Cancer Res Treat.
2020 Jan;52(1):117-127. 10.4143/crt.2019.092.
Pediatric Adenocarcinoma in Korea: A Multicenter Study
- Affiliations
-
- 1Department of Pediatric Surgery, Seoul National University Children’s Hospital, Seoul, Korea
- 2Department of Pediatric Surgery, Asan Medical Center Children’s Hospital, University of Ulsan College of Medicine, Seoul, Korea
- 3Department of Surgery, Haeundae Paik Hospital, Inje University College of Medicine, Busan, Korea
- 4Department of Surgery, Kyungpook National University Hospital, School of Medicine, Kyungpook National University, Daegu, Korea
- 5Department of Surgery, Chonbuk National University School of Medicine, Jeonju, Korea
- 6Department of Surgery, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
- 7Department of Pediatrics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
- 8Department of Pediatric Surgery, Seoul National University College of Medicine, Seoul, Korea
- 9Department of Pediatric Surgery, Seoul St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul, Korea
- 10Department of Pediatric Surgery, Inha School of Medicine, Incheon, Korea
- 11Department of Surgery, Ilsan Paik Hospital, Inje University, Goyang, Korea
- 12Department of Pediatric Surgery, Severance Children’s Hospital, Yonsei University College of Medicine, Seoul, Korea
- KMID: 2501207
- DOI: http://doi.org/10.4143/crt.2019.092
Abstract
- Purpose
Adenocarcinoma is an extremely rare malignancy in the pediatric population. Research regarding pediatric adenocarcinoma is very rare in Korea. This study aimed to investigate the clinical features of pediatric adenocarcinomas of various primary organ sites in Korea.
