Nutr Res Pract.
2019 Oct;13(5):384-392. 10.4162/nrp.2019.13.5.384.
Effect of immune-enhancing enteral nutrition formula enriched with plant-derived n-3 fatty acids on natural killer cell activity in rehabilitation patients
- Affiliations
-
- 1National Leading Research Laboratory of Clinical Nutrigenetics/Nutrigenomics, College of Human Ecology, Yonsei University, Seoul 03722, Republic of Korea. jhleeb@yonsei.ac.kr
- 2Department of Food and Nutrition, College of Human Ecology, Yonsei University, Seoul 03722, Republic of Korea.
- 3Department of Rehabilitation Medicine, Nowon Eulji Medical Center, Eulji University, Seoul 01830, Republic of Korea.
- 4Department of Nutrition and Dietetics, Yonsei University Severance Hospital, Seoul 03722, Republic of Korea.
- 5Research Institute of Rehabilitation Medicine, Yonsei University College of Medicine, Seoul 03722, Republic of Korea.
- 6Department & Research Institute of Rehabilitation Medicine, Yonsei University College of Medicine, Seoul 03722, Republic of Korea. kimdy@yuhs.ac
- KMID: 2464117
- DOI: http://doi.org/10.4162/nrp.2019.13.5.384
Abstract
- BACKGROUND/OBJECTIVES
Enteral nutrition formulas with immune-enhancing nutrients, such as n-3 fatty acids, may manage patients' nutritional status and pathophysiological processes. The aim of our study was to investigate natural killer (NK) cell activity alterations and related cytokine changes resulting from feeding with soybean oil-containing enteral nutrition formula (control group) and plant-derived n-3 fatty acid-enriched enteral nutrition formula.
SUBJECTS/METHODS
Subjects participated for 14 consecutive days and consumed enteral formula containing canola and flaxseed oil (n3EN, test group) in nonsurgical patients hospitalized for rehabilitation. Blood samples were collected on the first day and 14 days after the consumption of each formula daily, and anthropometric parameters were collected. Hematology and biochemical values were analyzed, and NK cell activities and serum cytokine concentration were measured. A total of sixty subjects were included in the analysis, excluding dropouts.
RESULTS
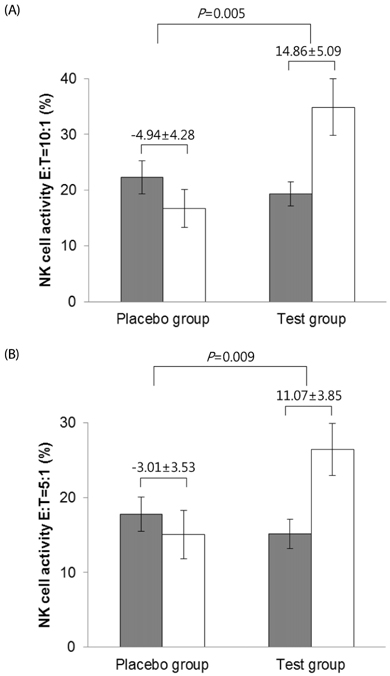
No significant differences were found in biochemical parameters. The n3EN group's NK cell activities at effector:tumor cell ratios of 10:1, 5:1, 2.5:1 and 0.625:1 were significantly higher than those of the control group after two weeks (P < 0.05). However, there were no statistically significant differences in serum cytokine interleukin (IL)-12, interferon-γ, IL-1β, IL-6 and tumor necrosis factor-α values between the two groups.
CONCLUSIONS
In conclusion, this study elucidates the beneficial effects of plant-derived n-3 fatty acid supplementation in enteral formula on NK cell activity.
MeSH Terms
Figure
Reference
-
1. Corrigan ML, Escuro AA, Celestin J, Kirby DF. Nutrition in the stroke patient. Nutr Clin Pract. 2011; 26:242–252.
Article2. Wirth R, Smoliner C, Jäger M, Warnecke T, Leischker AH, Dziewas R. DGEM Steering Committee. Guideline clinical nutrition in patients with stroke. Exp Transl Stroke Med. 2013; 5:14.
Article3. Liu H, Ling W, Shen ZY, Jin X, Cao H. Clinical application of immune-enhanced enteral nutrition in patients with advanced gastric cancer after total gastrectomy. J Dig Dis. 2012; 13:401–406.
