DG, Argo CK. Inflammation in nonalcoholic steatohepatitis. Expert Rev Gastroenterol Hepatol. 2011; 5:189–200. PMID: 21476914.
Article11. Muniandy K, Gothai S, Badran KM, Suresh Kumar S, Esa NM, Arulselvan P. Suppression of proinflammatory cytokines and mediators in LPS-induced RAW 264.7 macrophages by stem extract of alternanthera sessilis via the inhibition of the NF-κB pathway. J Immunol Res. 2018; 2018:3430684. PMID: 30155492.
Article12. Madamanchi NR, Vendrov A, Runge MS. Oxidative stress and vascular disease. Arterioscler Thromb Vasc Biol. 2005; 25:29–38. PMID: 15539615.
Article13. Ho FM, Liu SH, Liau CS, Huang PJ, Lin-Shiau SY. High glucose-induced apoptosis in human endothelial cells is mediated by sequential activations of c-Jun NH(2)-terminal kinase and caspase-3. Circulation. 2000; 101:2618–2624. PMID: 10840014.14. Abe S, Obata Y, Oka S, Koji T, Nishino T, Izumikawa K. Chondroitin sulfate prevents peritoneal fibrosis in mice by suppressing NF-κB activation. Med Mol Morphol. 2016; 49:144–153. PMID: 26880269.
Article15. Bauerova K, Ponist S, Kuncirova V, Mihalova D, Paulovicova E, Volpi N. Chondroitin sulfate effect on induced arthritis in rats. Osteoarthritis Cartilage. 2011; 19:1373–1379. PMID: 21884808.
Article16. Melgar-Lesmes P, Garcia-Polite F, Del-Rey-Puech P, Rosas E, Dreyfuss JL, Montell E, Vergés J, Edelman ER, Balcells M. Treatment with chondroitin sulfate to modulate inflammation and atherogenesis in obesity. Atherosclerosis. 2016; 245:82–87. PMID: 26714044.
Article17. Cañas N, Valero T, Villarroya M, Montell E, Vergés J, García AG, López MG. Chondroitin sulfate protects SH-SY5Y cells from oxidative stress by inducing heme oxygenase-1 via phosphatidylinositol 3-kinase/Akt. J Pharmacol Exp Ther. 2007; 323:946–953. PMID: 17885094.18. Song YO, Kim M, Woo M, Baek JM, Kang KH, Kim SH, Roh SS, Park CH, Jeong KS, Noh JS. Chondroitin sulfate-rich extract of skate cartilage attenuates lipopolysaccharide-induced liver damage in mice. Mar Drugs. 2017; 15:E178. PMID: 28617322.
Article19. Hu SW, Tian YY, Chang YG, Li ZJ, Xue CH, Wang YM. Fucosylated chondroitin sulfate from sea cucumber improves glucose metabolism and activates insulin signaling in the liver of insulin-resistant mice. J Med Food. 2014; 17:749–757. PMID: 24949837.
Article20. Kim BH, Ahn SH, Choi BD, Kang SJ, Kim YL, Lee HJ, Oh MJ, Jung TS. In vivo evaluation of chondroitin sulfates from midduk (Styela clava) and munggae tunics (Halocynthia roretzi) as a cosmetic material. J Korean Soc Food Sci Nutr. 2004; 33:641–645.21. da Cunha AL, Aguiar JA, Correa da Silva FS, Michelacci YM. Do chondroitin sulfates with different structures have different activities on chondrocytes and macrophages? Int J Biol Macromol. 2017; 103:1019–1031. PMID: 28536017.
Article22. Baek JM, Kang KH, Kim SH, Noh JS, Jeong KS. A study on development of high functional materials producing technique using by-products from skate processing (1)-development of chondroitin sulfate materials using skate cartilages. J Environ Sci Int. 2016; 25:645–654.23. Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972; 18:499–502. PMID: 4337382.
Article24. Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 1979; 95:351–358. PMID: 36810.
Article25. Schmidt HH, Warner TD, Nakane M, Förstermann U, Murad F. Regulation and subcellular location of nitrogen oxide synthases in RAW264.7 macrophages. Mol Pharmacol. 1992; 41:615–624. PMID: 1373797.26. Dietschy JM. Dietary fatty acids and the regulation of plasma low density lipoprotein cholesterol concentrations. J Nutr. 1998; 128(2 Suppl):444S–448S. PMID: 9478045.
Article27. Ichimura M, Kawase M, Masuzumi M, Sakaki M, Nagata Y, Tanaka K, Suruga K, Tamaru S, Kato S, Tsuneyama K, Omagari K. High-fat and high-cholesterol diet rapidly induces non-alcoholic steatohepatitis with advanced fibrosis in Sprague-Dawley rats. Hepatol Res. 2015; 45:458–469. PMID: 24827559.
Article28. Kainuma M, Fujimoto M, Sekiya N, Tsuneyama K, Cheng C, Takano Y, Terasawa K, Shimada Y. Cholesterol-fed rabbit as a unique model of nonalcoholic, nonobese, non-insulin-resistant fatty liver disease with characteristic fibrosis. J Gastroenterol. 2006; 41:971–980. PMID: 17096066.
Article29. Bieghs V, Van Gorp PJ, Wouters K, Hendrikx T, Gijbels MJ, van Bilsen M, Bakker J, Binder CJ, Lütjohann D, Staels B, Hofker MH, Shiri-Sverdlov R. LDL receptor knock-out mice are a physiological model particularly vulnerable to study the onset of inflammation in non-alcoholic fatty liver disease. PLoS One. 2012; 7:e30668. PMID: 22295101.