Materials and Methods
Pediatric patients under 18 years, diagnosed with adenocarcinoma of various sites between January 1995 and December 2016, were included. We retrospectively reviewed patient and tumor characteristics and calculated survival estimates, reported as 5-year survival rate and 95% confidence interval.
Results
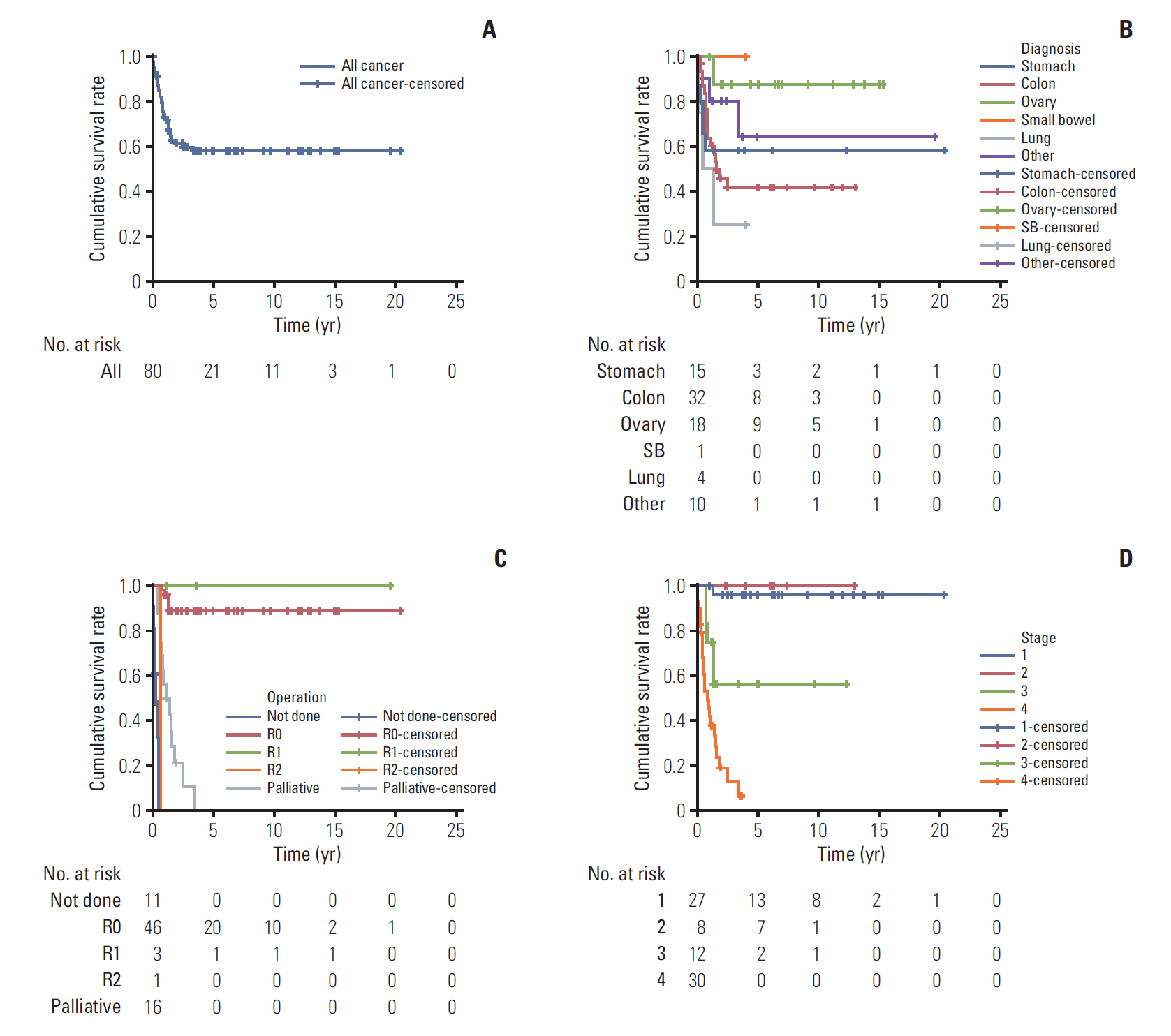
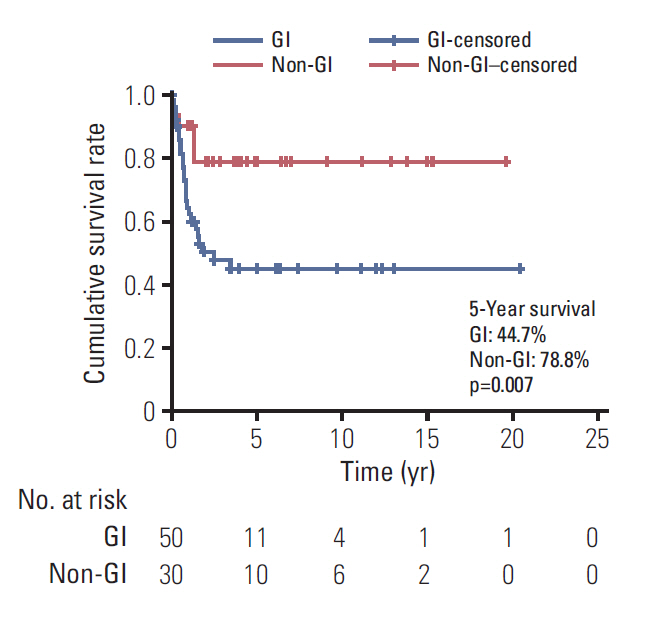
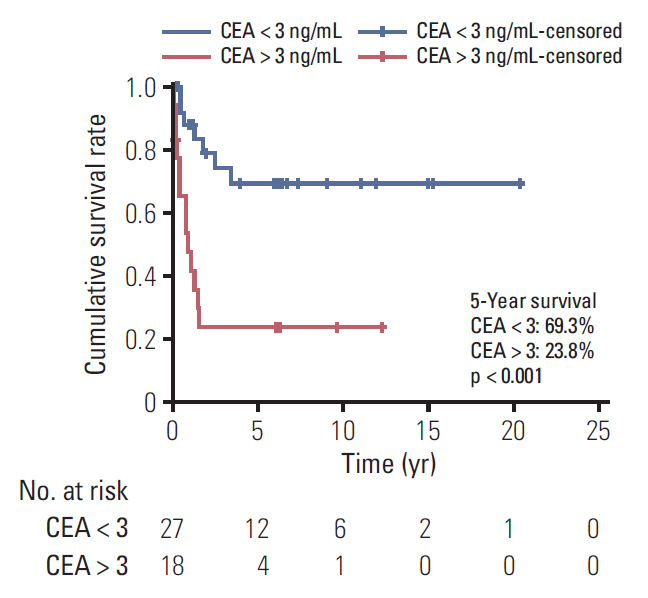
Of 80 patients (median age, 15 years; range, 10 to 17 years), 37 (46.3%) were men, and 24 (30%) had a family history of cancer or underlying disease relevant to malignancy. The cancer locations were the colon and rectum (n=32), ovaries (n=18), stomach (n=15), lung (n=4), small bowel (n=1), and other sites (n=10). Totally, 54.8% patients (42/77) had stage 3 or 4 disease. The median follow-up period was 2.0 years (range, 0 to 20.4). The 5-year overall survival estimate for all patients, and for those with stomach, colorectal, ovarian, and other cancer sites were 57.9%±11.5%, 58.2%±25.7%, 41.5%±18.2%, 87.5%±16.2%, and 64.0%±34.4%, respectively. The 5-year survival rate differed significantly between categories of adenocarcinomas into gastrointestinal (GI) (44.7%) and non-GI adenocarcinomas (78.8%) (p=0.007). The 5-year survival rate also differed significantly according to carcinoembryonic antigen level (69.3% in < 3 ng/mL, 23.8% in > 3 ng/mL; p < 0.001).