Article4. Seike J, Tangoku A, Yuasa Y, Okitsu H, Kawakami Y, Sumitomo M. The effect of nutritional support on the immune function in the acute postoperative period after esophageal cancer surgery: total parenteral nutrition versus enteral nutrition. J Med Invest. 2011; 58:75–80.
Article5. Calder PC. Rationale and use of n-3 fatty acids in artificial nutrition. Proc Nutr Soc. 2010; 69:565–573.
Article6. Ruxton CH, Reed SC, Simpson MJ, Millington KJ. The health benefits of omega-3 polyunsaturated fatty acids: a review of the evidence. J Hum Nutr Diet. 2004; 17:449–459.
Article7. Hardin-Fanning F, Boissonneault GA, Lennie TA. Polyunsaturated fatty acids. Immunomodulators in older adults. J Gerontol Nurs. 2011; 37:20–28.8. Kim YS, Sayers TJ, Colburn NH, Milner JA, Young HA. Impact of dietary components on NK and Treg cell function for cancer prevention. Mol Carcinog. 2015; 54:669–678.
Article9. Beale RJ, Bryg DJ, Bihari DJ. Immunonutrition in the critically ill: a systematic review of clinical outcome. Crit Care Med. 1999; 27:2799–2805.
Article10. Heys SD, Walker LG, Smith I, Eremin O. Enteral nutritional supplementation with key nutrients in patients with critical illness and cancer: a meta-analysis of randomized controlled clinical trials. Ann Surg. 1999; 229:467–477.
Article11. Heyland DK, Novak F, Drover JW, Jain M, Su X, Suchner U. Should immunonutrition become routine in critically ill patients? A systematic review of the evidence. JAMA. 2001; 286:944–953.
Article12. Zheng Y, Li F, Qi B, Luo B, Sun H, Liu S, Wu X. Application of perioperative immunonutrition for gastrointestinal surgery: a meta-analysis of randomized controlled trials. Asia Pac J Clin Nutr. 2007; 16 Suppl 1:253–257.13. Ma C, Tsai H, Su W, Sun L, Shih Y, Wang J. Combination of arginine, glutamine, and omega-3 fatty acid supplements for perioperative enteral nutrition in surgical patients with gastric adenocarcinoma or gastrointestinal stromal tumor (GIST): A prospective, randomized, double-blind study. J Postgrad Med. 2018; 64:155–163.
Article14. Lochs H, Allison SP, Meier R, Pirlich M, Kondrup J, Schneider S, van den Berghe G, Pichard C. Introductory to the ESPEN guidelines on enteral nutrition: terminology, definitions and general topics. Clin Nutr. 2006; 25:180–186.
Article15. Snyderman CH, Kachman K, Molseed L, Wagner R, D'Amico F, Bumpous J, Rueger R. Reduced postoperative infections with an immune-enhancing nutritional supplement. Laryngoscope. 1999; 109:915–921.
Article16. Kudsk KA, Minard G, Croce MA, Brown RO, Lowrey TS, Pritchard FE, Dickerson RN, Fabian TC. A randomized trial of isonitrogenous enteral diets after severe trauma. An immune-enhancing diet reduces septic complications. Ann Surg. 1996; 224:531–543.17. Weimann A, Braga M, Carli F, Higashiguchi T, Hübner M, Klek S, Laviano A, Ljungqvist O, Lobo DN, Martindale R, Waitzberg DL, Bischoff SC, Singer P. ESPEN guideline: clinical nutrition in surgery. Clin Nutr. 2017; 36:623–650.
Article18. Sydnor ER, Perl TM. Hospital epidemiology and infection control in acute-care settings. Clin Microbiol Rev. 2011; 24:141–173.
Article19. Khanapure SP, Garvey DS, Janero DR, Letts LG. Eicosanoids in inflammation: biosynthesis, pharmacology, and therapeutic frontiers. Curr Top Med Chem. 2007; 7:311–340.