Article30. Subramanian S, Goodspeed L, Wang S, Kim J, Zeng L, Ioannou GN, Haigh WG, Yeh MM, Kowdley KV, O'Brien KD, Pennathur S, Chait A. Dietary cholesterol exacerbates hepatic steatosis and inflammation in obese LDL receptor-deficient mice. J Lipid Res. 2011; 52:1626–1635. PMID: 21690266.
Article31. Han LK, Sumiyoshi M, Takeda T, Chihara H, Nishikiori T, Tsujita T, Kimura Y, Okuda H. Inhibitory effects of chondroitin sulfate prepared from salmon nasal cartilage on fat storage in mice fed a high-fat diet. Int J Obes Relat Metab Disord. 2000; 24:1131–1138. PMID: 11033981.
Article32. Wang J, Mazza G. Effects of anthocyanins and other phenolic compounds on the production of tumor necrosis factor α in LPS/IFN-γ-activated RAW 264.7 macrophages. J Agric Food Chem. 2002; 50:4183–4189. PMID: 12105943.
Article33. Kleemann R, Zadelaar S, Kooistra T. Cytokines and atherosclerosis: a comprehensive review of studies in mice. Cardiovasc Res. 2008; 79:360–376. PMID: 18487233.
Article34. Gil-Cardoso K, Ginés I, Pinent M, Ardévol A, Blay M, Terra X. Effects of flavonoids on intestinal inflammation, barrier integrity and changes in gut microbiota during diet-induced obesity. Nutr Res Rev. 2016; 29:234–248. PMID: 27841104.
Article35. Yuan Y, Naito H, Jia X, Kitamori K, Nakajima T. Combination of hypertension along with a high fat and cholesterol diet induces severe hepatic inflammation in rats via a signaling network comprising NF-κB, MAPK, and Nrf2 pathways. Nutrients. 2017; 9:E1018. PMID: 28906458.36. Anavi S, Eisenberg-Bord M, Hahn-Obercyger M, Genin O, Pines M, Tirosh O. The role of iNOS in cholesterol-induced liver fibrosis. Lab Invest. 2015; 95:914–924. PMID: 26097999.
Article37. Angeli JK, Cruz Pereira CA, de Oliveira Faria T, Stefanon I, Padilha AS, Vassallo DV. Cadmium exposure induces vascular injury due to endothelial oxidative stress: the role of local angiotensin II and COX-2. Free Radic Biol Med. 2013; 65:838–848. PMID: 23973752.
Article38. Stabler TV, Montell E, Vergés J, Huebner JL, Kraus VB. Chondroitin sulfate inhibits monocyte chemoattractant protein-1 release from 3T3-L1 adipocytes: a new treatment opportunity for obesity-related inflammation? Biomark Insights. 2017; 12:1177271917726964. PMID: 28890654.
Article39. Jomphe C, Gabriac M, Hale TM, Héroux L, Trudeau LÉ, Deblois D, Montell E, Vergés J, du Souich P. Chondroitin sulfate inhibits the nuclear translocation of nuclear factor-kappaB in interleukin-1β-stimulated chondrocytes. Basic Clin Pharmacol Toxicol. 2008; 102:59–65. PMID: 17983423.40. Kim HJ, Hwangbo MH, Lee JW, Im HG, Lee IS. Antioxidant effects of red ginseng powder on liver of benzo(α)pyrene-treated mice. Korean J Food Sci Technol. 2007; 39:217–221.
Article41. Lovlin R, Cottle W, Pyke I, Kavanagh M, Belcastro AN. Are indices of free radical damage related to exercise intensity. Eur J Appl Physiol Occup Physiol. 1987; 56:313–316. PMID: 3569239.
Article42. Niess AM, Dickhuth HH, Northoff H, Fehrenbach E. Free radicals and oxidative stress in exercise--immunological aspects. Exerc Immunol Rev. 1999; 5:22–56. PMID: 10519061.43. Carr AC, McCall MR, Frei B. Oxidation of LDL by myeloperoxidase and reactive nitrogen species: reaction pathways and antioxidant protection. Arterioscler Thromb Vasc Biol. 2000; 20:1716–1723. PMID: 10894808.44. Dahech I, Harrabi B, Hamden K, Feki A, Mejdoub H, Belghith H, Belghith KS. Antioxidant effect of nondigestible levan and its impact on cardiovascular disease and atherosclerosis. Int J Biol Macromol. 2013; 58:281–286. PMID: 23624165.
Article45. Furukawa S, Fujita T, Shimabukuro M, Iwaki M, Yamada Y, Nakajima Y, Nakayama O, Makishima M, Matsuda M, Shimomura I. Increased oxidative stress in obesity and its impact on metabolic syndrome. J Clin Invest. 2004; 114:1752–1761. PMID: 15599400.
Article46. Lee JY, Lee SH, Kim HJ, Ha JM, Lee SH, Lee JH, Ha BJ. The preventive inhibition of chondroitin sulfate against the CCl4-induced oxidative stress of subcellular level. Arch Pharm Res. 2004; 27:340–345. PMID: 15089041.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Glucosamine Sulfate and Chondroitin Sulfate for the Treatment of Osteoarthritis
- Chondroitin sulfate in Corneal Preaervation Media Assessed by monitoring the Transendothelial Electrical Potential Difference
- Measurement of Transendothelial Potential Difference to Evaluate the Chondroitin Sulfate Effect in TC-l99 Cornea Preservation Media
- Comparison of Intravesical Hyaluronic Acid, Chondroitin Sulfate, and Combination of Hyaluronic Acid-Chondroitin Sulfate Therapies in Animal Model of Interstitial Cystitis
- Effects of Glycosaminoglycan on the Growth of Human Gingival Fibroblast