Conclusion
In pediatric patients, adenocarcinomas arise from various organs and are often diagnosed at advanced stages. Large, prospective studies for their accurate clinical characteristics and prognostic factors are needed.
Keyword
Figure
Reference
-
References
1. Kaatsch P. Epidemiology of childhood cancer. Cancer Treat Rev. 2010; 36:277–85.
Article2. Park HJ, Moon EK, Yoon JY, Oh CM, Jung KW, Park BK, et al. Incidence and survival of childhood cancer in Korea. Cancer Res Treat. 2016; 48:869–82.
Article3. Subbiah V, Varadhachary G, Herzog CE, Huh WW. Gastric adenocarcinoma in children and adolescents. Pediatr Blood Cancer. 2011; 57:524–7.
Article4. Chantada GL, Perelli VB, Lombardi MG, Amaral D, Cascallar D, Scopinaro M, et al. Colorectal carcinoma in children, adolescents, and young adults. J Pediatr Hematol Oncol. 2005; 27:39–41.
Article5. Durno C, Aronson M, Bapat B, Cohen Z, Gallinger S. Family history and molecular features of children, adolescents, and young adults with colorectal carcinoma. Gut. 2005; 54:1146–50.
Article6. Hatzaras I, Palesty JA, Abir F, Sullivan P, Kozol RA, Dudrick SJ, et al. Small-bowel tumors: epidemiologic and clinical characteristics of 1260 cases from the connecticut tumor registry. Arch Surg. 2007; 142:229–35.7. Menczer J, Sadetzki S, Murad H, Barda G, Andreev H, Barchana M. Childhood and adolescent ovarian malignant tumors in Israel: a nationwide study. Acta Obstet Gynecol Scand. 1999; 78:813–7.
Article8. Hill DA, Furman WL, Billups CA, Riedley SE, Cain AM, Rao BN, et al. Colorectal carcinoma in childhood and adolescence: a clinicopathologic review. J Clin Oncol. 2007; 25:5808–14.
Article9. Pratt CB, Rivera G, Shanks E, Johnson WW, Howarth C, Terrell W, et al. Colorectal carcinoma in adolescents implications regarding etiology. Cancer. 1977; 40(5 Suppl):2464–72.
Article10. Karnak I, Ciftci AO, Senocak ME, Buyukpamukcu N. Colorectal carcinoma in children. J Pediatr Surg. 1999; 34:1499–504.
Article11. Ferrari A, Rognone A, Casanova M, Zaffignani E, Piva L, Collini P, et al. Colorectal carcinoma in children and adolescents: the experience of the Istituto Nazionale Tumori of Milan, Italy. Pediatr Blood Cancer. 2008; 50:588–93.
Article12. Takeyama E, Tanaka M, Fujishiro J, Kitagawa N, Iwanaka T, Tanaka Y. A case of alpha-fetoprotein-producing gastric cancer in a child presenting with rupture of multiple liver metastases. Pediatr Surg Int. 2015; 31:885–8.
Article13. Al-Tonbary Y, Darwish A, El-Hussein A, Fouda A. Adenocarcinoma of the colon in children: case series and mini-review of the literature. Hematol Oncol Stem Cell Ther. 2013; 6:29–33.
Article14. Blumer SL, Anupindi SA, Adamson PC, Lin H, Price AP, Markowitz RI, et al. Sporadic adenocarcinoma of the colon in children: case series and review of the literature. J Pediatr Hematol Oncol. 2012; 34:e137–41.15. Park JA, Park HJ, Lee JS, Ha JO, Lee GK, Park BK, et al. Adenocarcinoma of lung in never smoked children. Lung Cancer. 2008; 61:266–9.
Article16. Pasternak BA, Kaistha A. Jejunal adenocarcinoma: a rare cause of chronic anemia in children. J Pediatr Gastroenterol Nutr. 2006; 42:100–3.