Article20. Massaro M, Habib A, Lubrano L, Del Turco S, Lazzerini G, Bourcier T, Weksler BB, De Caterina R. The omega-3 fatty acid docosahexaenoate attenuates endothelial cyclooxygenase-2 induction through both NADP(H) oxidase and PKCε inhibition. Proc Natl Acad Sci U S A. 2006; 103:15184–15189.
Article21. Thomas DR, Ashmen W, Morley JE, Evans WJ. Council for Nutritional Strategies in Long-Term Care. Nutritional management in long-term care: development of a clinical guideline. J Gerontol A Biol Sci Med Sci. 2000; 55:M725–M734.
Article22. Bankhead R, Boullata J, Brantley S, Corkins M, Guenter P, Krenitsky J, Lyman B, Metheny NA, Mueller C, Robbins S, Wessel J. A.S.P.E.N. Board of Directors. Enteral nutrition practice recommendations. JPEN J Parenter Enteral Nutr. 2009; 33:122–167.23. United States Department of Agriculture (USDA). Composition of Foods Raw, Processed, Prepared - USDA National Nutrient Database for Standard Reference, Release 28. Beltsville (MD): USDA;2005.24. World Health Organization. Global Database on Body Mass Index: an Interactive Surveillance Tool for Monitoring Nutrition Transition. Geneva: WHO;2012.25. Sprecher H. The roles of anabolic and catabolic reactions in the synthesis and recycling of polyunsaturated fatty acids. Prostaglandins Leukot Essent Fatty Acids. 2002; 67:79–83.
Article26. Yaqoob P, Calder PC. Fatty acids and immune function: new insights into mechanisms. Br J Nutr. 2007; 98 Suppl 1:S41–S45.
Article27. Calder PC. The relationship between the fatty acid composition of immune cells and their function. Prostaglandins Leukot Essent Fatty Acids. 2008; 79:101–108.
Article28. Weiss G, Meyer F, Matthies B, Pross M, Koenig W, Lippert H. Immunomodulation by perioperative administration of n-3 fatty acids. Br J Nutr. 2002; 87 Suppl 1:S89–S94.
Article29. Barrett AH, Porter WL, Marando G, Chinachoti P. Effect of various antioxidants, antioxidant levels, and encapsulation on the stability of fish and flaxseed oils: assessment by fluorometric analysis. J Food Process Preserv. 2011; 35:349–358.
Article30. Kolanowski W, Laufenberg G. Enrichment of food products with polyunsaturated fatty acids by fish oil addition. Eur Food Res Technol. 2006; 222:472–477.
Article31. Kolanowski W, Swiderski F, Berger S. Possibilities of fish oil application for food products enrichment with omega-3 PUFA. Int J Food Sci Nutr. 1999; 50:39–49.
Article32. Moretta A, Bottino C, Mingari MC, Biassoni R, Moretta L. What is a natural killer cell? Nat Immunol. 2002; 3:6–8.
Article33. Forel JM, Chiche L, Thomas G, Mancini J, Farnarier C, Cognet C, Guervilly C, Daumas A, Vély F, Xéridat F, Vivier E, Papazian L. Phenotype and functions of natural killer cells in critically-ill septic patients. PLoS One. 2012; 7:e50446.
Article34. Calder PC. Long-chain n-3 fatty acids and inflammation: potential application in surgical and trauma patients. Braz J Med Biol Res. 2003; 36:433–446.
Article35. Wada M, DeLong CJ, Hong YH, Rieke CJ, Song I, Sidhu RS, Yuan C, Warnock M, Schmaier AH, Yokoyama C, Smyth EM, Wilson SJ, FitzGerald GA, Garavito RM, Sui X, Regan JW, Smith WL. Enzymes and receptors of prostaglandin pathways with arachidonic acid-derived versus eicosapentaenoic acid-derived substrates and products. J Biol Chem. 2007; 282:22254–22266.
Article36. Souza-Fonseca-Guimaraes F, Adib-Conquy M, Cavaillon JM. Natural killer (NK) cells in antibacterial innate immunity: angels or devils? Mol Med. 2012; 18:270–285.
Article37. Narni-Mancinelli E, Jaeger BN, Bernat C, Fenis A, Kung S, De Gassart A, Mahmood S, Gut M, Heath SC, Estellé J, Bertosio E, Vely F, Gastinel LN, Beutler B, Malissen B, Malissen M, Gut IG, Vivier E, Ugolini S. Tuning of natural killer cell reactivity by NKp46 and Helios calibrates T cell responses. Science. 2012; 335:344–348.
Article38. Kreymann KG, Berger MM, Deutz NE, Hiesmayr M, Jolliet P, Kazandjiev G, Nitenberg G, van den Berghe G, Wernerman J, Ebner C, Hartl W, Heymann C, Spies C. DGEM (German Society for Nutritional Medicine). ESPEN (European Society for Parenteral and Enteral Nutrition). ESPEN guidelines on enteral nutrition: intensive care. Clin Nutr. 2006; 25:210–223.
Article39. Drewry AM, Ablordeppey EA, Murray ET, Beiter ER, Walton AH, Hall MW, Hotchkiss RS. Comparison of monocyte human leukocyte antigen-DR expression and stimulated tumor necrosis factor alpha production as outcome predictors in severe sepsis: a prospective observational study. Crit Care. 2016; 20:334.
Article40. Kjaergaard AG, Nielsen JS, Tønnesen E, Krog J. Expression of NK cell and monocyte receptors in critically ill patients--potential biomarkers of sepsis. Scand J Immunol. 2015; 81:249–258.
Article41. Hall MW, Knatz NL, Vetterly C, Tomarello S, Wewers MD, Volk HD, Carcillo JA. Immunoparalysis and nosocomial infection in children with multiple organ dysfunction syndrome. Intensive Care Med. 2011; 37:525–532.
Article42. Hall MW, Geyer SM, Guo CY, Panoskaltsis-Mortari A, Jouvet P, Ferdinands J, Shay DK, Nateri J, Greathouse K, Sullivan R, Tran T, Keisling S, Randolph AG. Pediatric Acute Lung Injury and Sepsis Investigators (PALISI) Network PICFlu Study Investigators. Innate immune function and mortality in critically ill children with influenza: a multicenter study. Crit Care Med. 2013; 41:224–236.
Article43. Lee JG, Kim YS, Lee YJ, Ahn HY, Kim M, Kim M, Cho MJ, Cho Y, Lee JH. Effect of immune-enhancing enteral nutrition enriched with or without beta-glucan on immunomodulation in critically ill patients. Nutrients. 2016; 8:E336.
Article44. Lee DH, Kim M, Kim M, Lee YJ, Yoo HJ, Lee SH, Lee JH. Age-dependent alterations in serum cytokines, peripheral blood mononuclear cell cytokine production, natural killer cell activity, and prostaglandin F2α. Immunol Res. 2017; 65:1009–1016.
Article45. Flick DA, Gifford GE. Pharmacokinetics of murine tumor necrosis factor. J Immunopharmacol. 1986; 8:89–97.
Article46. Hayes KC, Hull TC, Delaney GA, Potter PJ, Sequeira KA, Campbell K, Popovich PG. Elevated serum titers of proinflammatory cytokines and CNS autoantibodies in patients with chronic spinal cord injury. J Neurotrauma. 2002; 19:753–761.
Article47. Agarwal N, Chitrika A, Bhattacharjee J, Jain SK. Correlation of tumour necrosis factor-α and interleukin-6 with anthropometric indices of obesity and parameters of insulin resistance in healthy north Indian population. J Indian Acad Clin Med. 2011; 12:196–204.48. Jason J, Archibald LK, Nwanyanwu OC, Byrd MG, Kazembe PN, Dobbie H, Jarvis WR. Comparison of serum and cell-specific cytokines in humans. Clin Diagn Lab Immunol. 2001; 8:1097–1103.
Article49. Nordenström J, Carpentier YA, Askanazi J, Robin AP, Elwyn DH, Hensle TW, Kinney JM. Metabolic utilization of intravenous fat emulsion during total parenteral nutrition. Ann Surg. 1982; 196:221–231.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Sources and Formulation of Macro- and Micro-Nutrients in Enteral Nutrition Formula
- A Study of Natural Killer Cell Activities , T Cells and T Cell Subsets in Vitiligo
- Enteral Nutrition and Its Clinical Application
- Involvement of Hepatic Innate Immunity in Alcoholic Liver Disease
- Association between serum fatty acid composition and innate immune markers in healthy adults