Article17. Michalek J, Kopecna L, Tuma J, Hrstkova H, Feit J. Gastric carcinoma in a 9-year-old boy. Pediatr Hematol Oncol. 2000; 17:511–5.18. Lamego CM, Torloni H. Colorectal adenocarcinoma in childhood and adolescent: report of 11 cases and review of the literature. Pediatr Radiol. 1989; 19:504–8.19. Goldthorn JF, Canizaro PC. Gastrointestinal malignancies in infancy, childhood, and adolescence. Surg Clin North Am. 1986; 66:845–61.
Article20. Maconi G, Kurihara H, Panizzo V, Russo A, Cristaldi M, Marrelli D, et al. Gastric cancer in young patients with no alarm symptoms: focus on delay in diagnosis, stage of neoplasm and survival. Scand J Gastroenterol. 2003; 38:1249–55.
Article21. MedlinePlus Medical Encyclopedia. How childhood cancers are different from adult cancers [Internet]. Bethesda, MD: U.S. National Library of Medicine;2019. [cited 2019 Apr 28]. Available from: https://medlineplus.gov/ency/patientinstructions/000845.htm.22. Fearon ER. Human cancer syndromes: clues to the origin and nature of cancer. Science. 1997; 278:1043–50.
Article23. Zoller ME, Rembeck B, Oden A, Samuelsson M, Angervall L. Malignant and benign tumors in patients with neurofibromatosis type 1 in a defined Swedish population. Cancer. 1997; 79:2125–31.
Article24. Shearer P, Parham D, Kovnar E, Kun L, Rao B, Lobe T, et al. Neurofibromatosis type I and malignancy: review of 32 pediatric cases treated at a single institution. Med Pediatr Oncol. 1994; 22:78–83.
Article25. Jo KM, Yang SY, Park JH, Kim TO, Jeong HJ, Heo CM, et al. Childhood colon cancer in a patient with ataxia telangiectasia. Ann Transl Med. 2016; 4:11.26. Majumdar SR, Fletcher RH, Evans AT. How does colorectal cancer present? Symptoms, duration, and clues to location. Am J Gastroenterol. 1999; 94:3039–45.
Article27. Esteva M, Leiva A, Ramos M, Pita-Fernandez S, GonzalezLujan L, Casamitjana M, et al. Factors related with symptom duration until diagnosis and treatment of symptomatic colorectal cancer. BMC Cancer. 2013; 13:87.
Article28. Sultan I, Rodriguez-Galindo C, El-Taani H, Pastore G, Casanova M, Gallino G, et al. Distinct features of colorectal cancer in children and adolescents: a population-based study of 159 cases. Cancer. 2010; 116:758–65.29. Deprest J, Moerman P, Corneillie P, Ide P. Ovarian borderline mucinous tumor in a premenarchal girl: review on ovarian epithelial cancer in young girls. Gynecol Oncol. 1992; 45:219–24.
Article30. Teng CJ, Hu YW, Chen SC, Yeh CM, Chiang HL, Chen TJ, et al. Use of radioactive iodine for thyroid cancer and risk of second primary malignancy: a nationwide population-based study. J Natl Cancer Inst. 2016; 108:djv314.
Article31. Rojas Y, Shi YX, Zhang W, Beierle EA, Doski JJ, Goldfarb M, et al. Primary malignant pulmonary tumors in children: a review of the national cancer data base. J Pediatr Surg. 2015; 50:1004–8.
Article32. Yoon HJ, Jeon TY, Yoo SY, Kim JH, Eo H, Lee SK, et al. Hepatic tumours in children with biliary atresia: single-centre experience in 13 cases and review of the literature. Clin Radiol. 2014; 69:e113–9.
Article33. Kulkarni PB, Beatty E Jr. Cholangiocarcinoma associated with biliary cirrhosis due to congenital biliary atresia. Am J Dis Child. 1977; 131:442–4.
Article34. Angel CA, Pratt CB, Rao BN, Schell MJ, Parham DM, Lobe TE, et al. Carcinoembryonic antigen and carbohydrate 19-9 antigen as markers for colorectal carcinoma in children and adolescents. Cancer. 1992; 69:1487–91.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Adenocarcinoma of the Urinary Bladder: Report of Three Cases
- An Overview of the Korean Intermittent Exotropia Multicenter Study by the Korean Association for Pediatric Ophthalmology and Strabismus
- A Case of Primary Jejunal Adenocarcinoma in Children
- Papillary gastric adenocarcinoma
- Prostatic Mucinous Adenocarcinoma